Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Ciencia del suelo
versión On-line ISSN 1850-2067
Cienc. suelo v.25 n.1 Buenos Aires ene./jul. 2007
The Rietveld method applied to the quantitative mineralogical analysis of some soil samples from Argentina
Stella M. Zabala; María S. Conconi; Margarita Alconada1 & Rosa M. Torres Sánchez*
Centro de Tecnología de Recursos Minerales y Cerámica (CETMIC), Cno. Centenario y 506, CC 49 (B1896ZCA) M.B. Gonnet. Argentina. e-mail: rosats@netverk.com.ar
1UNLP, Fac. Cs. Agrarias y Forestales, Cat. Edafología. C/60 y 119. (1900) La Plata; CISAUA. (MAA-UNLP) Calle 3 N° 584. (1900) La Plata
(*) corresponding author.
Recibido: 11/09/06
Aceptado: 08/05/07
ABSTRACT
The mineralogical quantification of soils species is mainly obtained by analysis of X-ray diffraction (XRD) patterns. The classic method, for these analyses, is limited to semi quantitative determinations due to the overlapping of different specie's peaks and processes of absorption of the constituents, the wide range of particle size distribution, etc.
The use of the Rietveld method for XRD quantitative analysis, in mixtures of minerals, has allowed to improve the accuracy of the quantitative results and to extend it to complex systems, as soils, transforming it in a meaningful tool for soils investigation.
In this work, quantitative results obtained by the application of classic and Rietveld methods, are compared in different soils samples from Argentina. A set of mixtures of minerals with similar composition to three studied samples, was also quantified by Rietveld method, with absolute error lower than 3%. Obtained results indicate that quantitative analysis by Rietveld method, can improve the results found by XRD classic method and may be used for the quantitative determination of soil minerals.
Key words. Soils; Quantification of minerals; X-ray diffraction; Rietveld method.
Aplicación del método de Rietveld al análisis mineralógico cuantitativo de algunas muestras de suelos de la Argentina
RESUMEN
La cuantificación mineralógica de componentes del suelo es obtenida principalmente por análisis de espectros de difracción de rayos X (DRX). El método clásico aplicado para dicho análisis está limitado a determinaciones semi-cuantitativas debido a la superposición de picos de las distintas especies y procesos de absorción de los constituyentes, el amplio rango de distribución de tamaño de partícula, etc.
El uso del método de Rietveld para el análisis cuantitativo por DRX, en mezclas de minerales, ha permitido mejorar la exactitud de los resultados cuantitativos y extenderlo a sistemas complejos, como lo son los suelos, transformándose en una herramienta significativa para su investigación.
En este trabajo se comparan los resultados cuantitativos obtenidos por la aplicación del método clásico y de Rietveld, a diferentes muestras de suelos de la Argentina. Un grupo de mezclas de minerales con composición similar a tres de los suelos estudiados, también se cuantificó por el método de Rietveld, obteniéndose un error absoluto menor al 3%. Los resultados indican que el análisis cuantitativo por el método de Rietveld, mejoran los resultados obtenidos por el método clásico de DRX y puede ser usado para la determinación cuantitativa de los minerales presentes en suelos.
Palabras clave. Suelos; Cuantificación de minerales; Difracción de rayos X; Método de Rietveld.
INTRODUCTION
Detailed information on the soil constituents and their chemical state can be obtained by means of a great diversity of spectroscopic and electronic microscopy techniques (Fendorf & Sparks, 1996). However quantification of crystalline components in the fine fraction continues being limited to the analysis of each specie's peaks from the X rays diffraction (XRD) patterns.
The first limitation in the XRD classic method (which requires mineral standards with XRD properties similar to those of the mineral phases in the unknown sample), is the heterogeneous nature of clays in soils.
