Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO  uBio
uBio
Compartir
Boletín de la Sociedad Argentina de Botánica
versión On-line ISSN 1851-2372
Bol. Soc. Argent. Bot. vol.52 no.1 Córdoba mar. 2017
ANATOMÍA Y MORFOLOGÍA
Morpho anatomical characterization and essential oils of Tagetes terniflora and Tagetes minuta (Asteraceae) growing in Tucumán (Argentina)
Emilio Lizarraga1,2, Maria I. Mercado3*, Carolina Galvez4, Ana I. Ruiz3, Graciela I. Ponessa3 and Cesar A. N. Catalan4
1 Instituto de Fisiología Animal, Área Zoología, Fundación Miguel Lillo. Miguel Lillo 251 (T4000JFE), San Miguel de Tucumán.
2 Facultad de Ciencias Naturales e Instituto Miguel Lillo, Universidad Nacional de Tucumán. Miguel Lillo 205 (T4000JFE), San Miguel de Tucumán.
3 Instituto de Morfología Vegetal, Área Botánica, Fundación Miguel Lillo. Miguel Lillo 251 (T4000JFE), San Miguel de Tucumán. Argentina.
4 Instituto de Química de Noroeste Argentino. CONICET - Facultad de Bioquímica, Química y Farmacia, Universidad Nacional de Tucumán, Batalla de Ayacucho 471 (T4000JFE), San Miguel de Tucumán.
* mimercado@lillo.org.ar
Summary
The essential oil composition and morpho-anatomy of Tagetes minuta, collected at "El Mollar" and two different populations of Tagetes terniflora collected in Tafí del Valle (Tucumán, Argentina) were studied. The essential oil of aerial parts (flowers and leaves) from both species was analyzed by gas chromatography-mass spectrometry. Qualitative and quantitative differences were observed in the composition of the essential oil from both species. Oxygenated monoterpenes were the dominant compounds in the essential oil from both collections of T. terniflora where the main component was cis-tagetone (33.6% and 58.4% respectively) accompanied by significant amounts of the monotepene hydrocarbon cis-β-ocimene (17.1% and 17.4% respectively). cis-Tagetone (53.2%) was also a dominant component in T. minuta along with dihydrotagetone (10.4%) and cis-β-ocimene (19.9%). cis- and trans-ocimenone were distinctive components of T. terniflora which were not detected in T. minuta. Anatomically stands out the presence of secretory cavities and glandular trichomes in foliar blades and secretory ducts in stems and petiole of both species.
Key words: Tagetes terniflora; Tagetes minuta; Essential oil; Secretory tissues; Morpho-anatomy.
Resumen
Caracterización morfo-anatomía y aceites esenciales de Tagetes terniflora y Tagetes minuta (Asteraceae) de Tucumán (Argentina)
Se estudió la composición del aceite esencial y morfo-anatomía de Tagetes minuta coleccionada en "El Mollar" y de dos poblaciones diferentes de Tagetes terniflora de Tafí del Valle (Tucumán, Argentina). El aceite esencial de las partes aéreas (flores y hojas) de ambas especies se analizó por cromatografía gaseosa-espectrometría de masa. Se observaron diferencias cuali- y cuantitativas en la composición del aceite esencial de ambas especies. En las dos colecciones de T. terniflora los compuestos dominantes del aceite esencial fueron monoterpenos oxigenados donde el componente principal fue la cis-tagetona (33.6% y 58.4% respectivamente) acompañado por cantidades significativas del hidrocarburo monoterpénico cis-β-ocimeno (17.1% y 17.4% respectivamente). La cis-tagetona (53.2%) fue también el componente dominante en T. minuta junto con dihidrotagetona (10.4%) y cis-β-ocimeno (19.9%). La cis- y trans-ocimenona fueron componentes característicos de T. terniflora que no fueron detectados en T. minuta. Anatómicamente se destaca la presencia de cavidades secretoras y tricomas glandulares secretores en los bordes foliares y conductos secretores en tallos y pecíolo de ambas especies.
Palabras clave: Tagetes terniflora; Tagetes minuta; Aceite esencial; Tejidos secretores; Morfo-anatomía.
Introduction
Tagetes L. (Asteraceae, tribe Heliantheae) is a genus of about 43 species originating in Central and South America (Kaplan, 1958). Their natural range extends from the south of the United States and México to Argentinean Patagonia. The genus comprises ornamental, aromatic herbs and shrubs popularly known as marigolds. In Argentina is represented by 23 species, among which, Tagetes terniflora Khunt "wild tagetes" is native from Ecuador to the north of Argentina, and introduced in South Africa, Australia and the South of Europe; and T. minuta L. "chinchilla", "mexican marigold" is distributed from the South of the United States to the Argentinean Patagonia, and introduced in Europe, Asia, Africa, Madagascar, India, Australia, Japan, Croatia and Hawaii (Soulé, 1993; Flora Argentina, 2016).
Several species of Tagetes are extensively used as condiment, ornamentals, medicinals, and ritual plants (Soulé, 1996; Barboza et al., 2009), being T. minuta, T. erecta and T. patula the most studied (Vasudevan et al., 1997).
