Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO  uBio
uBio
Compartir
Phyton (Buenos Aires)
versión On-line ISSN 1851-5657
Phyton (B. Aires) v.76 Vicente López ene./dic. 2007
ARTÍCULOS ORIGINALES
Light and temperature effects on seed germination of four native species of Mediterranean high mountains (Spain) (With 1 Table & 1 Figure)
Efectos de la luz y de la temperatura sobre la germinación de semillas de cuatro especies autóctonas de alta montaña Mediterránea (España) (Con 1 Tabla y 1 Figura)
Serrano-Bernardo F1, JL Rosúa1, M Díaz-Miguel2
1 Departamento de Botánica. Edificio Politécnico. Universidad de Granada. Campus Fuentenueva, s/n. 18071-Granada, España. e-mail: fserber@ugr.es
2Departamento de Fisiología Vegetal. Facultad de Ciencias. Universidad de Granada. Campus Fuentenueva, s/n. 18071-Granada, España.
Recibido/Received: 02.11.2006. Aceptado/Accepted 30.03.2007.
Abstract. The Mediterranean region is considered one of the most important zones worldwide in terms of plant diversity. Sierra Nevada (S Spain) is a unique mountain massif within the high-mountain systems of the Mediterranean. It is of great floristic importance and very rich in endemic species. However, it requires numerous tasks for restoring plant cover due to the constant anthropic degradation, especially because of the ski station located near the second-highest peak of the mountain. One of the restoration efforts consists of trying to reproduce, in the laboratory, the ideal conditions to pre-germinate seeds of some of the species most threatened by these perturbations. This is to transplant the seedlings afterwards in the mountain, thereby contributing to the recovery of these species. With this purpose, a germination experiment was performed, under controlled conditions, with seeds of Genista versicolor Boiss. Hormathophylla spinosa (L.) P. Küpfer, Reseda complicata Bory, and Thymus serpylloides Bory. These four species, native to the Mediterranean high mountains, were germinated under constant darkness, 12 or 24 h light followed by constant darkness, and constant light, at temperatures of 15 ºC or 25 ºC. The germination process was monitored during 15 days. Results were species-dependent: some species germinated easier under darkness at low temperatures, such as Genista versicolor, whereas Thymus serpylloides presented maximum germination under constant light to a higher temperature.
Key words: Germination; Plant-cover restoration; Mediterranean high mountains; Sierra Nevada; Light; Temperature.
Resumen. La región mediterránea es considerada como una de las zonas más importantes a nivel mundial en cuanto a diversidad vegetal. Sierra Nevada (Sur de España) constituye un área importante dentro de esta región. Es un macizo montañoso único dentro de los sistemas de alta montaña mediterránea, de gran importancia florística y muy rico en endemismos. Esto hace necesarias numerosas tareas de restauración de la cubierta vegetal, debido a las constantes alteraciones antrópicas que sufre, especialmente como resultado de la estación de esquí que hay en él. Una de estas tareas de restauración consiste en intentar reproducir, en laboratorio, las condiciones ideales para pregerminar semillas de algunas de las especies más amenazadas por estas alteraciones. Esto permitiría posteriormente transplantar dichas especies al campo, contribuyendo a su recuperación. En el presente trabajo, se llevó a cabo un experimento de germinación, en condiciones controladas, con semillas de Genista versicolor Boiss. Hormathophylla spinosa (L.) P. Küpfer, Reseda complicata Bory, and Thymus serpylloides Bory. Estas especies, autóctonas de alta montaña mediterránea, fueron puestas a germinar en condiciones de Oscuridad Constante, 12 ó 24 horas de luz y posterior traslado a Oscuridad Constante, y Luz Constante, a temperaturas de 15 ºC o 25 ºC. Durante 15 días se siguió la evolución de la germinación. Los resultados obtenidos fueron heterogéneos. Hubo especies con mayor facilidad para germinar en oscuridad a bajas temperaturas, como Genista versicolor, en oposición a Thymus serpylloides, que presentó un máximo porcentaje germinativo con luz constante a la temperatura más elevada.
Palabras clave: Germinación; Restauración de la cubierta vegetal; Alta montaña Mediterránea; Sierra Nevada; Luz; Temperatura.
