Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO  uBio
uBio
Compartir
Boletín de la Sociedad Argentina de Botánica
versión On-line ISSN 1851-2372
Bol. Soc. Argent. Bot. v.42 n.1-2 Córdoba ene./jul. 2007
Screening of Colletotrichum (Ascomycota) isolates, causal agents of Soybean Anthracnose, for Laccase production*
L. Levin1 , A .M. Ramos1 , M . Parisi1 y M. Gally2
*Trabajo publicado en homenaje a la Dra Irma J. Gamundi en conmemoración de su 80º aniversario.
1Laboratorio de Micología Experimental, Dpto. de Biodiversidad y Biología Experimental, Facultad de Ciencias Exactas y Naturales
2Cátedra de Fitopatología, Facultad de Agronomía, Universidad de Buenos Aires, Ciudad Autónoma de Buenos Aires, Argentina. E-mail: lale@bg.fcen.uba.ar
Summary: Colletotrichum truncatum is the most common pathogen fungus associated with soybean anthracnose. Although the lignin-degrading enzyme laccase has been implicated in pathogenicity of a wide range of plant pathogenic fungi, its biological role in the Colletotrichum -soybean disease system is unknown. The extent of the infection in our country led us to examine laccase production in Argentinean Colletotrichum strains isolated from diseased soybean plants from different geographic locations. Ten strains (eight of them identified as C. truncatum) , were screened for in vitro laccase production. Only six of the isolates, all of them C. truncatum , produced laccase activity when cultured on a defined medium based on pectin and asparagine as carbon and nitrogen sources, respectively. Strain BAFC 3102 (isolated from Chaco province), yielded the highest laccase titers (44 U/L) in this medium. Denaturing polyacrylamide gel electrophoresis of extracellular culture fluids revealed one band with laccase activity (mol wt 67 kDa). CuSO 4 addition to media with either glucose or pectin as carbon sources increased up to 7-fold laccase production (280 U/L in the glucose medium), but the pattern of isoenzyme was not affected by culture age or medium composition. This is the first report on laccase production by C. truncatum.
Key words: Anthracnose; Colletotrichum; Laccase; SDS-PAGE; Soybean.
Resumen: Relevamiento de la producción de lacasa en aislamientos de Colletotrichum (Ascomycota), agente causal de antracnosis de la Soja. Colletotrichum truncatum es el hongo patógeno más comúnmente asociado con la antracnosis de soja. Aunque la enzima ligninolítica lacasa se relaciona con la patogenicidad de un amplio rango de hongos fitopatógenos, su rol biológico en la interacción Colletotrichum -soja aún se desconoce. La extensión de la infección en la Argentina , nos ha llevado a examinar la producción de lacasa en cepas aisladas de plantas enfermas de soja de diferentes regiones de nuestro país. Se evaluó la producción in vitro de lacasa en diez cepas (ocho de ellas identificadas como C. truncatum ). Sólo seis, todas correspondientes a C. truncatum , produjeron lacasa cuando fueron cultivadas en un medio con pectina y asparagina. La máxima actividad en este medio (44 U/L) se detectó en la cepa BAFC 3102 (aislada en la Provincia del Chaco). La electroforesis en SDS PAGE de los sobrenadantes de cultivo reveló una única banda con actividad lacasa (peso molecular 67 kDa). La adición de CuSO 4 al medio incrementó hasta siete veces su producción (280 U/L en medio con glucosa), sin embargo la edad del cultivo o la composición del medio no produjeron variaciones en el patrón isoenzimático. Este es el primer reporte sobre producción de lacasa por C. truncatum.
Palabras clave: Antracnosis; Colletotrichum; Lacasa; SDS-PAGE; Soja.