T. terniflora essential oil showed bactericidal (Tereschuk et al., 2003) and insecticidal activities (Stefanazzi et al., 2006), while T. minuta is an economically important worldwide resource due to the agrochemical, phytotoxic, fungicidal, insecticidal and pharmacological properties of its essential oil (Maradufu et al., 1978; Perich et al., 1995; Macedo et al., 1997; Bii et al., 2000; López et al., 2009; Karimian et al., 2014; Shirazi et al., 2014). The industry produces "tagetes oil" which is marketed for its various medicinal properties, health benefits and uses (Martijema et al., 1998; Sadia et al., 2013). T. minuta and T. teniflora essential oils are rich in monoterpenes, sequiterpenes, flavonoids, thiophenes and aromatics compounds. The major components reported for T. minuta were cis-tagetone, trans-tagetone, cis-β-ocimene, dihydrotagetone, cis-ocimenone, trans-ocimenone and the sesquiterpene alcohol spathulenol (López et al., 2009; Karimian et al., 2014.) while cis and trans-tagetone were the main components of T. terniflora (Stefanazzi et al., 2006). Studies related to differences in oil composition and activity among species, different populations of a same species, throughout the life cycle, and even between different organs in the same species have been reported (Zygadlo et al., 1990, 1993; Bii et al., 2000; López et al., 2009).
Tagetes are characterized by the presence of oil glands (= secretory cavities), ducts and secretory cells that contain volatiles constituents (Metcalfe & Chalk, 1950; Lawrence, 1985; Simon et al., 2002). These secretory structures have been analyzed in T. minuta (Del Fueyo, 1986; Simon et al., 2002; López et al., 2009), but they are not described for T. terniflora.
In the province of Tucumán T. minuta and T. terniflora overlap in their distribution range being often misidentified by local villagers. Tagetes minuta is easily distinguishable from T. terniflora by capitula features (Fig. 1 A-B) (Cabrera, 1978; Flora Argentina, 2016), however, the identification of both species becomes difficult in vegetative state. Thus, the aim of this paper is to characterize the essential oil composition and vegetative organs morpho-anatomy of both Tagetes species, collected in different places of Tucumán province.

Fig. 1. General Aspect. A: Tagetes minuta. B: Tagetes terniflora.
Materials and Methods
Sample collection
Aerial parts of T. minuta were collected at "El Mollar" Prov. Tucumán, Dpto. Tafí del Valle, 26º 55' S 65º 41' O (1906 masl); and T. terniflora from two population, from Tafi del Valle, Dpto. Tafí del Valle, Prov. Tucumán, Argentina, Route 307, km 43 and km 77 (1988 masl and ca. 2700 masl). Each collection was named as Tm and Tt (CI and CII respectively). Voucher specimens of each collection were deposited at the Herbarium of Fundación Miguel Lillo (LIL).
Extraction of essential oil
Fresh aerial parts of vegetal material (100g) of each collection were hydrodistilled in a Clevenger's type apparatus for 2 hours in accordance with European Pharmacopeia's procedure (Clevenger European Pharmacopeia, 1983). The essential oils obtained were dried over anhydrous sodium sulfate to remove traces of moisture, flushed with argon and stored in a refrigerator in the dark at 4°C in a well-sealed bottle until analysis. All samples were analyzed within 72 hs of obtaining them. The essential oils of both T. minuta and T. terniflora deteriorate rapidly (oxidation, polymerization) in contact with air and light.
Analysis
The chemical analysis was undertaken by gas chromatography-mass spectroscopy (GC-MS) techniques using a Hewlett Packard 6890 gas chromatograph coupled to a Hewlett-Packard 5973 quadrupole mass spectrometer equipped with a Perkin-Elmer Elite-5MS capillary column (5% phenyl methyl siloxane, length= 30 m, inner diameter= 0.25 mm, film thickness= 0.25 mm) and a selective mass detector was used for GC-MS detection; an electron ionization system with ionization energy of 70 eV was used. Helium was used as a carrier gas at a flow rate of 1.0 mL/min. Injector and ion source temperature were set at 250 and 300 ºC respectively. Injection volume was 1 mL with a split ratio 80:1 and the oven temperature was programmed as follows: 60 ºC (5 min), 60-180 ºC (3.0 ºC/min), 180 ºC (10min), 180-250 ºC (7 °C/ min), 250 ºC (3 min).
Identification of components
The components percentage reported in Table 1 was taken from capillary GC traces with FID (Flame Ionization Detector) using an integrator HP 3395 without FID response factor correction. The identification of the individual components was based on (a) computer matching with commercial mass spectra libraries (NBS75K, NIST 98, WILEY275) and published data (Adams, 2007); (b) comparison with spectra available in our files; (c) comparison of their GC arithmetic index (AI) on a HP-5 column. The arithmetic indices were calculated using a homologous series of n-alkanes C8-C18 (Adams, 2007).