INTRODUCTION
The Mediterranean region is considered one of the most important zones worldwide regarding plant diversity (Médail & Quézel, 1997; Myers et al., 2000; Malcolm et al., 2006; Médail & Diadema, 2006; Vogiatzakis et al., 2006), including Mediterranean high-mountain areas (Väre et al., 2003; Giménez-Benavides et al., 2005). One of these mountain systems is Sierra Nevada (S Spain), with the only high-mountain national park in Europe, covering 86,208 ha. Also, this is the most southern European massif, and its proximity to Africa gives it particular climatic characteristics. These characteristics, in combination with its altitude (among other factors) make it the only Mediterranean mountain of Spain that has five bioclimatic levels (Rivas-Martínez, 1990). This contributes to explain the high biodiversity of this massif. Thus, Sierra Nevada has more than 2000 vascular plant species and subspecies, constituting more than 30% of the flora of the Spanish peninsula. Of this high number of vascular plant species, more than 80 are endemic, especially in the peak zones, where the percentage of endemic species exceeds 30%. In exclusive ecological niches such as the rocky or gravely areas, over 80% of the species are endemic (Blanca, 2002). In these fragile areas, stands the Sierra Nevada ski station, the most southern ski resort of Europe. It receives a great number of skiers and visitors year round: roughly a million people per year for a surface area of around 350 ha. Works preparing the runs have seriously perturbed the ecosystems of these slopes, resulting in a major loss of biodiversity and increased soil erosion. It is essential to restore the plant cover for preventing further ecosystem degradation and loss of biodiversity.
Chambers (1997) and Macyk (2000) have described the alpine ecosystem as one of the most difficult for restoring disturbances, because of the exclusive physical determinants of the environment, such as low temperatures, high levels of solar radiation, persistent winds, irregular topography and steep slopes. Among these determinants, temperature and light play a major role. Temperature regulates germination in three ways (Bewley & Black, 1994): 1) by determining the capacity and percentage of germination; 2) by eliminating primary and/or secondary dormancy; and 3) by inducing secondary dormancy. The optimal germination temperature for most seeds which are not in dormancy is 25 to 30 ºC. However, temperature ranges for germination depend largely on the (1) period of the year in which plants complete their life cycle and (2) geographic origin of the species (Besnier, 1989). Thus, seeds usually require a thermal periodicity to germinate: a cold season (of activation), between 0 and 10 ºC (stratification), alternating with another one where the embryonic axis grows at intermediate temperatures (20-30 ºC). In fact, low temperatures can break dormancy of some seeds (e.g. woody plants, cereals), and are necessary for germination at higher temperatures (Bewley & Black, 1982). High temperatures sometimes reduce dormancy of some seeds but induce secondary dormancy in others. Therefore, the response of seeds to these conditions can be essential for the regeneration of communities after a perturbation.
In addition, light is also important for seed germination. Many species respond to the environment with optimal growth and development according to the light they receive (Maloof et al., 2000). Some seeds germinate similarly in light and darkness (Baskin & Baskin, 1988), while others do it more readily either under light (Colbach et al., 2002) or darkness conditions (Thanos et al., 1989). Also, light requirements for germination can vary with temperature. It has been demonstrated that some species need a constant temperature and light to germinate and others can germinate either under light or darkness conditions but need temperature fluctuations (Felippe, 1978). In other species, stratification (Farmer et al., 1984) or high temperatures (Amritphale et al., 1989) replace light requirements for germination.
Our objective was to determine some of the light and temperature requirements for germination of the study species under laboratory conditions. This would contribute to know the required light and temperature conditions necessary for optimum germination of these species in soils of Sierra Nevada. After seed germination in the laboratory, seedlings were transplanted to the natural environment in an effort to improve methods of restoring plant cover in degraded areas.
MATERIALS AND METHODS
Seeds from four endemic species of the Betic Sierras and Sierra Nevada were used in this study: Genista versicolor Boiss., Hormathophylla spinosa (L.) P. Küpfer), Reseda complicata Bory, and Thymus serpylloides Bory. They were collected from different areas near the ski station, between 2000 and 3000 m in altitude. Before the study initiation, similar-size seeds from each species were kept in darkness at 4 ºC for at least three months. Thereafter, seeds were sorted out for uniform size and similar external characteristics, discarding those with obvious alterations or malformations.