INTRODUCTION
Species of the anamorphic genus Colletotrichum are implicated in plant diseases (generally referred to as anthracnoses) throughout the world. Their ability to cause latent or quiescent infections places them amongst the most important of post-harvest pathogens, and numerous growing crops are affected (Bailey & Jeger, 1992). Soybean anthracnose reduces stands, seed quality and yields by 16-26% or more in US, and 100% in certain areas of Brazil and India . The most common pathogen associated with anthracnose is Colletotrichum truncatum (teleomorph Glomerella truncata ) (Armstrong-Cho & Banniza, 2006)). Other Colletotrichum spp. that can be involved include C. coccodes , C. destructivum (teleomorph Glomerella glycines ), C. gloeosporioides (teleomorph Glomerella cingulata ) and C. graminicola (teleomorph Glomerella graminicola ) (Hartman et al., 1999).
Laccase ( p -diphenol: oxygen oxidoreductase; EC 1.10.3.2), an extracellular enzyme produced by some fungi, is a copper-containing enzyme that catalyzes the oxidation of phenolic substrates by coupling it to the reduction of oxygen to water. The cell wall complex aromatic polymer lignin can provide a barrier to fungal entry and to the diffusion of fungal toxins and enzymes into plant cells (Elfstrand et al., 2002). But, in contrast to the cell wall-degrading enzymes and cutinase, which could be involved in breaching the physical barriers to entry into the plant, the lignindegrading enzyme, laccase, is thought additionally, to play a role in protecting phytopathogens from plant defense compounds (Bar-Nun & Mayer, 1989; Pezet et al., 1991). Proposed roles of plant pathogen laccases include detoxification of phytoalexins and other plant phenolics compounds (Rigling & Van Alfen, 1993; Anderson & Nicholson, 1996; Binz & Canevascini, 1996; Breuil et al., 1999) and protection against host oxidative responses (Edens et al., 1999). Even though laccases have been implicated in pathogenicity of a range of plant pathogenic fungi that includes the soft rot fungus Botrytis cinerea , the chestnut blight fungus Cryphonectria parasitica , the Dutch elm disease fungus Ophiostoma novo-ulmi and the take-all pathogen Gaeumannomyces graminis (Bar-Nun & Mayer, 1990; Sbaghi et al., 1996; Viterbo et al., 1994; Rigling & Van Alfen, 1993; Binz & Canevascini, 1996; Edens et al., 1999), the roles of laccases in plant-pathogen interactions remain poorly understood. The production of cucurbitacins by cucumber, suppresses disease symptoms through repression of the levels of both intracellular and extracellular laccases of B. cinerea (Bar-Nun & Mayer, 1989; 1990); in addition, cucurbitacins are observed to alter the electrophoretic mobility of the extracellular laccase (Viterbo et al . , 1994). Significant reduction in laccase activity is one of the factors associated with hypovirulence in viral infected strains of the chestnut blight fungus C. parasitica (Rigling & Van Alfen, 1993). Likewise, hypovirulence in Diaporthe ambigua , isolated from apple rootstocks, and Didymella bryoniae , the causal agent of gummy stem blight disease in cucurbitaceae, again correlates with reduced levels of a laccase-type phenol oxidase activity (Smit et al., 1996; Urs et al . , 2004). Moreover inhibitors of fungal laccase reduced C. gloeosporioides symptom development when applied to ripening susceptible fruits (Guetsky et al., 2005). Levels of resistance of crops to fungi could be increased by understanding and then genetically manipulating metabolic events that lead to production of antimicrobial compounds that are toxic to pathogens or that can strengthen the barriers of plant cells to pathogen entry (Dixon et al., 2002). The biological role of laccases in the Colletotrichum soybean disease system is unknown. The extent of the infection in our country, one of the main world soybean producers, and the potential value of laccases to this plant pathogen, led us to examine laccase production in Argentinean Colletotrichum strains isolated from diseased soybean plants from different geographic locations.