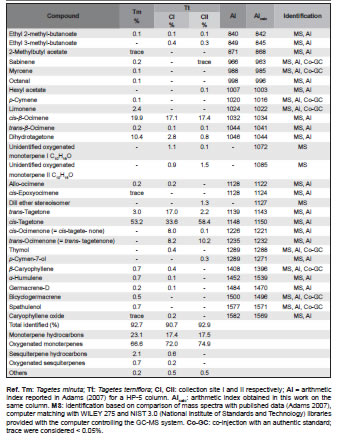
Table 1. Chemical composition of the essential oils from Tagetes minuta and Tagetes terniflora collected in Tucumán.
Microscopy
For histological preparations samples were fixed in FAA (formalin, ethanol, acetic acid, water, 100:500:50:350 mL). Sections of 3-4 mm, of the midregion of leaves (petiole and apical segment of fully expanded leaves), roots (first lateral root, next to the soil surface) and stems (third internode from the apex) were placed between two plates of dental wax and sectioned. Sections 15-20 μm were obtained with a rotation microtome MICROM HM 315, cleared with sodium hypochlorite (50% commercial bleach) and washed with distilled water. They were then stained with astra blue-safranin and mounted in 50% glycerol (Zarlavsky, 2014).
Whole foliar segments (third pair of leaves from the apex) and stems (third-fourth internode from the apex) were cleared according to Dizeo de Strittmatter (1973) and stained with cresyl violet 1% in distilled water. Leaf architecture was described using the terminology proposed by Hickey (1979) and Ellis et al. (2009). Stomata types were described according to Dilcher (1974). For light microscopy a Zeiss Axiolab optic microscope equipped with a Zeiss Axiocam ERc 5s digital camera and AxioVision Rel.4.3 acquisition software was used.
For scanning electron microscopy (SEM) samples were fixed in glutaraldehyde phosphate 5% buffered with 0.1 M sodium cocadylate at pH 7, and postfixed in 1.5% osmium tetroxide buffered with 0.1 M sodium cocadylate at pH 7.2. Leaf slices were dehydrated in a graded acetone solutions series, and coated with gold (Fine Coat Ion Sputter JEOL JFC-1100). Scanning electron microscopy (SEM) of gold coated samples was performed using a ZEISS SUPRA-55 VP field emission scanning electron microscope at Centro Integral de Microscopía Electrónica (CIME), CONICET-UNT.
Statistical analyses
Epidermal characteristics, stomatal sizes and stomatal densities were compared within and among species by analyses of variance (ANOVA) and differences among means were tested by the Tukey (InfoStat, 2002).
Results
Essential oil
The hydrodistillation of fresh aerial parts of each collection yielded yellowish oils, 0.91% for Tm and 2.4% and 2.8% for Tt, CI and CII respectively. For Tm 20 compounds were identified accounting for 92.7% of total, while for Tt 17 compounds were identified in CI and 14 compounds in CII constituting 90.7% and 92.9% of the oil respectively. The components identified in each collection and their relative percentages are listed in Table 1.
Tm was characterized by high percentages of oxigenated monoterpenes, i.e., dihydrotagetone (10.4 %), cis- and trans-tagetone (53.2% and 3.0% resp.) accompanied by lower amounts of monoterpene hydrocarbons the most relevant being cis-β-ocimene (19.9%) and limonene (2.4 %). Only minor amounts of sesquiterpenoids (2.8%) were detected in the oil: β-caryophyllene (0.7%), α-humulene (0.7%), germacrene-D (0.2%), bicyclogermacrene (0.5%), spathulenol (0.7%) and caryophyllene oxide (trace); ocimenones (= tagetenones) were not detected (Table 1).
The most distinctive difference between the oils from Tm and Tt was the presence of ocimenones in the latter. Both collections of Tt showed a high percentage of oxigenated moneterpenes with tagetones (50.6% and 60.6% in CI and CII resp.) and ocimenones (16.2% and 10.3% in CI and CII resp.). cis-Tagetone was the dominant component in both collections (33.6% and 58.4% respectively). It is noteworthy the variation in the relationship of cis- and trans-tagetone between collections, ca. 2:1 for CI and ca. 26:1 for CII. A quantitative difference was also found for dihydrotagetone: 2.8% in CI and 0.8% in CII. cis- β-Ocimene was the main monoterpene hydrocarbon with almost the same percentage in both collections (17.1% and 17.4% resp.). Also significant differences were also observed in the content of sesquiterpenoids. Significant differences were also observed in the content of sesquiterpenoids. Thus, the essential oil of CI contains small amounts of β-caryophyllene, α-humulene, germacrene-D and caryophyllene oxide while none of these were detected in CII (see Table 1).