Previous to the germination test, a sample of seeds was tested for viability, applying the tetrazolium test (AOSA, 2000). It showed a positive staining reaction in 80-90% of the seeds. All seeds were surface sterilized by immersion in 1% sodium hypochlorite for 5 min, and washed afterwards with distilled water. Thereafter, petri dishes 10 cm in diameter were prepared with filter paper (both previously sterilized). Sixty seeds were placed on each of 6 petri dishes (replicates; 10 seeds x dish), 10 ml of distilled water was added, and the following four treatments were applied: Constant Darkness; Light 12 h followed by constant darkness; Light 24 h followed by constant darkness; and Constant Light. Germination tests were conducted in a cultivation chamber having fluorescent tubes of white light and a light/darkness timer, at 15 ºC or 25 ºC. Dishes were sampled daily during 15 days. A seed was considered germinated when the radicle emerged. Days to reach half the maximum germination percentage (T50 : estimate of germination rate) was determined in each case. Distilled water was added whenever moisture loss was detected.
Data were analyzed using one-way ANOVA. When F tests were significant, means were compared using the Duncan's multiple-range test (Duncan, 1955).
RESULTS
1) Temperature: 15 ºC
In G. versicolor, germination percentages under Darkness or with a pre-treatment of 12 h Light were more than 18% greater than those under a pre-treatment of 24 h Light (Fig. 1A). Germination rate of seeds germinated in Darkness was greater than that in the other treatments; these seeds presented a T50 value about half of that on seeds germinated with Constant Light (Table 1). Hormathophylla spinosa seeds, in all cases where light was involved, registered germination values greater than those of seeds germinated under Constant Darkness. Values increased by 10% in the case of 12 h Light, 16% under the pre-treatment with 24 h Light, and 28% under Constant Light, in relation to seeds kept in Darkness (Fig. 1B; Table 1). Seed germination was significantly greater (p<0.05) under Constant Light than under Constant Darkness (Table 1). Similarly, results for R. complicata showed that seeds in the light treatments reached higher germination percentages than under Constant Darkness, although differences were not significant in any case (p>0.05). Treatments with 12 h and 24 h Light reached the higher germination rates (48% and 52% germination) than under Constant Light and Constant Darkness, respectively (Fig. 1C). Treatments with 12 and 24 h Light also showed faster germination (T50= 3.8 days) than those under Constant Light and Constant Darkness (T50=8.1 and 3.9 days, respectively; Table 1). Finally, T. serpylloides reached 82% germination under any light pre-treatment. This value was 24% higher than that for seeds germinated in Constant Darkness. However, differences were non-significant (p>0.05) in any case. When the evolution of germination was studied, no significant differences (p>0.05) were found among Constant Light, Constant Darkness and Light pre-treatments of 12 and 24 h (Figure 1D).
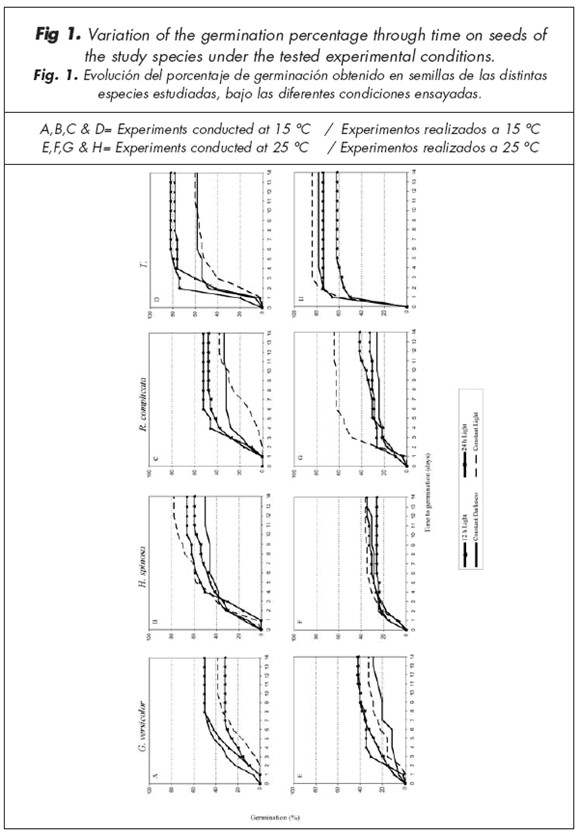

2) Temperature: 25 ºC
No differences (p>0.05) in germination percentages were found among treatments in any species at this temperature (Table 1). When G. versicolor seeds were germinated at 25 ºC, or 15 ºC, there were no significant differences (p>0.05) in final germination percentage in the Constant Light and 24 h pre-treatment (Table 1). Seeds in the 12 h pre-treatment showed greater (p<0.05) germination percentages at 25 than 15 ºC (Table 1). The reverse was true (p<0.05) under Constant Darkness (Table 1). All treatments with light presented curves situated above those corresponding to seeds germinated in Constant Darkness (Fig. 1E). Seeds of H. spinosa had greater (p<0.05) germination percentages at 15 than 25 ºC under all study treatments (Table 1, Fig. 1F). Germination percentages for R. complicata were greater (p<0.05) at 25 than 15 ºC under the Constant Light or Constant Darkness treatment (Table 1; Fig. 1G). In addition, at 25 ºC, all treatments had lower T50 values than at 15 ºC (Table 1). Seeds of T. serpylloides under Constant Light and Constant Darkness showed greater (p<0.05) germination percentages at 25 than 15 ºC (Table 1; Fig. 1H). However, seeds of T. serpylloides pre-teated with 12 h Light showed greater (p<0.05) germination percentages at 15 than 25 ºC (Table 1).