MATERIALS and METHODS
Microorganisms - Strains BAFC 3093-3102 (BAFC: Mycological Culture Collection of the Department of Biological Sciences, Faculty of Exact and Natural Sciences, University of Buenos Aires ) of the anamorphic genus Colletotrichum Corda were used in these experiments. The isolates were collected by Marcela Gally during April/May 2004, from lesions of stems and pods of symptomatic soybean plants from seven Argentinean localities in the provinces of Buenos Aires (3099, 3100, 3101), Santa Fe (3093, 3096, 3097, 3098), Chaco (3102), La Rioja (3095) and Misiones (3094). Strains 3093, 3094, 3096, 3098, 3099, 3100, 3101 and 3102 were identified as C. truncatum (Schweinitz) Andrus et Moore. Pieces of symptomatic tissue bearing immature acervuli were surface sterilized in 1.5 % sodium hypochlorite for 2 minutes, rinsed twice in sterile water and incubated in humid chambers at 25- 27 ºC under 12 hours near ultraviolet light (nuv) and 12 hours of darkness, until liberation of masses of conidia, which were cultured in potatodextrose agar (PDA) plates at 25- 27 ºC in darkness. Hyphal tips were aseptically transferred to PDA and incubated at 25- 27 ºC under nuv light/darkness (12/ 12 hours) to obtain pure cultures. Stock cultures were maintained on potato-dextrose agar slants at 4 ºC .
Basal culture medium - Pectin from apple, 10 g ; asparagine monohydrate, 4 g ; MgSO4 ·7H2O, 0.5 g ; H2KPO4, 0.5 g ; HK2PO4 , 0.6 g ; CuSO4 ·5H2O, 0.4 mg; MnCl2 ·4H2O, 0.09 mg; H3BO3 , 0.07 mg; Na2MoO4·2H2O, 0.02 mg; FeCl3 , 1 mg; ZnCl2 , 3.5 mg; thiamine hydrochloride, 0.1 mg; distilled water up to 1 L. Final pH: 3.5. Alternatively glucose 10g/L, was used as carbon source and CuSO 4 ·5H 2 O (667 µ M) was added to the medium. Final pH was then adjusted to 3.5 with either NaOH or HCl 1N.
Culture conditions - 100 mL Erlenmeyer flasks with 20 mL of medium were inoculated with one agar plug (of 0.25 cm 2 ) cut out from a colony grown on Bacto-agar 2%. Incubation was carried out at 23 ± 1 ºC under stationary conditions. Cultures were harvested at different incubation periods, filtered through a filter paper using a Büchner funnel and dried overnight at 70 ºC . Dry weight of mycelia was then determined. The culture supernatants were used as enzyme sources.
Enzyme assays - Laccase activity was measured with 2,2'-azino bis(3-ethylbenzthiazoline-6-sulphonic acid) (ABTS) in 0.1 M sodium acetate buffer (pH 3.5) at 50 ºC . Oxidation of ABTS was determined by the increase in A 420 ( 420 = 36/mM cm) (Levin et al., 2002).
International enzymatic units (U) were used ( µ mol/ min). Enzyme activity was expressed as U/L of culture filtrate. The results are the average of three triplicate experiments with a standard error of less than 5%. Polyacrylamide gel electrophoresis (PAGE) and activity staining of gels - Separation was carried out on vertical polyacrylamide slab gels. Electrophoresis was performed on 9 % polyacrylamide gel under denaturing conditions (SDS PAGE) (Laemmli, 1970). The buffer solution for the separating gel was Tris- HCl 50 mM (pH 8.8). For electrophoresis if necessary, culture filtrates were concentrated in a dialysis tube against sacarose. Up to 50 µ L of supernatants from different culture days and conditions (to render a laccase activity of approx 0.005 U) were loaded onto the gel and electrophoresed with Tris-glycine buffer (pH 8.3) at 120 V. After electrophoresis the gel was fixed for 10 min in 10% (v/v) acetic acid and 40% (v/v) methanol, and then soaked in 50 mM acetate buffer (pH 3.5) containing 3 mg/mL of ABTS. Protein bands exhibiting laccase activity stained green with ABTS within 5 min. For the determination of the molecular weights of the laccase isoenzymes, samples were run with Full range Rainbow Molecular Weight Markers (10-250 kDa), Amersham Biosciences, U.K. All chemicals were purchased from Sigma Chemical Co.