Several steps were used to improve the accuracy of this method. Preliminary treatments of sample preparation as dispersion of particles, removal of organic matter, etc. (Bish, 1994; Whittig & Allardice, 1986) were used to disperse the sample and to remove crystalline solids whose diffraction peaks are superimposed interfering in the determination and/or quantification of certain minerals. Also, separation of different particle fractions (<2-0,2 mm by Stokes's law) was managed to improve the quantitative results obtained by XRD classic method.
The overlapping of peaks, which also contribute to the inaccuracy of the XRD classic method, can be eliminated using single line fitting procedure (Naidu & Houska, 1982), peak deconvolutions or using differential XRD; the high absortivity (that can cause error higher than double of the real value) can be overcome by changing the radiation source (Cu to Mo or Co, etc.; Brindley, 1961) and the use of external standard technique would decrease the technique error to [always higher than 7%, (Pawloski, 1985)]. Generally, to improve the results furnished by XRD quantitative analysis, it is necessary to use other spectroscopy (Mössbauer, electronic Microscopy, etc.) or chemical analysis of the samples.
Hugo Rietveld (1969) developed a method to refine crystalline structures using neutron diffraction data. The utilization of Rietveld's method to XRD has advanced quickly, extending its use to structural analysis (McCuster et al., 1999), crystalline perfection investigation, reticular parameters measurement, phase transformations (Bish & Howard, 1988; Pascoal et al., 1999; Ortiz et al., 2000), determination of amorphous content (De la Torre et al., 2001) and to quantitative analysis of mineral mixtures (Jones & Bish, 1991; Taylor & Rui, 1992; Bish & Post, 1993; Raudsepp et al., 1999).
This method fits point-to-point the difference between experimental intensities of the whole X-ray pattern and the calculated intensities, based on a certain model of crystalline structure, optic effects of diffraction, instrumental factors and other characteristics of the sample. All lines for each phase are included explicitly in the refinement, and linear background is obtained when a good correlation between the calculated and experimental X-ray patterns exists. Among others, the advantages over traditional methods are reduction of effects of preferred orientation, calibration without use of internal or external standards, separately handling of the overlapped reflections and mass absorption effects, etc.
The Rietveld method has shown an appropriate correspondence in diverse applications in soil studies. It has been used in the determination of clays in soils (Weidleret al.,1998; Bravoet al.,2003) and in coal (Chuande & Ward, 2003), of mixtures of minerals (Bonetto et al., 2003), of montmorillonites quantification in soils (Dermatas & Dadachov, 2003) and in the adjustment among experimental and calculated values of densities of surface soil charges (Taubaso et al., 2004).
The objective of this work is to use the Rietveld method for quantitative analysis of the mineral composition of some soils samples from Argentina of different origin and composition. Some soils were also evaluated by a classic XRD method and also different radiation source was used for two soils with high iron content. Also, three different measurements and refinements were carried out on artificial mixtures of similar mineral composition of three studied soils samples.
MATERIALS AND METHODS
Samples of surface horizon (A) of all soils were used. Three of them belong to the Pampean region (a Typic Ustipsamment, a Typic Hapludert and a Typic Argiudoll, named Anguil, Gorina and Castelar, respectively), four other samples belong to soils from a subtropical zone (Rhodudult, identified as S. Jose, C. Sierra and C. Azul and Udult, C. Navaja) and the last one is the surface horizon of a soil in a cold area near the Cordillera de los Andes (Typic Haplocryoll, Chalten).
The references used for classic XRD analysis were: Quartz, (99.9%, particle size <44 µm, Argentina), Montmorillonite (99% sodium Montmorillonite, particle size <2 µm, Neuquén), Illite (85% and 15% quartz, particle size <74µm, Illinois, USA), Kaolinite (98%, particle size <2µm, Georgia, USA), Magnetite (97.5% Fe3O4, Argentina), Hematite (99% Fe2O3, analytical grade BDH), Anatase (TiO2 analytical grade Fluka), two feldspar were used: Albite (99%, particle size <74µm, Argentina) and Sanidine (99%, particle size <74µm, VASA), and micronized Fluorite (99.9%, Argentina) obtained by contrition milling for 15 hr (80% <2mm) as amorphous.