DISCUSSION
Mediterranean high-mountain ecosystems are especially sensitive to disturbances, such as overgrazing and agriculture, increase in tourist pressure or factors which act synergycally (Lorite et al., 2007). They are also vulnerable to natural climatic variations (Hódar & Zamora, 2004). In this sense, interactions between light and temperature can determine the germination time and seed capacity to germinate of many species. Thus, some seeds require light at a certain temperature but not at others (Pons, 1992). Light characteristics that affect germination include photon length, quality, and irradiance reaching the seed (Casal et al., 1998). For many species in different habitats, light responses are measured by perception via the phytochrome (Vázquez-Yanes et al., 1996; Casal & Sánchez, 1998).
In addition, it is generally recognized that high-mountain species do not need a specific temperature to promote germination (Körner, 2003), although most of them usually prefer high temperatures (Billings & Mooney, 1968). This also seems to occur in the case of Mediterranean high-mountain species in the oromediterranean level (Giménez-Benavides et al., 2005). However, this does not apply to the four species of the present study, which is situated also at this same bioclimatic level. This is very likely because of the extraordinary combination of natural factors that mix and interact in the ecosystems of this type of Mediterranean mountain. In this sense, Sierra Nevada is a zone where the solar radiation is especially intense during the dry months. This produces great thermal gradients during the day between the atmosphere, which is always cold, and an overheated soil. This is especially true because rocky soils present dark colours, which reflect little the incident radiation (Ortega, 1989). The fruiting period of the study species spans from the end of July (H. spinosa) to the end of September (R. complicata). This implies, as occurs in other Mediterranean highmountain species (Angosto & Matilla, 1994), that light sensitivity of each species can be a major factor inducing possible secondary dormancy (Baskin & Baskin, 1998). This is because of the few weeks that the seeds have to germinate after snowmelt, and the harsh climatic conditions of the Mediterranean high mountains (Giménez-Benavides, 2005). Except in specific cases, light can also slow down the germination process. The fact that seeds of G. versicolor need light at 25 ºC but not at 15 ºC to activate germination may be related to an increase of the ABA levels at this latter temperature, which can inhibit growth that precedes germination (Karssen, 1976).
The study species are chamephytes (the replacement buds are near the ground surface during the unfavourable season). This, and factors such as seed weight and size, can cause any amount of light to be sufficient for promoting germination in a very short response-time period (Casal et al., 1997). This happened with R. complicata and T. serpylloides seeds at 15 ºC, and can make differences hardly appreciable between the exposure to light for 12 or 24 h. The exception was G. versicolor at 15 ºC, where the germination percentages reached after the seeds were exposed to light for 24 h (identical to the results under Constant Darkness) did not appear to follow the general pattern of the other species.
The cold stratification in darkness, used to increase germination percentages in many high-mountain species (Bewley & Black, 1982; Baskin & Baskin, 1998) helps to increase light sensitivity, especially in seeds with permeable and semi-permeable coats (Hartmann et al., 1997). This can be explained by the greater easiness of the seeds of H. spinosa, R. complicata, and T. serpylloides to germinate in the presence of light. It could be that G. versicolor seeds do not respond efficiently to darkness stratification because either the lack or excess of time, and this could have determined secondary dormancy (skotodormancy). This would involve less light sensitivity (Steadman, 2004), giving rise to the germination percentages found when seeds were exposed to light. Nevertheless, it would be necessary to increase knowledge of these four species, specially in those that require light to germinate, and in particular, in the study of the phytochrome response (specially Pfr). This, when present in sufficient quantities, can stimulate germination (Hillhorst, 1998; Vleeshouwers & Bouwmeester, 2001). Phytochrome may also be one of the factors responsible for the relationship between temperature and light-exposure time in the germination process of these Mediterranean high-mountain plants.
