Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO  uBio
uBio
Compartir
Phyton (Buenos Aires)
versión On-line ISSN 1851-5657
Phyton (B. Aires) v.77 Vicente López ene./dic. 2008
ARTÍCULOS ORIGINALES
Phototoxic compounds and biological activity of extracts from Eupatorium morifolium Mill. (Asteraceae) (With 5 Tables)
Compuestos fototóxicos y actividad biológica de los extractos de Eupatorium morifolium Mill. (Asteraceae) (Con 5 Tablas)
Pérez-Amador1 MC, V Muñoz Ocotero1, F García Jiménez2
1 Facultad de Ciencias, Departamento de Biología.
2 Instituto de Química. Universidad Nacional Autónoma de México. Ciudad Universitaria. 04510. México, D.F. México.
Address Correspondence to: Dr. Maria Cristina Pérez-Amador; Facultad de Ciencias. UNAM. Circuito Exterior s/n Ciudad Universitaria. C.P. 04510. México D.F.; e-mail: perez_amador@yahoo.com, mcpa@hp.fciencias.unam.mx
Recibido/Received 21.09.2007. Aceptado/Accepted 27.11.2007.
Abstract. Leaves of Eupatorium morifolium Mill. were analyzed for phototoxic compounds, and the toxicity of their hexane extract was evaluated against Bacillus subtilis. The phototoxic compounds were analyzed by TLC. In the plate, pale blue spots indicated the presence of these compounds. This was confirmed by the UV extract spectrum and the antibacterial activity against Bacillus subtilis. The biological activity of the leaf extracts (hexane, ethyl acetate or methanol) was also studied. The antiinflammatory activity was determined using the edema test on mouse ears, and the bactericide activity was assayed against Bacillus subtilis and Escherichia coli. A strong anti-inflammatory activity was shown by the leaf extracts. The ethyl acetate extract exhibited the strongest bactericide activity, followed by the methanol and hexane extracts.
Key words: Asteraceae; Phototoxic compounds; Bactericide activity; 12-O-tetradecanoylphorbol-13-acetate (TPA).
Resumen. En las hojas de Eupatorium morifolium Mill. se analizaron los compuestos fototóxicos y la toxicidad del extracto hexánico frente a Bacillus subtilis. Los compuestos fototóxicos se hicieron evidentes mediante TLC. En la placa, las manchas de color azul claro indicaron la presencia de estos compuestos. Esto fue confirmado por el espectro al UV del extracto hexánico y por la actividad antibacteriana frente a Bacillus subtilis. También se analizó la actividad biológica de los extractos de hoja (hexano, acetato de etilo o metanol). Se determinó la actividad anti-inflamatoria empleando la prueba del edema en la oreja de ratón, y la actividad bactericida se midió frente a Bacillus subtilis y Escherichia coli. Las hojas mostraron una fuerte actividad anti-inflamatoria. El extracto de acetato de etilo presentó la mayor actividad bactericida, seguido por el de metanol y hexano.
Palabras clave: Asteraceae; Compuestos fototóxicos; Actividad bactericida; 12-O-tetradecanoilforbol-13-acetato (TPA).
INTRODUCTION
The genus Eupatorium, of the Asteraceae family, comprises nearly 600 species which are distributed in the tropical regions of America (Jones, 1987). In Mexico, it includes several species which have been used in traditional medicine for their pharmacological properties as antibacterial, antifungal, anti-inflammatory and hepatoprotective. Some of these species have also been used as a treatment for cough, tracheitis, diarrhea, headache, muscle pain, etc. (Argueta et al. 1994; Martínez et al., 2001).
The phototoxic compounds isolated from Eupatorium morifolium Mill. are polyacetylenes and thiophenes that require U.V. radiation for expression of their toxicity, which acts as a plant defense.
We have been studying these compounds to observe the frequency of their presence in the Asteraceae family. They could also be used as taxonomic markers at the scale of family or genera.
The anti-inflammatory study was carried out by the edema test on mouse ears, and the antimicrobial properties were assayed against Bacillus subtilis and Escherichia coli. We confirmed the presence of the phototoxic compounds by the U.V. spectrum, the chromatographic profile and the bactericide activity of hexane extract of the plant leaves.