The soil samples were manually crushed in a porcelain mortar and sieved (<1mm) to eliminate plant residues and gravel, then leached with distilled water and finally air-dried. In all soils except Chalten, due to its low OM content, the organic matter was removed by treating the sample with 30% w/w H2O2, at 80 °C under constant stirring (Kunze & Dixon, 1986); the OM remaining in the samples after the treatment was determined following the procedure of Richter & Von Wistinghausen (1981). The obtained samples were crushed, sieved (<125 µm) and used for the XRD analysis. They will be called hereafter with their origin name (Anguil, Castelar, C. Sierra, etc.).
To simulate soil composition, mixtures of same minerals in similar amounts to those of soil chosen were homogenized, mixing them in an agate mortar for 5 minutes with acetone until its evaporation.
A Philips 3020 apparatus was used to record XRD patterns. The operating conditions were: 40kV and 30mA, Cuk Á radiation, Ni filter. Diffraction data were collected over a 2q range of 3°-70°, with a step width of 0.02° and a counting time of 2.0 sec/ step. The samples S. José and C. Sierra were also analyzed, under the same working conditions, in a Philips PW1810 with Co radiation. All samples were analysed by means of the powder mounting technique.
The classic XRD method for quantitative determination of the different species was carried out on Castelar, Anguil, C. Sierra, S. Jose, C. Navaja and C. Azul samples. The identification and quantification of montmorillonite was carried out on samples treated with glycol. The peaks used to measure the intensities were: Quartz, 3.36 Å (Wilding et al., 1977); Montmorillonite, 17.0 Å (Borchardt, 1977); Kaolinite, 7.00 Å (Dixon, 1977), Anatase, 3.52 Å (Hutton, 1977), Hematite, 2.70 Å and Magnetite, 2.97Å (Schwertmann & Taylor, 1977); Illite, 9.3 Å (Fanning & Keramidas, 1977) and Feldspar, 3.30 Å (Huang, 1977). The content of each species was determined by the ratio of the indicated peak area in the sample with respect to that of the reference, same crystalline species in pure condition, expressed as percentage. The absorptive properties of the soil components were considered by correcting the values of the obtained areas (I) using the mass adsorption coefficients (m) of each component.
Ix/Io=(mx/mo) xi (1)
Where: the subscripts x ando correspond to crystalline species in the sample and in the standard, respectively; while xi is the mass fraction of the species in the sample.
The Rietveld method, as was indicated, fits point-to-point the experimental intensities of the whole pattern to those calculated. The "Profile Matching" refinement mode included in the programs "FULLPROF" (Rodríguez Caravajal, 1990), was used in the present work and allows the refinement and subsequent quantitative analysis to be made even when some phases may have incomplete structural data (Bonetto et al., 2003). The starting crystallographic data used for each phase were extracted from the literature. For montmorillonite quantification, the fitting was carried out without taking into account the atomic positions. The structure factors were then calculated based on the measured intensities of a sample containing 99% of sodium montmorillonite (Lombardi et al., 2002) whose pattern match with that present in the samples. These structure factors were used to calculate the integrated intensities of the sample under study, whereas other parameters as preferred orientation, profile function parameters, asymmetry, etc. were refined. The illitic material was adjusted as 1M mica. As Rietveld analysis does not take into account the presence of phases not detected by XRD and amorphous materials, micronized fluorite was added as internal standard to quantify these components (Madsen et al., 1991). Quartz present in the samples was used to fit the background and the 2q shift for the displacement of each specimen.
RESULTS AND DISCUSSION
Table 1 shows the iron and the organic matter content, specific surface and particle size after H2O2 treatment of the soil samples.
Table 1. Iron oxide and organic matter contents, specific surface area and particle size composition after H2O2 treatment. Values from (#) Taubaso et al. (2003) and (*) Torres Sánchez et al. (2001).