AKNOWLEDGEMENTS
Our appreciation to Oficina del Parque Nacional-Natural de Sierra Nevada for donating the seeds of the different study species, and to Mr. David Nesbitt for translating the original manuscript into English. This work has been granted by the Plan Propio de Investigación de la Universidad de Granada to Dr. Francisco Serrano-Bernardo.
REFERENCES
1. Amritphale, D., S. Iyengar & R.K. Sharma (1989). Effect of light and storage temperature on seed germination in Hygrophila auriculata (Schumach.) Haines. Journal of Seed Technology 13: 39-43. [ Links ]
2. Angosto, T. & A.J. Matilla (1994). Modifications in seeds of Festuca indigesta from two different altitudinal habitats. Seed Science Technology 22: 319-328. [ Links ]
3. AOSA, Association of Official Seed Analysts (2000). Tetrazolium Testing Handbook, no. 29, Las Cruces, New Mexico. [ Links ]
4. Baskin, C.C. & J.M. Baskin (1988). Germination ecophysiology of herbaceous plant species in a temperature region. American Journal of Botany 75: 286-305. [ Links ]
5. Baskin, C.C. & J.M. Baskin (1998). Seeds: Ecology, biogeography, and evolution of dormancy and germination. Acad. Press, San Diego, California. 666 p. [ Links ]
6. Besnier, F. (1989). Semillas: Biología y Tecnología. Ed. Mundiprensa, Madrid. 524 p. [ Links ]
7. Bewley, J.D. & M. Black (1982). Physiology and biochemistry of seeds in relation to germination. Vol. 2. Viability dormancy and environmental control. Springer, Berlín. [ Links ]
8. Bewley, J.D. & M. Black (1994). Seeds: Physiology of development and germination. 2nd ed. Plenum Press, NY. 445 p. [ Links ]
9. Billings, W.D. & H.A. Money (1968). The ecology of artic and alpine plants. Biological Reviews 43: 481-529. [ Links ]
10. Blanca, G. (2002). Flora amenazada y endémica de Sierra Nevada. Junta de Andalucía. Ed. Univ. Granada, Granada. 410 p. [ Links ]
11. Casal, J.J. & R.A. Sánchez (1998). Phytochromes and seed germination. Seed Science and Research 8: 317-319. [ Links ]
12. Casal, J.J., R.A. Sánchez & J.F. Botto (1998). Modes of action of phytochromes. Journal of Experimental Botany 49: 127-138. [ Links ]
13. Casal, J.J., R.A. Sánchez & M.J. Yanovsky (1997). The function of phytochrome A. Plant, Cell and Environment 20: 813-819. [ Links ]
14. Chambers, J.C. (1997). Restoring alpine ecosystems in the western United States: environmental constraints, disturbance characteristics, and restoration success. In: Urbanska, K.M., Webb, N.R. and Edwards, P.J. (eds), pp. 161-187. Restoration Ecology and Sustainable Development, Cambridge Univ. Press, UK. 397 p. [ Links ]
15. Colbach, N., B. Chauvel, C. Dürr & G. Richard (2002). Effect of environmental conditions on Alopecurus myosuroides germination. I. Effect of temperature and light. Weed Research 42: 210-221. [ Links ]
16. Duncan, D.B. (1955). Multiple range and multiple F tests. Biometrics 11: 1-42. [ Links ]
17. Farmer, R.E., P. Charrette, I.E. Searle & D.P. Trajan (1984). Interaction of light, temperature and chilling in the germination of black spruce. Canadian Journal of Forest Research 14: 131-133. [ Links ]
18. Felippe, G.M. (1978). Estudos de germinacao, crescimento e floracao de Bidens pilosa L. Revista do Museo Paulista 25: 183-217. [ Links ]
19. Giménez-Benavides, L., A. Escudero & F. Pérez-García (2005). Seed germination of high mountain Mediterranean species: altitudinal, interpopulation and interannual variability. Ecological Research 20: 433-444. [ Links ]
20. Hartmann, K, C. Kroo â & A. Mollwo (1997). Phytochrome-mediated photocontrol of the germination of the Scentless Mayweed, Matricaria inodora L. and its sensitization by nitrate and temperature. Journal of Photochemistry and Photobiology B-Biology 40: 240-252. [ Links ]
21. Hillhorst, H.W.M. (1998). The regulation of secondary dormancy. The membrane hypothesis revisited. Seed Science and Research 8: 77-90. [ Links ]
