Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO  uBio
uBio
Compartir
Phyton (Buenos Aires)
versión On-line ISSN 1851-5657
Phyton (B. Aires) v.73 Vicente López ene./dic. 2004
ARTÍCULOS ORIGINALES
Nutrient levels of the soil solution obtained by means of suction cups in intensive tomato cultivation (with 5 tables & 1 figure)
Lao MT1*, S Jiménez2, E Eymar3, EJ Fernández1
1Dpto. Producción Vegetal. Escuela Politécnica Superior. Universidad de Almeria. Almeria (España)
2 Centro de Investigación y Formación Agrícola. Departamento de Horticultura. Apdo. 91. El Ejido. Almeria (España)
3Dpto. Química Agrícola, Geología y Geoquímica. Facultad de Ciencias C-VII. Universidad Autónoma de Madrid. Madrid (España)
* Corresponding author, E-mail: mtlao@ual.es
Received 14.I.2003; accepted 18.II.2003
Abstract. Suction cups are the only viable method to obtain in situ the soil solution along the culture in real time (24 h). The soil solution is the objective criterion to modify the nutrient solution, so that it can be an appropriate tool for fertirrigation management. The nutrient levels of the soil solution must be known to establish the appropriate approach to nutrients management. In this work, levels of pH, EC,NO3-,NH4+, H+2PO4- , K+, Ca2+, Mg2+, Na+ and Cl- of the soil solution of three tomato crops were followed for periods 1995/96 and 1996/97 culture estimated in a trial carried out during two years.
Key words: fertirrigation management, nutrient parameters, nutrient solution, tomato production.
Fertirrigation is the combined application of water and fertilisers. The fertirrigation system is at the moment the most rational method, to carry out an optimised fertilisation with due respect of the environment (1).
To satisfy the needs of the crop by fertirrigation, two approaches may be taken:
One, more traditional: the nutrients supply in quantitative sense is adapted to the theoretical needs of the crop. This system requires to use the classic diagnostic methods. A second, one more physiological and more qualitative approach that tries to apply to the crop a physiologically balanced solution, with all elements required by the crop. This is the approach used in soil less culture (2). Table 1 shows nutrient solutions proposed by some authors applying the last criteria.

We have the appropriate technology to apply the nutrient solutions accurately. With a quality of water is applied the necessary nutrient to obtain a theoretical solution. However, keeping in mind the interactions among soil, nutrient solution, soil solution, plants and atmosphere, the final soil solution differs of the required one (3).
The roots of the plants uptake the soil solution, is the direct productive parameter on the crop; it is fundamental to know it as the basis of the fertirrigation management.
The suction cups are the only viable method to extract in situ the soil solution, without proceeding to dilutions of the same one. In consequence, in the cases, where is necessary to know the ionic composition of the soil solution, only this technique can be used because other techniques only measured the EC in situ (4). The first ones to introduce the suction cups technique, were Briggs & McCall, proposing their use like an artificial root to suction the really available water in the soil for the plants.
In this work the nutritional levels of the soil solution are established for tomato crop to use it in management fertirrigation.
MATERIALS & METHODS
This work was carried out in 6 commercial greenhouses type "Almería" with tomato var. Daniela, during two years 95/96 and 96/97. Located in southeastern Spain. The election of these greenhouses was made in function of high production, fertirrigation equipment and the horticulturist's capacity to understand the process being tested.
In each greenhouse a watering subsector was chosen with approximately 2.500 m2, the watering uniformity was studied (up to 90%), 15 suction cups were installed in a random way considering the perimetral edge. The installation and taking of samples are described for (3). The suction cups remained in the soil during the crop.
6 samplings by greenhouse were taken for each year (95/96 and 96/97). Each sampling was constituted by 16 samples, one of nutrient solution and 15 samples of the soil solution. Of each 15 samples of soil solution were taken 20 cc, mixed and homogenised.
Analytical methods described by the Spanish Ministry of Agriculture H2PO-4 , were used to determine the following: pH; EC at 25ºC; NO3- , phosphate and NH4+ (VIS-UV spectrophotometry); K+, Ca2+, Mg2+, Na+ (atomic absorption) & Cl- (photometry).
The production by qualities was registered weekly. The samplings are classified in three periods: period 1: vegetative state (1-90 DAT); period 2: full production (91-250 DAT) and period 3: final of the cycle (251-320 DAT).
An analysis of variance was carried out for the experimental design is multifactorial with three factors: plots, period and year.
RESULTS
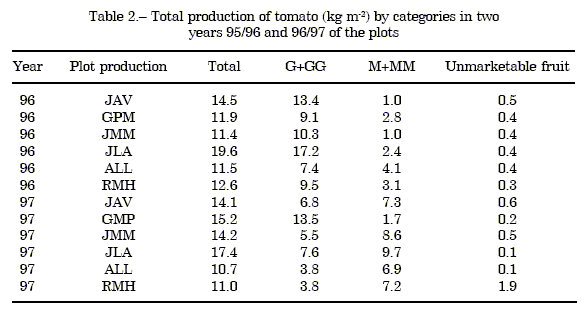
The total productions and categories, as well as the unmarketable fruits corresponding to each plot and year are presented in the Table 2.