Foliar morphology architecture and anatomy
The two species of Tagetes studied in this work showed green yellowish, elliptic, pinnatisect noto-mesophyll leaves (4-20 cm long. x 3-8.5 cm lat. in T. minuta and 5-15 cm long. x 3-9 cm lat. in T. terniflora); constituted by 4-10 (2.5-8 cm long., 1-3 cm lat.) segments in T. minuta and 3-8 segments (1.2-0.50 cm long. x 0.2-1.2 cm lat.) in T. terniflora. Segments are opposite in the base to alternate in the apex, elliptic-lanceolate, with acute apex, lineal raquis and serrate-dentate margin, sometimes with a fully developed second order teeth (apical side straight, basal side convex to flexuous with angular sinus) (Fig. 2 A-B). Oil glands appeared macroscopically as translucent dots on the surface of the leaves. In Tagetes minuta conspicuous rounded-elliptic, pellucid glands (446.3 ± 32.7 mm long. x 153.3 ± 19.0 mm lat.), occur in the basal region of the teeth, and less frequently smaller rounded glands (124.7 ± 47.3 mm long. x 123.1 ± 24.5 mm lat.) occur along leaf blade in the proximity of the midvein, 10-15 per segment (Fig. 2 A) while in T. terniflora glands were slightly elliptic or rounded (210.7 ± 55.8 mm long. x 148.9 ± 22.9 mm lat.) distributed the largest ones in the basal region of the teeth and the smallest in the foliar lamina (Fig. 2 B). Short petiole with stipules surrounds the steam.
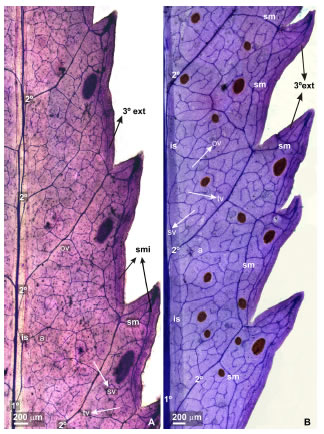
Fig. 2. Leaf architecture. A: Tagetes minuta. B: Tagetes terniflora. Abbreviations: 1º, primary vein; 2º, secondary vein; 3º ext, exterior tertiary; a, areole; is, intersecondary vein; sm, minor secondary; smi, intramarginal minor secondary; sv, simple veinlet; ov, one brunch veinlet; tv, two brunch veinlet.
The venation pattern was similar in both species. Terminal leaf segment presented primary vein pinnate, massive with straight course. Major secondary veins were decurrent, alternated, eucamptodromous becoming reticulodromous distally, sometimes, with irregular spacing gradually increasing proximally, forming straight to acute angles with the main vein. In T. minuta they form an intramarginal sencondary vein. Minor secondary veins were semicraspedodromous terminating at the apex of the teeth or becoming reticulodromous. In both species intersecondary veins were rare, parallel to major secondaries with a ramifying distal course, some times basiflex. Intercostal and epimedial terciary veins were irregular reticulated, forming areoles sometimes with one or two freely ending veinlets, often with one or two branchs. In T. terniflora tertiary exterior veins form looped marginal ultimate venation and teeth vascularization (Fig. 2 B).
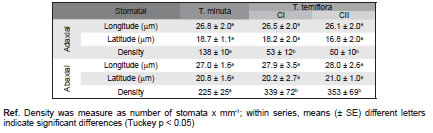
In frontal view Tagetes species showed amphistomatic leaves, striated cuticle (Fig. 3 E) and anomocytic stomata (Fig. 3 A-D). Stomata lengths were similar in both species (Table 2), significant differences were found only for the stomatal density (Table 2), been mayor for the adaxial suface in T. minuta and for the abaxial surface in T. terniflora.
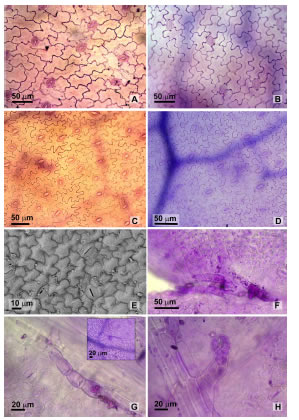
Fig. 3. Leaf paradermal view. A-B: Adaxial epidermis, Tagetes minuta and T. terniflora respectively. C-D: Abaxial epidermis, T. minuta and T. terniflora respectively. E: SEM showing abaxial striated cuticle on T. minuta. F. Tuft of non glandular trichomes at the tooth sinus. G. Uniseriate non glandular trichome. Detail of the insertion. H. Biseriate capitate glandular trichome.
Table 2. Stomatal characteristics in Tagetes sp.
Both species presented polygonal adaxial and abaxial epidermal cells, isodiametric with sinuous-undulating anticlinal walls (Fig. 3 A-D), uniseriate non glandular trichomes and uni to biseriate capitate glandular trichomes. Non glandular trichomes showed 5-8 cells with rounded hyaline apex (164.3 ± 54.8 mm long.), were located along principal veins and clustered in the sinus of some teeth on both epidermal surfaces (Fig. 3 F-G). Glandular trichomes present a stalk, with 3-6 pairs of cells distributed symmetrically or asymmetrically, and a head formed by an apical pair of secretory cells (108.1 ± 16.2 mm long.), they were found in the proximity of the main veins (Fig. 3 H).
Table 2 In transverse section, the midvein at the medium third part of the leaf blade had a uniseriate epidermis. Sub epidermically, two to three layers of angular collenchymas and a collateral vascular bundle immersed in a fundamental parenchyma (Fig. 4 A-B).