22. Hódar, J.A. & R. Zamora (2004). Herbivory and climatic warning: a Mediterranean outbreaking caterpillar attacks a relict, boreal pine species. Biodiversity and Conservation 13: 493-500. [ Links ]
23. Karssen, C.M. (1976). Two sites of hormonal action during germination of Chenopodium album seeds. Physiologia Plantarum 36: 264-270. [ Links ]
24. Körner, C. (2003). Alpine plant life - Functional Plant Ecology of High Mountain Ecosystems. Springer, Heidelberg. 344 p. [ Links ]
25. Lorite, J., F.B. Navarro & F. Valle (2007). Estimation of threatened orophytic flora and priority of its conservation in the Baetic range (S. Spain). Plant Biosystems 141: 1-14. [ Links ]
26. Macyk, T.M. (2000). Reclamation of alpine and subalpine lands. In: Barnhisel, R.L., Darmody, R.G. and Lee Daniels, W.L. (eds), pp. 537-566. Reclamation of drastically disturbed lands. Am. Soc. Agr. Publ., Madison, WI. [ Links ]
27. Malcolm, J.R., C. Liu, R.P. Nelson, L. Hansen & L. Hannah (2006). Global warming and extinctions of endemic species from biodiversity hotspots. Conservation Biology 20: 538-548. [ Links ]
28. Maloof, J.N., J.O. Borevitz, D. Weigel & J. Chory (2000). Natural variation in phytochrome signaling. Seminars in Cell and Developmental Biology 11: 523-530. [ Links ]
29. Médail, F. & K. Diadema (2006). Mediterranean plant biodiversity and human impact: Macro and micro-regional approaches. Annales de Geographie 115: 618-640. [ Links ]
30. Médail, F. & P. Quézel (1999). Biodiversity hotspots in the Mediterranean Basin: setting global conservation priorities. Conservation Biology 13: 1510-1513. [ Links ]
31. Myers, N., R.A. Mittermeier, C.G. Mittermeier, G.A. Fonseca & J. Kent (2000). Biodiversity hotspots for conservation priorities. Nature 403: 853-858. [ Links ]
32. Ortega, F. (1989). Principales aspectos climáticos de Sierra Nevada. In: Chacón, J., Ortega, F., Pulido, A. & Rosúa, J.L. (eds), pp. 25-31. Avance del estudio de impacto ambiental del anteproyecto de reparación y acondicionamiento de la pista del río para el Campeonato del Mundo de esquí alpino con instalación de nieve artificial y nuevos remontes, Univ. Granada. [ Links ]
33. Pons, T.L. (1992). Seed responses to light. In: Fenner, M. (ed), pp. 259-284. Seeds: the ecology of regeneration in plant communities. CAB Int., UK. [ Links ]
34. Rivas-Martinez, S (1990). Bioclimatology and biogeography of West Europe. In: Duplessey, V.C., Poms, A. and Fantecli, R. (eds), pp. 225-246. Climate and global change. Proceedings of European School of Climatology and Natural Hazards. European Commission, Brussels. [ Links ]
35. Steadman, K.J. (2004). Dormancy release during hydrated storage in Lolium rigidum seeds is dependent on temperature, light quality, and hydration status. Journal of Experimental Botany 55: 929-937. [ Links ]
36. Thanos, C.A., K. Georghious & F. Skarou (1989). Glaucium flavum seed germination: An ecophysiological approach. Annals of Botany 63: 121-130. [ Links ]
37. Väre, H., C. Lampinen, C. Humphries, P. Williams (2003). Taxonomic diversity of vascular plants in the European alpines areas. In: Nagy, L. Grabherr, G., Körner, C., and Thompson, D.B.A. (eds), pp. 133-148. Alpone biodiversity in Europe. Ecological studies, vol. 167. Springer, Berlín. [ Links ]
38. Vázquez-Yanes, C., M. Rojas-Arechiga, M.E. Sánchez-Coronado & A. Orozco-Segovia (1996). Comparison of light-regulated seed germination in Ficus spp. and Cecropia obtusifolia: ecological implications. Tree Physiology 16: 871-875. [ Links ]
39. Vleeshouwers, L.M. & H.J. Bouwmeester (2001). A simulation model for seasonal changes in dormancy and germination of weed seeds. Seed Science and Research 11: 77-92. [ Links ]
40. Vogiatzakis, I.N., A.M. Mannion & G.H. Griffiths (2006). Mediterranean ecosystems: problems and tools for conservation. Progress in Physical Geography 30: 175-200. [ Links ]