RESULTS and DISCUSSIONS
The kinetics of in vitro production of extracellular laccase activity in ten strains of Colletotrichum solated from diseased soybean plants with anthracnose symptoms from diverse Argentinean geographical locations was studied in a synthetic medium based on pectin as carbon source and asparagine as nitrogen source. Figure 1 depicts the relationship between growth and laccase activity in all the strains evaluated. Only six of the isolates evaluated, produced laccase activity in these conditions, all of them identified as C. truncatum. Laccase activity appeared in all the isolates before mycelial biomass peaked (i.e. it was present in the primary phase growth) (data not shown), but showed its maximum at days 13-15, generally in coincidence with the highest biomass recorded (Fig. 1). On the contrary, the ligninolytic enzyme system of white rot of growth, usually is triggered in response to N or C depletion, attaining its maximum in the idiophase when the mycelial dry weight is decreasing (Kaal et al., 1995). Biomass production in all the strains evaluated varied among 2.5-3.5 mg/ml medium. Differences in laccase production seemed to be not related to mycelial yield, considering that very low levels of laccase activity were detected in strain 3093, and in contrast, strain 3102 rendered the uppermost laccase titres (44 U/L), but the maximum of biomass measured (3.5 mg/ml) was similar in both isolates. Likewise, when comparing laccase activity of strains of Heterobasidion annosum belonging to the more aggressive P and less aggressive S intersterility groups, Johansson et al. (1999) found out that enzyme activity was significantly higher for the P strains tested, compared with a similar number of S strains, although biomass production was significantly lower in P cultures than in S cultures. The extracellular laccase production by C. truncatum BAFC 3102 in this medium was comparable with that of Fusarium solani f. sp. glycines (agent of sudden death syndrome), and two known lignin-degrading fungi: Polyporus tulipifera and Schizophyllum commune on modified Murashige-Skoog medium (Lozovaya et al., 2006).
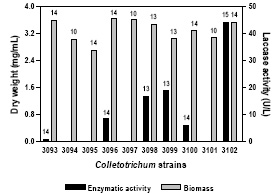
Fig. 1. Maximum biomass level attained by ten Colletotrichum strains grown in a synthetic medium with pectin 10 g/L and asparagine 4 g/L, and laccase production in this medium. The numbers over the bars indicate the day when the highest value was achieved.
Nevertheless, the choice of culture conditions had a great impact on the final amount of laccase produced. Figure 2 shows growth and laccase production by C. truncatum strain 3102 cultured in different media. In the pathogen B. cinerea , pectin not only induces pectic enzymes but also serves as a second inducer of laccase synthesis and secretion (Marbach et al., 1985), but in the case of C. truncatum, when using glucose as carbon source laccase production (41 U/ L) was similar than in pectin medium (44 U/L). CuSO 4 (667 M) addition increased laccase production in both media, attaining a maximum of 280 U/L in the medium with glucose and copper. Higher copper concentrations were not assayed since they may be toxic for fungi, affecting their growth and enzymatic activities (Baldrian, 2003). Copper has been reported to be a strong laccase inducer in several white rot fungi, among them Trametes pubescens (Galhaup et al., 2002), Trametes versicolor (Collins & Dobson, 1997), Grammothele subargentea (Saparrat, 2004) and Grifola frondosa (Xing et al., 2006). It is known that copper induces both laccase transcription and activity (Collins & Dobson, 1997), and the increase in activity is proportional to the amount of copper added. Production of extra laccase isoenzymes, was observed after copper addition in Phanerochaete chrysosporium and Marasmius quercophilus (Klonowska et al., 2001), and in G. frondosa (Xing et al., 2006), but not in Trametes trogii (Levin et al., 2002).