Nutrient solution: The Table 3 shown the average values and standard deviations of the nutritional parameters of the nutrient solution by periods for two years.
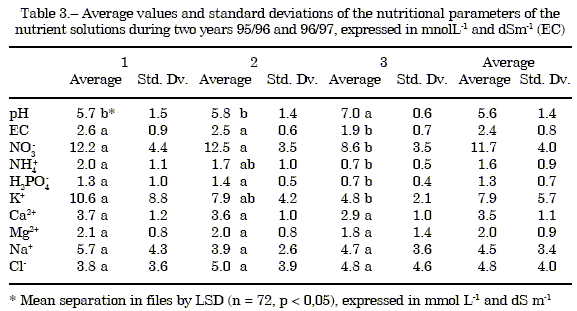
The different nutritional parameters between years don't show significant differences.
In pH: two groups are observed; ones acidifies the nutrient solution up to values of 4.7; the other group presents an average value of 6.4. The pH is higher in period 3 due to the smaller amount of applied acid.
EC: between plots the range values is 1.60 and 3.00 dS m-1. The EC in the period 3 are inferior due to the minor fertilizers contribution (nitrates, ammonium, phosphates and potassium) that is justified for the increment of the transpiration crops and the mayor water consumption.
The concentrations of NO3- , NH4+ and Ca2+ in the nutrient solution, don't differ significantly between plots.
The differences found in the concentrations of Mg2+, Na+ and Cl- depend fundamentally on the quality of the irrigation waters employed. Plots don't show differences (p<0.05).
The concentration of phosphates in the nutrient solution presents differences between plots, a group applies a concentration around 2 mmol L-1 and another group around 1 mmol L-1. In period 3 the concentration is lower.
Great variability exists in the concentration of K+ due to the lack of clear criteria for application. Between periods there are differences diminishing the quantity applied along the crop.
Soil solution:In table 4 the average values and standard deviations of the nutritional parameters of the soil solution are presented by periods for both years.

Between years the nutritional parameters don't differ significantly.
The pH of the soil solution doesn't differ significantly between plots, although the nutrient solutions employed present different acidity, neither are there pH differences between different productive periods, due to the buffer effect of the soil.
There are no differences between plots, it presents them between productive periods; in period 1 is higher, due to a larger evapotranspiration rate but fertilizers accumulation is not observed.
The concentration of Nitrates differs widely. There exist 3 groups where the concentrations vary from 7.20 to 18.70 mmol L-1; however the nutrient solutions didn't differ between plots. This indicates that the NO3- concentration is not be the appropriate parameter to standardize fertirrigation (Figure 1).

Between productive periods significant differences are not observed (p <0.05) in the concentration of NO3- , H2PO4- and K due fundamentally to the high degree of variability.
The average levels found in the soil solution can be compared with the nutrient solutions applied in soil less culture; we find that (6) proposes a level of 20.3 mmol L-1, (7) recommends levels in the substrate solution of 11 mmol L-1.
In ammonium concentration there are no differences between plots but there are between productive periods, diminishing the concentration in period 2.
The differences between the soil solution and the nutrient solution could be due to a nitrification process, or the volatilization of ammonia. The values recommended in nutrient solutions are 0.5 mmol L-1(8 & 9) and 2 mmol L-1(10). The use of ammonium nitrate has been recommended traditionally in soil cultivation (11).
The concentration of phosphates in the soil solution differs significantly between plots, although the range of concentrations is very narrow (0.05 - 0.33 mmol L-1). Nutrient solutions present higher values (average 1.26 mmol L-1). This effect of phosphates retrogradation can be due to pH (12) in recirculating system observes the same phenomenon, a decrease in phosphates concentration correlated with an increase of pH (13) find levels in cultivated soil between 0.1-0.01 mmol L-1. In this work with the levels found, the crops did not show deficiency symptoms; nevertheless it seems more convenient to know the available levels of P by means of the Olsen method to management the fertilization.
The concentration of potasium in the soil solution presents differences between plots with values in the range of 3.2 to 8.7 mmol L-1.
The concentration of calcium in the soil solution ranges between plots with values in the range of 3.3 and 6.9 mmol L-1. The concentration of Ca in the soil solution is low in period 3. According to references, the values obtained in this trial can be considered acceptable or high.
One of the important approaches are the balances between cations; the Ca shows a low ratio with K and Mg (35: 39: 26) in comparison with(14) (45: 35: 20); (15) suggest a value of 2 and 2.97 as ratio of Ca/Mg uptake for the vegetative and productive periods respectively and (16) finds a ratio of uptake of 2 for 2.46; the ratio of concentrations in the soil solution presents an average value of 1.35 in our trial and corresponds to a high value of Mg more than to a low application of Ca (17) doesn't find differences in the B.E.R. in tomato crop, as manifestation of the metabolic imbalance of Ca, with values of the ratio Ca/Mg in nutrient solutions superiors to one. Nevertheless the level of applied calcium is conditioned by the level of magnesium in the soil solution. The coefficient Ca/Mg should be adjusted, to diminish the application of Ca, without producing imbalances, because the uptake is very good at this pH.