Tabla 1. Contenido en óxido de hierro y materia orgánica, superficie específica y composición granulométrica después del tratamiento del suelo con H2O2. Los valores indicados con (#) corresponden a Taubaso et al. (2003) y (*) Torres Sánchez et al. (2001).
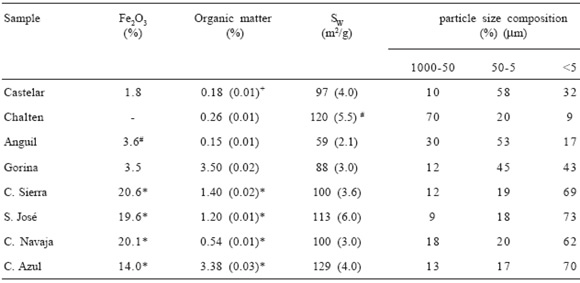
The high iron content in C. Sierra, S. Jose, C. Navaja and C. Azul samples denotes its origin and the high clay content is reflected in the measured values of surface area. Surface area values of all samples are in agreement with those found by Torres Sánchez & Falasca (1997). The high surface area value obtained for Chalten sample can not be directly correlated to any organic matter or clay content.
Figures 1 to 4 show the observed and calculated patterns for Castelar, Anguil, C. Sierra and S. Jose.

Figure 1. Observed (+) and calculated (line) patterns for Castelar. Crystalline species Bragg position lines. The lower curve shows the difference between observed and calculated patterns (Rwp = 35.0%, Rexp = 15.0%).
Figura 1. Difractogramas experimental (+) y calculado (línea) para el suelo Castelar. Posiciones de las líneas de Bragg de lasespecies cristalinas. La curva inferior muestra la diferencia entre los difractogramas experimental y calculado (Rwp = 35.0%, Rexp = 15.0%).
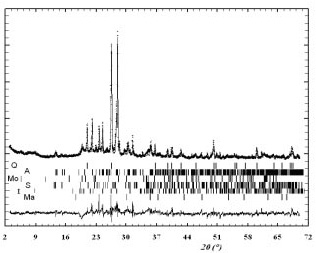
Figure 2. Observed (+) and calculated (line) patterns for Anguil. Crystalline species Bragg position lines. (Rwp = 35.1%, Rexp = 16.3%).
Figura 2. Difractogramas experimental (+) y calculado (línea) para el suelo Anguil. Posiciones de las líneas de Bragg de las especies cristalinas. (Rwp = 35.1%, Rexp = 16.3%).

Figure 3. Observed (+) and calculated (line) patterns for C. Sierra.. Crystalline species Bragg position lines. (Rwp = 30.3%, Rexp = 20.6%).
Figura 3. Difractogramas experimental (+) y calculado (línea) para el suelo C. Sierra. Posiciones de las líneas de Bragg de las especies (Rwp = 30.3%, Rexp = 20.6%).

Figure 4. Observed (+) and calculated (line) patterns for S. Jose. Crystalline species Bragg position lines. (Rwp = 29.6%, Rexp = 20.2%).
Figura 4. Difractogramas experimental (+) y calculado (línea) para el suelo S. José. Posiciones de las líneas de Bragg de las especies (Rwp = 29.6%, Rexp = 20.2%).
The difference between observed and calculated patterns (Figures 1 and 2) was significant from around 23 to 30° (2q) where the relative moisture of clay samples play an important role (Runpinget al.,2003). Soil samples with high iron content (Figures 3 and 4) as will be discussed latter, showed a better fit than that obtained for the other soils. The best least square fit between observed and calculated patterns was indicated by Rwp, weighted pattern R-factor, which indicates the quality of the fitting procedure, and Rexp values, R-Bragg factor: which indicates the correspondence between the calculated and the measured diffraction intensities of individual phases. The Rwp for all samples were within 29-35%, which is typically adequate in X-ray refinements (Young, 1993) and Rexp values were close to Rwp in soils C. Sierra and S. José and also similar to those obtained by Bish & Post (1993) and Bonetto et al. (2003) for mechanical mixtures of different oxides.