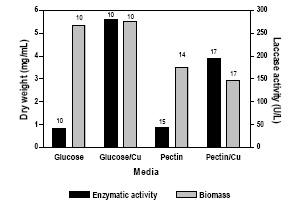
Fig. 2. Maximum biomass level and laccase production by C. truncatum (BAFC 3102) in different media: either with glucose or pectin (10 g/L) as carbon sources, alternatively supplemented with CuSO4 ·5H2O (667 µ M).
Strains of Colletotrichum were compared in respect of their laccase isozyme pattern. Denaturing polyacrylamide gel electrophoresis confirmed the spectrophotometric assays. Either strains 3094, 3095, 3097 and 3101 did not produce laccase or their levels were too low to be detected by gel electrophoresis or by the spectrophotometric assay. However, their laccase production under different culture conditions fungi produce multiple laccase isozymes, encoded by differentially expressed genes that may fulfill different functions (Burke & Cairney, 2002). Many plant pathogenic fungi produce different laccase isoenzymes encoded by multiple genes (i.e. four laccase genes have been detected in Rhizoctonia solani (Wahleithner et al . , 1996) and three in G. graminis var. tritici (Litvintseva & Henson, 2002). Expression of these genes can be either constitutive or sensitive to the presence of inducers.
Consequently, in different fungi, the production of minor laccase forms can be enhanced under appropriate culture conditions; but the zymograms of extracellular culture fluids of Colletotrichum revealed only one band with laccase activity, with similar electrophoretic mobility, in the strains: 3093, 3096, 3098, 3099, 3100 and 3102 (Figure 3A), and even under different growth conditions (Figure 3B). The molecular weight of laccase, determined by SDSPAGE, was 67 kDa (Figure 3B). This finding is generally in keeping with those for laccases obtained from strains within a wide variety of fungal genera (Thurston, 1994). As in the white-rot fungus T. trogii (Levin et al . , 2002) the pattern of isoenzyme production by Colletotrichum spp. was not affected by culture age (data not shown). On the contrary, expression of the laccase isozymes was dependent on culture phase in Fusarium proliferatum an opportunistic pathogen isolated from wheat (Known & Anderson , 2001).

Fig. 3. (A) Laccase activity staining after SDS PAGE of Colletotrichum concentrated culture filtrates. Cultures grown in a synthetic medium with pectin 10 g/L and asparagine 4 g/L. Lanes 1-10 respectively strains: BAFC 3094 (1), 3093 (2), 3096 (3), 3097 (4), 3098 (5), 3099 (6), 3100 (7), 3101 (8), 3102 (9), 3095 (10). (B) Laccase activity staining after SDS PAGE of C. truncatum (BAFC 3102) culture filtrates. Cultures grown in a synthetic medium with either glucose (10 g/L) (Lanes 1 and 2) or pectin (10 g/L) (Lanes 3 and 4) as carbon sources, with (Lanes 2 and 4) or without (Lanes 1 and 3) CuSO4·5H2O (667 µ M) addition. Samples were run with prestained molecular weight protein markers
In conclusion, although laccase production has been investigated in other Colletotrichum species (among them C. graminicola (Anderson & Nicholson, 1996), C. lagenarium (Tsuji et al . , 2001) and C. gloeosporioides (Guetsky et al., 2005), as far as we know this is the first report on laccase production by C. truncatum . Taking into account that we found remarkable differences in laccase production in the strains of Colletotrichum evaluated, the aim of our prospect studies will be to test the hypothesis that differences in pathogenicity and aggressiveness in strains of Colletotrichum causing anthracnose in soybean, stem from differences in laccase activity.
ACKNOWLEGDMENTS
The authors are grateful to Ing. Marcelo Carmona for supplying the diseased soybean plants, and to Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET, Argentina ) and University of Buenos Aires, for financial support.