The concentration of magnesium doesn't present differences between plots. The concentrations are higher in the nutrient solution due to the enrichment by the soil. There also exist differences between periods, diminishing toward the end of the crop. (18) considers that under Almería conditions it isn't necessary to apply Mg.
Sodium & chlorides: the concentration of Na+ and Cl- in the soil solution present significant differences between plots due to the quality of the irrigation water, and between periods with superior values in the periods 1 & 3. The Cl don't show variations differences between periods.
Ratios between the concentrations in the nutrient solution and in the soil solution: Table 5 shows the ratios between concentrations in nutrient solution and in soil solution and figure 1 shows the average level of different parameters in nutritional and soil solutions. The ratios of ammonium, phosphates and potassium are superior to 1, it supposes a net consumption due to uptake by plant, retrogradation in the soil or/and leaching. For the rest of the analysed parameters these ratios is smaller than one, it implies a net accumulation due to a low uptake or contribution by the soil.

CONCLUSIONS
* The levels of the different nutritients parameters of the soil solution in the productive periods are presented in the table 4, as basic criteria for the management fertirrigation in tomato crop in the Mediterranean area.
* The soil solution presents a stable pH due to the tampon effect of the soil that doesn't modify the acidity of the nutrient solution.
* The EC should be managed jointly by the nutritional parameters and by irrigation.
* The application of nitrates should be managed in function of soil solution levels because the nitrates are very mobile.
Level of phosphates in the soil are very low in relation to the nutrient solution, without deficiency symptoms in the crop. The phosphates buffer in soil should be evalueted by means of the Olsen method (basic soils). The form, time and amount of application ofphosphates should be carefully studied.
1.Cadahía C, Fertirrigación. Conceptos básicos. I Congreso ibérico y III Nacional de Fertirrigación (1997) 46 [ Links ]
2.Domínguez A, In Fertirrigación, Edit. Mundi-Prensa (1996) [ Links ]
3.Lao MT, Gestión del fertirriego de los invernaderos de Almería mediante el uso de sondas de succión. Tesis doctoral, Universidad de Almería (1998) [ Links ]
4.Grossmann J, P Udluft, J Soil Sci 42 (1991) 83 [ Links ]
5.MAPA, Métodos oficiales de análisis. Publication´s Serv of MAPA, Ed. Madrid, Spain (1986) [ Links ]
6.White RAJ, Commercial use of soilless culture for tomatoes in New Zealand. ISOSC Proceedings of 8thInternat Congr Soilless Cult Hunters rest (1992) 483 [ Links ]
7.Jeannequin B, in Fertilisation de la tomate en culture hors sol en région méditerranéenne, en Les cultures hors sol. INRA (1987) 235 [ Links ]
8.Martínez E, M García. Cultivos sin suelo: Hortalizas en clima mediterráneo, Horticultura Ed. (1994) 41 [ Links ]
9.Sonneveld C, Growing cucumbers and tomatoes in rockwool. ISOSC, Proceedings of Fifth International congress on soilless culture, Wageningen (1980) 253 [ Links ]
10.Coïc Y, C Lesaint, La nutrition minerale et en eau des plantes en horticulture avancé. Le document Technique de la S.C.P.A. Mulhouse (France) (1975) [ Links ]
11.Palomar F, In Fertirrigación de cultivos hortícolas en invernadero. Hoja divulgadora 15/92. Junta de Andalucía. Consejería de Agricultura y pesca (1993) [ Links ]
12. García M, Eficacia en el uso de la fertirrigación recirculante en cultivos hortícolas sin suelo bajo condiciones de clima semiárido. Tesis doctoral. Univ de Almería (1997) [ Links ]
13.Mengel K, EA Kirkby, Principles of plant nutrition. Internat Potash Institute Bern, Switzerland, 3rd ed. (1982) [ Links ]
14. Steiner AA, The selective capacity of plants for ions and its importance for the composition and treatment of the nutrient solution. ISOSC 1980, Proceedings Wageningen (1980) 83 [ Links ]
15.Brun R, D Blanc, Cinétique de l´absorption hydrique et minérale. Composition ionique des solutions, en Les cultures hors sol. INRA (1987) 203 [ Links ]
16. Gertsson E, Nutrient uptake by tomatoes grown in hydroponics. Acta horticulturae 401 (1995) 351 [ Links ]
17.Segura-Pérez ML, Fertirrigación de cultivos hortícolas en condiciones salinas con sistemas enarenados y sustratos alternativos. Comarca Agrícola de Almería. Tesis doctoral, Univ autónoma de Madrid, Facultad de Ciencias (1995) [ Links ]
18. González P, La fertilización mediante el riego localizado. Curso Internacional sobre Agrotecnia del cultivo en invernaderos, F.I.A.P.A. Almería (1991) 223 [ Links ]