Table 2 summarizes the percentage values obtained for each species, by classic and Rietveld methods. Feldspar was obtained as the percentage addition of Albite and Sanidine species. Analysis of fraction < 2µm for Gorina, correction for the presence of amorphous species and estimated standard deviations for all soils were also included for the Rietveld method analysis in Table 2.
Table 2. Weight percentage of minerals obtained by the XRD Classic and the Rietveld methods for the indicated soils. Letters indicate: Q, Quartz, Mo, Montmorillonite, F, Feldspar (obtained as addition of Sanidine and Albite), K, Kaolinite, I, Illite, Ma, Magnetite, H, Hematite, T, Anatase and Amor, Amorphous. (1) values from Torres et al. (2001).
Tabla 2. Porcentaje en peso de minerales obtenidos por el método Clásico de DRX y Rietveld, para los suelos indicados. Las letras indican Q, Cuarzo, Mo, Montmorillonita, F, Feldespato (como suma de Sanidina y Albita), K, Caolinita, I, Illita, Ma, Magnetita, H, Hematita, T, Anatasa y Amor, Amorfo. (1) valores de Torres et al. (2001).
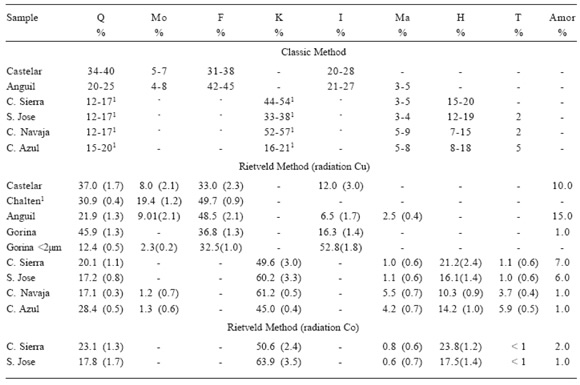
Gorina is classified as Typic Hapludert with the characteristic of a clay-rich soil that shrinks and swells with changes in moisture content. The absence of montmorillonite or expansive clays and the high illite content in Gorina, could be assigned to a smectite-to-illite transition (Moore & Reynolds, 1989) and stacking disordersof clays (Bish, 1993). The different values found for illite in Gorina and Gorina < 2 µm follows that indicated by Laird & Dowdy (1994) mineralogical composition is independent of particle size for quartz and kaolinite, however, this assumption is not valid for the illitic materials in soils.
The precision of Rietveld results (absolute error lower than 3%) was in all species higher than that obtained by the classical method, but one must remember that the standard deviation for the refined Rietveld parameters is related to their precision rather than to their accuracy; thus, accurate estimation of the errors in the quantitative species-composition analysis requires the use of standard mixtures.
An approach to evaluate the accuracy of the Rietveld method in samples of high iron content was to calculate the quartz and kaolinite percentage from the chemical analysis data [Al2O3%and SiO2% in C. Sierra were20.7 and 42.3%, respectively, while their corresponding values for S. Jose were 24.2 and 42.0% (Torres et al.,2001)]. For this purpose the Al2O3 amount was considered to come only from the kaolinite structure, so the quartz proportion was calculated as the difference of total SiO2 and kaolinite. Amounts calculated of kaolinite were of 50% in C. Sierra and 59% in S. Jose, while values of quartz in the same materials were 18 and 15%, respectively. The data differ 2% in kaolinite and quartz with respect to data obtained by the Rietveld method. The difference obtained for quartz was assigned to the inadequate hypothesis that all analytically determined SiO2 was crystalline, in agreement with data obtained by Bish & Post (1993) for hematite and corundum mixtures.