BIBLIOGRAPHY
ARMSTRONG-CHO, C. L. & S. BANNIZA. 2006. Glomerella truncata sp. nov., the teleomorph of Colletotrichum truncatum . Mycol. Res. 110: 951-956. [ Links ]
ANDERSON, D. W. & R. L. NICHOLSON. 1996. Characterization of laccase in the conidial mucilage of Colletotrichum graminicola . Mycologia 88: 996-1002. [ Links ]
BAILEY, J. A. & M. J. JEGER. 1992. Colletotrichum : biology, pathology and control. CAB International, Wallingford , Oxon. [ Links ]
BALDRIAN, P. 2003. Interaction of heavy metals with white rot fungi. Enzyme Microb. Technol. 32: 78-91. [ Links ]
BAR-NUN, N. & A. M. MAYER. 1989. Cucurbitacinsrepressors of induction of laccase formation. Phytochemistry 28: 1369-1371. [ Links ]
BAR-NUN, N. & A. M. MAYER. 1990. Cucurbitacins protect cucumber tissue against infection by Botrytis cinerea. Phytochemistry 29: 787-791. [ Links ]
BINZ, T. & G. CANEVASCINI. 1996. Differential production of extracellular laccase in the dutch elm disease pathogens Ophiostoma ulmi and O. novo-ulmi . Mycol. Res. 100: 1060-1064. [ Links ]
BREUIL, A. C. ,P. JEANDET & M. ADRIAN . 1999. Characterization of a pterostilbene dehydropolymer produced by laccase of Botrytis cinerea . Phytopathology 89: 298-302. [ Links ]
BURKE, R. M. & J: W: G: CAIRNEY. 2002. Laccases and other polyphenol oxidases in ecto- and ericoid mycorrhizal fungi. Mycorrhiza 12: 105-116. [ Links ]
COLLINS, P. J. & A: D: W: DOBSON.1997. Regulation of laccase gene transcription in Trametes versicolor . Appl. Environ. Microbiol . 63: 3444-3450. [ Links ]
DIXON , R. A., L: ACHNINE & P. KOTA . 2002. The phenylpropanoid pathway and plant defense-a genomic perspective. Mol. Plant. Biol . 3: 371-390. [ Links ]
EDENS , W.A., T: Q: GOINS & D: DOOLEY. 1999. Purification and characterization of a secreted laccase of Gaeumannomyces graminis var. tritici . Appl. Environ. Microbiol. 65: 3071-3074. [ Links ]
ELFSTRAND, M., F. SITBON & C. LAPIERRE. 2002. Altered lignin structure and resistance to pathogens in spi 2- expressing tobacco plants. Planta 214: 708-716. [ Links ]
GALHAUP, C., H. WAGNER & B. HINTERSTOISSER. 2002. Increased production of laccase by the wood-degrading basidiomycete Trametes pubescens . Enzyme Microb. Technol. 30: 529-536. [ Links ]
GUETSKY, R., I. KOBILER & X. WANG. 2005. Metabolism of the flavonoid epicatechin by laccase of Colletotrichum gloeosporioides and its effect on pathogenicity on avocado fruits. Phytopathology 95: 1341-1348. [ Links ]
HARTMAN, G. L., J. B. SINCLAIR & J. C. RUPE. 1999. Compendium of soybean diseases, 4th ed. APS Press, Saint Paul. [ Links ]
JOHANSSON, M, M. DENEKAMP & F: O. ASIEGBU. 1999. Production and isozyme pattern of extracellular laccase in the S and P intersterility groups of the root pathogen Heterobasidion annosum. Mycol. Res. 103: 365-371. [ Links ]
KAAL, E. E. J., J. A. FIELD & T. W. JOYCE. 1995. Increasing ligninolytic enzyme activity in several white-rot basidiomycetes by nitrogen-sufficient media. Bioresour. Technol . 53: 133-139. [ Links ]
KLONOWSKA, A., J. LE PETIT & T. TRON. 2001. Enhancement of minor laccases production in the basidiomycete Marasmius quercophilus C30. FEMS Microbiol. Lett. 200: 25-30. [ Links ]