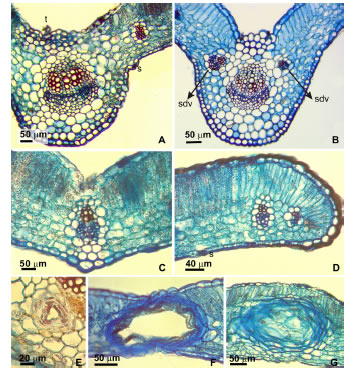
Fig. 4. Leaf transection. A-B: Mid vein, Tagetes minuta and T. terniflora respectively. C: Secondary veins, vascular bundle with collenchymatous cap on the phloem and surrounded by a parenchymatous sheath. D. Marginal minor order vein with parenchymatous sheath. E. Secretory cavity with multilayered epithelium surrounded by a parenchymatous sheath. F. Marginal large oil gland, occupied the entire mesophyll G. Smaller oil gland distributed on the lamina, occupied only part of the mesophyll leaving one layer of palisade parenchyma between the adaxial epidermis and the gland sheath. Abbreviations: t, trichome; s, stomata; sdv; secondary decurrent vein.
Secondary veins presented collateral vascular bundles with collenchymatous cap on the phloem side, surrounded by a parenchymatous sheath transcurrent to both epidermises. Minor order veins presented a one layer parenchymatous sheath (Fig. 4 C-D).
The apical segment of the lamina presented dorsiventral mesophyll, with one layer of palisade parenchyma and four to seven layers of compact spongy parenchyma. Along the blade margin next to the teeth basis on the proximal flank, large oil glands occupied the entire mesophyll (Fig. 4 F). The secretory cavity surrounded by a parenchymatic sheath showed a multilayered epithelium with a large light 170-300 mm partially fill with dense lipidic material (Fig. 4 E-F). Smaller oil gland distributed on the lamina (more frequents in T. terniflora), presented the same characteristics but light of the secretory cavity measured 66-150 mm and they only occupied part of the mesophyll, leaving one layer of palisade parenchyma between the adaxial epidermis and the gland sheath (Fig. 4 G).
In cross section the petioles of both species, were half-oval (near to the stem) to winged shaped (near to the blade) (Fig. 5 A-C). Trichomes were abundant in the adaxial surface with similar features to those previously described for the leaf. Sections showed epidermis uniseriate, one or two layers of angular collenchyma adjacent to the epidermis near the mid vein and margin. The wing mesophyll was dorsiventral formed by one layer of palisade parenchyma and four to seven layers of compact spongy parenchyma (Fig. 5 D). Seven to eight collateral vascular bundles arranged in a straight line or forming a semi circle open to the adaxial surface, were immersed in fundamental parenchyma, one large and central with scleremchymatous caps at phloem and xylem poles, two laterals half as large as the latter and two or four small marginal bundles which extend toward the wing. Three to four schizogenous ducts (23.4 ± 5.8 mm) with uni, rare incomplete biseriate ephitelium, were located in the ground parenchyma, abaxially between the vascular bundles at the phloem level (Fig. 5 E-F).
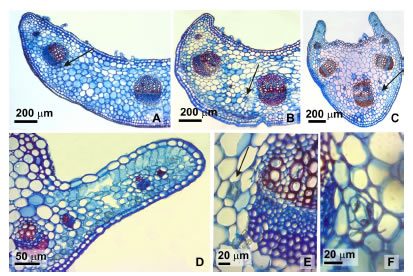
Fig. 5. Petiole transection. A: Half-oval shape near to the stem. B: Half-oval shape middle part of the organ. C: Winged shaped near to the blade. D: Dorsiventral wing. E: Schizogenous duct with uniseriate ephitelium. F: Schizogenous duct with incomplete biseriate ephitelium. Arrows indicate schizogenous ducts.
Stem anatomy
In both species caulinar axis were ramified, green, erect to decumbent. In face view, the epidermis presented anomocytic stomata (30.8 ± 4.0 long. x 20.3 ± 2,7 lat. mm) even or slightly raised regarding the other epidermal cells (Fig. 6 A), polygonal quadrangular cells with thick straight anticlinal cell walls, coated by a thick cuticle. Pluricellular uniseriate non-glandular trichomes (Fig. 6 B) and biseriate (rare uniseriate) glandular trichomes (Fig. 6 C), sparse, similar to the trichomes previously observed at the leaf.
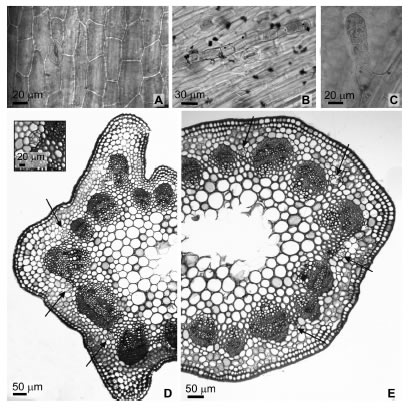
Fig. 6. Stem. A: Paradermal view, polygonal quadrangular epidermal cells with anomocytic stomata. B: Pluricellular uniseriate non-glandular trichome. C: Biseriate capitate glandular trichome. D: Tagetes minuta transection with quadrangular ribs. Detail of schizogenous duct. E: Tagetes terniflora circular transection. Arrows indicate schizogenous ducts.