By comparing the classical and the Rietveld method when applied to data obtained for the Castelar sample (Table 2) it is found that the values obtained with the first methods were within the XRD experimental error (± 7%) and with the Rietveld were ± 3% for all components.
The iron percentage obtained by the Rietveld method agreed with that found by chemical analysis (Table 1). For C. Navaja and C. Azul, it must be taken into account that some 20% of the total iron was in the kaolinite structure as isomorphic substitution (Torres et al., 2001). Data obtained with Co anode agreed within the method error found with Cu anode in the two samples (C. Sierra and S. Jose) taken as example for comparison.
Table 3 shows the nominal values in wt% of crystalline species identified in the oxides mixtures with similar composition of C. Sierra, S. Jose and Castelar.
Table 3. Nominal values in wt% of crystalline species determined by Rietveld method in the oxides mixtures with similar composition of C. Sierra, S. Jose and Castelar. Letters indicate same minerals as in Table 2.
Tabla 3.Valores nominales de porcentaje en peso de las especies cristalinas, determinados por el método de Rietveld, en mezclas de óxidos con composición similar a los suelos C. Sierra, S. José y Castelar. Las letras indican los mismos minerales que en la Tabla 2.

Quartz amount obtained by Rietveld method was slightly higher than that used in the oxide mixtures of similar composition of C. Sierra and S. Jose evaluated, while percentage of kaolinite, anatase and iron oxides were always lower than those of the nominal values used. It is important to mention the lower montmorillonite value (0.8%) obtained by Rietveld method in comparison to that of the mechanical mixture (8.0%), which point out the difficulty to identify expansible minerals. The concentrations obtained for the oxide mixtures show that the absolute errors do not exceed 3wt %.
CONCLUSIONS
An accurate and rapid quantification of minerals in soil samples is a long standing problem in soil science. The good results obtained with XRD techniques on oxide mixtures and on soil samples of different composition, confirm the usefulness of the profile matching mode for mineral quantification in soils. The Rietveld quantitative analysis applied to soil samples provides lower errors than those obtained by the classical method XRD (without requiring laborious sample pre-treatment).
Also, no significant difference in quantitative analysis was obtained between the uses of Cu or Co anode in high iron content samples.
The reference mixture measured and quantified 3 times demonstrates that the average error values obtained and the error values from a single refinement are similar.
The presence of amorphous soil phases must be taken in consideration to determine the percentage of soil minerals.
REFERENCES
1. Bish, DL. 1994. Quantitative XRD analysis of soils. Pp.267-295 In: (eds. JE Amonette & L Zelazny). Quantitative methods in soil mineralogy. S.S.S.A. Misc. Publ. Madison, WI. [ Links ]
2. Bish, DL. 1993. Studies of clays and clay minerals using XRD and the Rietveld Method. Pp.79-121 In: (eds. R Reynolds & J Walker) CMS workshop lectures. Computer applications to XRD powder analysis of clay Minerals. Vol. 5. The clay Min. Soc. Boulder, Colorado. [ Links ]
3. Bish, DL. & JE Post. 1993. Quantitative mineralogical analysis using the Rietveld full-pattern fitting method. American Mineralogist 78: 932-940. [ Links ]