KWON, S. I. & A: J: ANDERSON . 2001. Laccase isozymes: production by an opportunistic pathogen, a Fusarium proliferatum isolate from wheat. Physiol. Mol. Plant Pathol. 59: 235-242. [ Links ]
LAEMMLI, U. K. 1970. Cleavage of structural proteins during the assembly of the head of the bacteriophage T4. Nature 227: 680-685. [ Links ]
LEVIN, L., F. FORCHIASSIN & A. M. RAMOS. 2002. Copper induction of lignin modifying enzymes in the white rot fungus Trametes trogii . Mycologia 94: 377-383 [ Links ]
LITVINTSEVA, A. P. & J. M. HENSON. 2002. Cloning, characterization, and transcription of three laccase genes from Gaeumannomyces graminis var. tritici , the take-all fungus. Appl.. Environ. Microbiol . 68: 1305-1311 [ Links ]
LOZOVAYA, V. V., A. V. LYGIN & O. V. ZERNOV. 2006. Lignin degradation by Fusarium solani f. sp. glycines. Plant Dis. 90: 77-82 [ Links ]
MARBACH, I. , E. HAREL & A. M. MAYER. 1985. Pectin, a second inducer for laccase production by Botrytis cinerea. Phytochemistry 24: 2559-2561. [ Links ]
NELSON, N. 1944. A photometric adaptation of the Somogyi method for the determination of glucose. J. Biochem. 153: 375-380. [ Links ]
PEZET, R., V. PONT & K. HOANG-VAN. 1991. Evidence for oxidative detoxification of pterostilbene and resveratrol by a laccase-like stilbene oxidase produced by Botrytis cinerea . Physiol. Mol. Plant Pathol. 39: 441- 450. [ Links ]
RIGLING, D. & N. K. VAN ALFEN. 1993. Extra- and intracellular laccases of the chestnut blight fungus, Cryphonectria parasitica. Appl. Environ. Microbiol . 59:.3634-3639. [ Links ]
SAPARRAT, M. C. 2004. Optimizing production of extracellular laccase from Grammothele subargentea CLPS No. 436 strain. World J. Microbiol. Biotechnol. 20: 583-586. [ Links ]
SBAGHI, M., P. JEANDET & R. BESSIS. 1996. Degradation of stilbene-type phytoalexins in relation to the pathogenicity of Botrytis cinerea to grapevines. Plant Pathol. 45: 139-144. [ Links ]
SMIT, W.A., B. D. WINGFIELD & M. J. WINGFIELD. 1996. Reduction of laccase activity and other hypovirulenceassociated traits in dsRNA-containing strains of Diaporthe ambigua . Phytopathology 86: 1311-1316. [ Links ]
THURSTON, C. F. 1994. The structure and function of fungal laccases. Microbiology 140: 19-26. [ Links ]
TSUJI, G., J. FUJIKAWA & H. ISHIDA.2001. Laccase gene LAC1 of. Colletotrichum lagenarium is not essential for melanin biosynthesis and pathogenicity. J. Gen. Plant Pathol . 67: 182-190. [ Links ]
URS, R. R., D. E. MAYHEW & P. D. ROBERTS. 2004.Hypovirulence of Didymella bryoniae associated with dsRNA. J. Phytopathol. 152: 55-59. [ Links ]
VITERBO, A, R. C. STAPLES & B. YAGEN. 1994. Selective mode of action of cucurbitacin in the inhibition of laccase formation in Botrytis cinerea. Phytochemistry 35: 1137- 1142. [ Links ]
WAHLEITHNER, J. A., F. XU & K. M. BROWN. 1996. The identification and characterization of four laccases from the plant pathogenic fungus Rhizoctonia solani . Curr. Genet. 29: 395-403. [ Links ]
XING, Z.T., J. H. CHENG & Q. TAN. 2006. Effect of nutritional parameters on laccase production by the culinary and medicinal mushroom, Grifola frondosa. World J. Microbiol. Biotechnol. 22: 1215-1221. [ Links ]
Recibido el 04 de Diciembre de 2006,
aceptado el 24 de Abril de 2007.