Transections were quadrangular with evident ribs in T. minuta (Fig. 6 D) to circular in T. terniflora (Fig. 6 E), revealing insipient secondary growth at the stem level analyzed. Epidermis uniseriate, 1-4 layers of sub epidermal angular collenchyma, reinforced at the ribs level in T. minuta, and 1-2 layers of discontinuous angular to laminar collenchyma in T. terniflora. Two to five layers of thin walled parenchymatic cortex. In the cortical parenchyma, alternating with the vascular bundles, secretory ducts (20 ± 5 mm), with uniseriate epithelium surrounded by a parenchymatous sheath, were found. Collateral open vascular bundles form a eustele, with perivascular fibres which may adjoin the bundles and isolate the phloem. The pith, which tends to be collapsed, was formed by thick walled parenchymatic cells, larger than cortex cells.
Root anatomy
Transection of the root at the level analyzed reveals secondary growth in both species. Uniseriate rizodermis, 3-4 layers of thin-walled cortical parenchyma, endodermis with evident casparian band, one layer pericycle, secondary phloem well developed, xylem with diffuse porosity showed abundant fibers. Secretory ducts in the cortex and the phloem were not observed (Fig. 7 A-B).
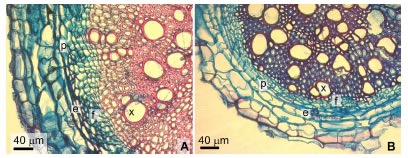
Fig. 7. Root transection at early secondary growth stage. A: Tagetes minuta. B: Tagetes terniflora. Abbreviations: e, endodermis, p, pericycle; f, phloem; x, xylem.
Discussion
Tagetes minuta for Tucumán province was characterized by high amounts of cis-tagetone. Collections of Tm from San Juan, Mendoza and Rio Negro showed dihydrotagetone as major constituent (Gil et al., 2000). Plants coming from Buenos Aires showed cis-tagetone, dihydrotagetone and cis-β-ocimene approximately 20% each; plants obtained from Jujuy showed significant amounts of cis-β-ocimene (26%), cis-tagetone (19%), cis-tagetenone (17%) and trans-tagetenone (12%); and a collection from Salta showed high amounts of trans-β-ocimene (34%) and α-phellandrene (29%) (Gil et al., 2000; Vázquez et al., 2011). Essential oils of Tm collected in various localities of Cordoba province were dominated by cis and trans-ocimene, cis and trans-tagetenone, and cis- and trans-tagetone (Zygadlo et al., 1990) while collections from Chaco contain mostly dihydrotagetone accompanied by β-ocimene, tagetone and tagetenone (Chamorro et al., 2008).
Our two collections of Tt from Tucumán province contained high amounts of cis-tagetone, cis-β-ocimene and dihydrotagetone. Our results are only partly in agreement with data previously reported for collections from Argentina and Perú (Zygadlo et al., 1993; De Feo et al., 2005; López et al., 2011). These facts indicate the existence of a high chemical plasticity in Tagetes as demonstrated by Zygadlo et al. (1990) and Gil et al. (2000, 2002).
In general, different growth stages of the plant exhibit variations in their essential oil composition (Chamorro et al., 2008). Similarly chemotypes can be found as result of intraspecific variations when the essential oil composition of individual plants of the same population are compare, other differences can arise as a result of the plant response to environmental conditions i.e. growth locations (Zygadlo et al., 1990; Gil et al., 2000). Thus further studies with a larger number of individuals from a same population and from other Tagetes populations in different phenological stages are necessary in order to determinate quantitative, qualitative, inter and intraspecific variations in their essential oils composition.
Since the species analyzed in this study do not share the exact same essential oil profile becomes important their correct identification according to the purpose of collection.
Regarding the morpho anatomy, both species were similar. They only showed slight variations in their morphological and anatomical features, such as venation patterns, type and distribution of leaf glands and stem transverse section aspect.
This paper describes the vegetative anatomy of both species, coinciding in the presence of structures associated with the synthesis of essences with those described by Del Fueyo (1986), Simon et al. (2002) and Visintin and Bernardello (2005) for T. minuta; García-Sánchez et al. (2012) and Martínez et al. (2013) for T. lucida, T. lunulata and T. micrantha.
In contrast to the observations of Simon et al. (2002), no secretory canals were observed at root level, possibly due to the stage of development.
This work describes for the first time the morpho-anatomy of vegetative organs of T. terniflora and the venation pattern and presence of non glandular trichomes in the lamina of T. minuta.