4. Bish, DL. & SJ Howard. 1988. Quantitive phase analysis using the Rietveld method. J. Appl. Cristallography 21: 86-91. [ Links ]
5. Bonetto, RD; PE Zalba; MS Conconi & M Manassero. 2003. The Rietveld method applied to quantitative phase analysis of minerals containing disordered structures.Rev. Geologica de Chile 30(1): 103-115. [ Links ]
6. Borchardt, GA. 1977. Montmorillonite and other smectite Minerals. Pp. 296 In: (eds. J Dixon & S Weed.). Minerals in Soil Environment. S.S.S.A. Misc. Publ. Madison, WI. [ Links ]
7. Bravo, C; A Rodríguez; L Saavedra; Q Trujillo; S Mejia & V Huaypar. Caracterización mineralógica de suelos agrícolas de Vicus, Región Piura, por DRX y espectroscopia Mössbauer. Rev. XIV Simposio Peruano de Física, 2003. [ Links ]
8. Brindley, GW. 1961. Chlorite minerals Pp. 51-131 In: (ed. G Brown.). The X-ray identification and crystal structures of clay Minerals. Miner. Soc. London. [ Links ]
9. Chuan-De, R & C Ward. 2003. Quantitative X-ray powder diffraction analysis of clay minerals in Australian coals using Rietveld methods. Appl. Clay Sci. 21: 227-240. [ Links ]
10. De la Torres, AG; S Bruqueand; MAG Aranda. 2001. Rietveld quantitative amorphous content analyses. J. of Appl. Crystallog. 34: 196-202. [ Links ]
11. Dermatas, D & M Dadachov. 2003. Rietveld quantification of montmorillonites in lead contaminated soils. Appl. Clay Sci. 23: 245-255. [ Links ]
12. Dixon, JB. 1977. Kaolinite and serpentine group minerals Pp 392 In: (eds. J Dixon & S Weed) Minerals in Soil Environment. S.S.S.A. Misc. Publ. Madison, WI. [ Links ]
13. Fanning, DS & VZ Keramidas. 1977. Micas Pp. 208 In: (eds. J Dixon & S Weed) Minerals in Soil Environment. S.S.S.A. Misc. Publ. Madison, WI. [ Links ]
14. Fendorf, S & D Sparks. 1996. Chemical methods Ch. 13 Pp 377 In: (eds. D. Sparks & L Page.). Methods of soil analysis. Vol. 3 S.S.S.A. Misc. Publ. Madison WI. [ Links ]
15. Huang, PM. 1977. Feldspars, Olivines, Pyroxenes, and Amphiboles Pp. 553-593. In: (eds. J Dixon & S Weed) Minerals in Soil Environment. S.S.S.A. Misc. Publ. Madison, WI. [ Links ]
16. Hutton, J.T. 1977. Titanium and zirconium minerals Pp 685. In: (eds. J Dixon & S Weed) Minerals in Soil Environment. S.S.S.A. Misc. Publ. Madison, WI. [ Links ]
17. Jones, R & D Bish. 1991. Quantitative X-ray diffraction analysis of soils: Rietveld full pattern mineral concentration and pattern curve fitting vs. P sorption of bauxite soils. Proc. of the Amer. Clay Min. Soc. Meeting 28: 84-94. [ Links ]
18. Kunze, GW & JB Dixon. 1986. Pretreatment for mineralogical analysis Pp. 95-97 In: (ed. A Klute). Methods in soil analysis. Vol. 3. S.S.S.A. Misc. Publ. Madison WI. [ Links ]
19. Laird, D & R Dowdy. 1994. Simultaneous mineralogical quantification and chemical characterization of soil clays. Clay and Clay Min. 42(6): 747-754. [ Links ]
20. Lombardi, B.; M Baschini & RM Torres Sánchez 2002. Characterization of montmorillonites from bentonite deposits of North Patagonia, Argentina: physicochemical and structural parameter correlation. J. Arg. Chem. Soc. 90 (4-6): 87-99. [ Links ]
21. McCuster, LB.; RB Von Dreele; DE Cox; D Löuer & P Scardi. 1999. Rietveld refinement guidelines. J. of Appl. Crystallography 32: 36-50. [ Links ]
22. Madsen, IC; RJ Finney; CA Flann; MT Frost & BW Wilson. 1991. Quantitative analysis of high alumina refractories using Xray powder diffraction data and the Rietveld method. J. Am. Ceram. Soc. 74(3): 619-624. [ Links ]