Bibliography
1. ADAMS, R. P. 2007. Identification of essential oil components by gas chromatography / mass spectrometry. 4th Ed. Allured Publishing Corp. USA. [ Links ]
2. BARBOZA, G., J. CANTERO, C. NÚÑEZ, A. PACCIARONI & L. ESPINAR. 2009. Medicinal plants: a general review and a phytochemical and ethnopharmacological screening of the native Argentine Flora. Kurtziana 34: 7-365. [ Links ]
3. BII, C. C., G. M. SIBOE & R. K. MIBEY. 2000. Plant essential oils with promising antifungal activity. Nairobi. East Afr. Med. J. 77: 319-322. [ Links ]
4. CABRERA, A. L. 1978. Flora de la Provincia de Jujuy. Compositae. Colección Científica del INTA, Buenos Aires. [ Links ]
5. CHAMORRO, E. R., G. BALLERINI, A. F. SEQUEIRA & G. VELASCO. 2008. Chemical composition of essential oil from Tagetes minuta L. leaves and flowers. J. Arg. Chem. Soc. 96: 80-86. [ Links ]
6. CLEVENGER European Pharmacopeia. 1983. Maissoneuve, S.A, Sainte Ruffinne, I. [ Links ]
7. DE FEO, V., E. URRUNAGA SORIA, R. URRUNAGA SORIA & C. PIZA. 2005. Composition and in vitro toxicity oh the essential oil of Tagetes terniflora HBK. (Asteraceae). Flavour Frag. J. 20: 89-92. [ Links ]
8. DEL FUEYO, G. M. 1986. Ontogenia de las glándulas foliares e involucrales de Tagetes minuta (Compositae). Bol. Soc. Argent. Bot. 24: 403-10. [ Links ]
9. DILCHER, D. L. 1974. Approaches to the identification of angiosperm leaf remains. Bot. Rev. 40: 1-157. [ Links ]
10. DIZEO DE STRITTMATTER, C. G. 1973. Nueva técnica de diafanización. Bolet. Soc. Argent. Bot. 15: 126-129. [ Links ]
11. ELLIS, B. D., C. DALY, L. J. HICKEY, H. R. JOHNSON, J. D. MITCHELL, P. WILF & S. L. WING. 2009. Manual of leaf architecture. Cornell University Press, USA. [ Links ]
12. FLORA ARGENTINA. 2016. http://www.floraargentina.edu.ar [Accessed 4th March 2016]. [ Links ]
13. GARCÍA-SÁNCHEZ, F., M. E. LÓPEZ-VILLAFRANCO, S. AGUILAR-RODRÍGUEZ & A. AGUILAR-CONTRERAS. 2012. Etnobotánica y morfo-anatomía comparada de tres especies de Tagetes que se utilizan en Nicolás Romero, estado de México. Bot. Sci. 90: 221-232. [ Links ]
14. GIL, A., C. M. GHERSA & S. LEICACH. 2000. Essential oil yield and composition of Tagetes minuta accessions from Argentina. Biochem. Systemat. Ecol. 28: 261-274. [ Links ]
15. GIL, A., C. M., GHERSA & S. PERELMAN. 2002. Root thiophenes in Tagetes minuta L. accessions from Argentina: genetic and environmental contribution to changes in concentration and composition. Biochem. Systemat. Ecol. 30: 1-13. [ Links ]
16. HICKEY L. J. 1979. A revised classification of the architecture of dicotyledonous leaves. In: METCALFE C. R., CHALK L. (eds.), Anatomy of the Dicotyledons, pp. 25-39. Volumen 1. Clarendon Press, Oxford, [ Links ]
17. INFOSTAT. 2002. Manual de Usuario. Versión 1.1. Grupo InfoStat, FCA, Universidad Nacional de Córdoba. Primera edición, Ed. Brujas. Argentina. [ Links ]
18. KAPLAN, L. 1958. Historical and ethnobotanical aspects of domestication in Tagetes. Econ. Bot. 14: 200 - 202. [ Links ]
19. KARIMIAN, P., G. KAVOOSI & Z. AMIRGHOFRAN. 2014. Anti-oxidative and anti-inflammatory effects of Tagetes minuta essential oil in activated macrophages. Asian Pac. J. Trop. Biomed. 4: 219- 227. [ Links ]
20. LAWRENCE, B. M. 1985. Essential Oils of the Tagetes genus. Perfum. Flavor. 10: 73–82.
21. LÓPEZ, M. L., N. E.BONZANI & J. A. ZYGADLO. 2009. Allelopathic potential of Tagetes minuta terpenes by a chemical, anatomical, and phytotoxic approach. Biochem. Syst. Ecol. 36: 882–890.
22. LÓPEZ, S. B., M. L. LÓPEZ, L. M. ARAGON, M. L. TERESCHUK, A. C. SLANIS, G. E. FERESIN, J. A. ZYGADLO & A. A. TAPIA. 2011. Composition and anti-insect activity of essential oils from Tagetes L. species (Asteraceae, Helenieae) on Ceratitis capitata Wiedemann and Triatoma infestans Klug. J. Agric. Food Chem. 59: 5286-5292. [ Links ]
23. MACEDO, M. E., R. CONSOLI, T. S. M. GRANDI, A. M. G. ANJOS, A. B. OLIVEIRA, N. M. MENDES, R. O. QUEIROZ & C. L. ZANI. 1997. Sceening of Asteraceae (Compositae) plant extracts for larvicidal activity against Aedes fluviatilis (Diptera: Culicidae). Mem. Inst. Oswaldo Cruz 92: 565-570. [ Links ]
24. MARADUFU, A., R. LUBEGA & F. DORN. 1978. Isolation of (5E)-ocimenone, a mosquito larvicide from Tagetes minuta. Llyodia 41: 181-183. [ Links ]
25. MARTIJEMA, J. D., D. A. GARCÍA, R. H. MARÍN, M. A. PERRILLO & J. P. ZYGADLO. 1998. Anxiogenic like and antidepressant like effects of the essential oil from Tagetes minuta. Fitoterapia 69: 161-164. [ Links ]
26. MARTÍNEZ, M. L., G. BETTUCCI, M. GATUSSO & A. CORTADI. 2013. Caracteres micrográficos analíticos de hojas, tallos, inflorescencias-flores de Tagetes lucida Cav. (Asteraceae – Helenieae). Dominguezia 29: 29-37.
27. METCALFE C. & L. CHALK. 1950. Anatomy of the Dicotyledons. Vol. II. Ed. Clarendon Press, Oxford. [ Links ]
28. NIST. 2005. Mass Spectral Search Program. For the NIST/EPA/NIH Mass Spectral Library. Version 2. [ Links ]
29. PERICH, J. M., C. WELLS, W. BERTSCH & K. E. TREDWAY. 1995. Isolation of the insecticidal components of Tagetes minuta (Compositae) against mosquito larvae and adults. J. Am. Mosquito Control Assoc. 11: 307-310. [ Links ]
30. SADIA, S., S. KHALID, R. QURESHI & A. A. BAJWA. 2013. Tagetes minuta L., a useful underutilized plant of family Asteraceae: a review. Pak. J. Weed Sci. Res. 19: 179-189. [ Links ]
31. SHIRAZI, M. T., H. GHOLAMI, G. KAVOOSI, V. ROWSHAN & A. TAFSIRY. 2014. Chemical composition, antioxidant, antimicrobial and cytotoxic activities of Tagetes minuta and Ocimum basilicum essential oils. Food Sci. Nutr. 2: 146-155. [ Links ]
32. SIMON, P. M., L. KATINAS & A. M. ARAMBARRI. 2002. Secretory structures in Tagetes minuta (Asteraceae, Helenieae). Bol. Soc. Argent. Bot.37:181-191. [ Links ]
33. SOULÉ, J. A. 1993. Tagetes minuta: A potential new herb from South America. In: JANICK, J. & J. E. SIMON (eds.), New crops, pp. 649-654. Wiley, New York. [ Links ]
34. SOULÉ, J. A. 1996. Novel annual and perennial Tagetes. In JANICK, J. (ed.), Progress in New Crops, pp. 546-551. ASHS Press: Arlington, Virginia. [ Links ]
35. STEFANAZZI, N., M. M. GUTIERREZ, T. STADLER, N. A.BONINI & A. A. FERRERO. 2006. Actividad biológica del aceite esencial de Tagetes terniflora Kunth (Asteraceae) en Tribolium castataneeum Herbst (Insecta, Coleoptera, Tenebrionidae). Bol. San. Veg. Plagas 32: 439-447. [ Links ]
36. TERESCHUK, M. L., M. D. BAIGORÍ & L. R. ABDALA. 2003. Antibacterial activity of Tagetes terniflora. Fitoterapia 74: 404-406. [ Links ]
37. VASUDEVAN, P., S. KASHYAP & S. SHARMA. 1997. Tagetes: A multipurpose plant. Bioresour. Technol. 62: 29-35. [ Links ]
38. VÁZQUEZ, A. M., G. I. DEMMEL, S. G. CRIADO, M. L. AIMAR, J. J. CANTERO, L. I. ROSSI & M. I. VELASCO. 2011. Phytochemistry of Tagetes minuta L. (Asteraceae) from Córdoba, Argentina: Comparative study between essential oil and HS-SPME analyses. B. Latinoam. Caribe Pl. 10: 351- 362. [ Links ]
39. VISINTIN, A. M. & G. BERNARDELLO. 2005. Morfología y anatomía floral de Tagetes minuta L. (Asteraceae). Arnaldoa 12: 08-15. [ Links ]
40. ZARLAVSKY, G. E. 2014. Histología vegetal: Técnicas simples y complejas. Sociedad Argentina de Botánica, Buenos Aires. [ Links ]
41. ZYGADLO, J. A., N. R. GROSSO, R. E. ALBURRA & C. A. GUZMAN. 1990. Essential oil variation in Tagetes minuta populations. Biochem. Syst. Ecol. 18: 405-407. [ Links ]
42. ZYGADLO, J.A., A. LAMARQUE, D. M. MAESTRI, C. A. GUZMAN & N. R GROSSO. 1993. Composition of the inflorescence oils of some Tagetes species from Argentina. J. Essent. Oil Res. 5: 679–681.
Recibido 13 de junio de 2016,
aceptado 26 de setiembre de 2016.