23. Moore, D & R Reynolds. 1989. Clays Pp 150 In: (eds. D Moore & R Reynolds) XRD and identification and analysis of clay minerals. Oxford Univ. Press. Oxford. [ Links ]
24. Naidu, S & C Houska. 1982. Profile separation in complex powder patterns. J. Appl. Physisc 15: 190-198. [ Links ]
25. Ortiz, A; F Cumbrera; F Sánchez Bajo; F Guibertau; H Xu & N Padture. 2000. Quartz phase composition analysis of liquidphase sintered silicon carbide using the Rietveld method. J. Am. Ceram. Soc. 83(9): 2282-2286. [ Links ]
26. Pascoal, C; R Macado & V Pandeolfelli. 1999. Composicao quimica e mineralogica de bauxitas refratarias. Anais do 43 Congresso Brasileiro de Ceramica. Pp 8301-8314. [ Links ]
27. Pawloski, GA. 1985. Quantitative determination of mineral content of geological samples by X-ray diffraction. Amer. Mineralogist 70: 663-667. [ Links ]
28. Raudsepp, M; E Pani & G Diplle. 1999. Measuring mineral abundance in skarn. I Rietveld method using X-ray powder diffraction data. Canadian Min. 37: 1-15. [ Links ]
29. Richter, M & E Von Wistinghausen. 1981. Unster. Von humusfraktionen in Boden. Z. Pfla. Bodenkunde 144: 395-406. [ Links ]
30. Rietveld, HM. 1969. A profile refinement method for nuclear and magnetic structures. J. Appl. Crystal. 2: 65-71. [ Links ]
31. Rodríguez-Caravajal, J. 1990. Fullprof, a program for Rietveld Refinement and Pattern Matching Analysis. Abstracts XV of Congress of the IUCr Pp. 127.Toulouse, France. [ Links ]
32. Runping, KB; R Dohrmann; R Jahn & M Kleber. 2003. XRD quantification of soil clay mineral. X conference of the European Clay Groups Assoc. Pp. 242. Modena. Italy. [ Links ]
33. Schwertmann, U. & RM Taylor. 1977. Iron oxides Pp. 147 In: (eds. J Dixon & S Weed) Minerals in Soil Environments. S.S.S.A. Misc. Publ. Madison, WI. [ Links ]
34. Taubaso, C; RM Torres Sánchez & M Dos Santos Afonso. 2004. Modelling soil surface charge density using mineral composition. Geoderma 121: 123-133. [ Links ]
35. Taylor, JC & Z Rui. 1992. Simultaneous use of observed and calculated standard profiles in quantiative XRD analysis of minerals by the multiphase Rietveld method: The determination of pseudorutile in mineral sands products. Powder Diffraction 7(3): 152-161. [ Links ]
36. Torres Sánchez, RM & S Falasca. 1997. Specific surface area and surface charges of some Argentinean soils. Z. Pflan. Bodenk 160: 223-226. [ Links ]
37. Torres Sánchez, RM; M Okumura & R Mercader. 2001. Charge properties of red Argentina soils as indicative of iron oxide/ clay associations. Aust. J. Soil Res. 39: 1-12. [ Links ]
38. Weidler, P; J Luster; J Scheider; H Sticher & U Gehring. 1998. The Rietveld method applied to quantitative mineralogical and chemical analysis of ferralitic soil. Europ. J. of Soil Sci. 49: 95-105 [ Links ]
39. Wilding, LP; NE Smeck & LR Drees. 1977. Silica in Soils: Quartz, Cristobalite, Trydimite, and Opal. Pp. 500 In: (eds. J Dixon & S Weed) Minerals in Soil Environments S.S.S.A. Misc. Publ. Madison, WI. [ Links ]
40. Whittig, L & W Allardice. 1986. Chap. X-ray diffraction techniques Pp 336In: (ed. A Klute) Methods of soil analysis. S.S.S.A. Misc. Publ. Madison WI. [ Links ]
41. Young, RA. 1993. In: (ed. RA Young.) The Rietveld Method. International Union Crystallography, Oxford University Press. Oxford. [ Links ]