MATERIALS AND METHODS
Plant material. Plants were collected in Los Tuxtlas, Veracruz, and the voucher specimens were deposited at the National Herbarium, Instituto de Biología, UNAM (MEXU).
Preparation of extracts. Dry and ground plant leaves (50 g) were extracted at room temperature with hexane, ethyl acetate or methanol (600 ml each), and the solvent was eliminated at reduced pressure. Dry extracts from hexane (0.13 mg), ethyl acetate (1.56 mg) or methanol (5.76 mg) were used for different biological tests.
Phototoxic compounds. They were determinated by TLC using the leaf hexane extract, at a concentration of 8 mg/ 150 ml. The extract (4 μl) was applied on a silica gel Merck 60 F254 plate, using hexane-ethyl acetate (85:15) as mobile phase. Detection was achieved using UV light (365 nm). The pale blue spots of the compounds appeared after utilizing ceric sulphate.
Photoxic activity. The bioassay was carried out with Bacillus subtilis (ATCC-6051) using the Daniel's method (1965). Paper disks containing the extracts (0.25, 0.5, 1 or 2 mg/ml) were applied on Petri dishes with agar containing a bacterial concentration of 10 6 UFC. Petri dishes were then incubated at 37 ºC during 24h. For comparison, one study was conducted in darkness, and another study under U.V. light (365 nm) exposure.
Antibacterial activity. The test was carried out using the paper disk diffusion method. Petri dishes with agar containing a bacterial concentration of 106 UFC were used. Two strains were evaluated: B. subtilis (ATCC-6633) and Escherichia coli (ATCC-6051) (Cavalieri, 2005). The three extracts, (hexane, ethyl acetate or methanol) were assayed at concentrations of 0.25, 0.5, 1 and 2 mg/ml each. Petri dishes were incubated at 37 ºC during 24h. Controls included the Mueller Hinton agar alone and anhydrous ampicilline (0.02 mg) (Sigma) as a positive control. Each assay was repeated 3 times.
Anti-inflammatory activity. Anti-inflammatory activity of the 3 extracts was carried out by the edema test on mouse ears induced with TPA (12-O-tetradecanoyl-phorbol-13-acetate) (De Young et al., 1989). Three male CDI mice (25 - 30 g) were used for each determination; 10 μL of an ethanolic solution (0.25 mg/ml) of TPA (2.5 μg/ear) were applied to the surface of the right ear on each mouse; the left ear was used as control; 10 min. after the application of TPA, 20 μL of each of the three extracts (hexane, ethyl acetate or methanol; 0.31 mg dissolved in ethanol) were applied topically. Animals were sacrificed after 4 h, and an ear section (7 mm) was weighted. Increases in weight of the right ears with respect to the left ones indicate swelling produced by the TPA application. Indometacine (0.046, 0.085, 0.15, 0.28 mg / ear) was used as the drug reference. The following experiments were carried out:
Edema A: edema induced by TPA alone
Edema B: edema induced by TPA plus extract application
Inhibitory ratio (%) = [(Edema A - Edema B) / Edema A] x 100
RESULTS AND DISCUSSION
Phototoxic compounds. The TLC on leaves showed pale blue spots, characteristic of the phototoxic compounds, with a Rf value of 0.46, when using ceric sulphate and irradiating with U.V. (365 nm). Presence of these compounds was confirmed with (1) the U.V. spectrum of the hexane extract, which showed peaks at 243, 273 and 324 nm, characteristic of polyacetylenes (Table 1), and (2) the bactericidal activity against B. subtilis which indicates its toxicity. For this determination, 4 extract concentrations were used: 0.25, 0.50, 1.0 and 2.0 mg. One study was conducted after irradiating with UV to develop toxicity, while the other study was developed in darkness (control). Values were analyzed by the Student's t-test. Their activity showed significant differences (p< 0.01) only under the two highest concentrations. At these concentrations, 52% and 57% of inhibition of the bacteria was obtained, compared with 42% inhibition without irradiation (Table 2).
Table 1. Characteristic U.V. absorptions of polyacetylenes.
Tabla 1. Absorción de U.V. de poliacetilenos característica.

Table 2. Phototoxic bactericide activity against Bacillus subtilis.
Tabla 2. Actividad bactericida fototóxica contra Bacillus subtilis.

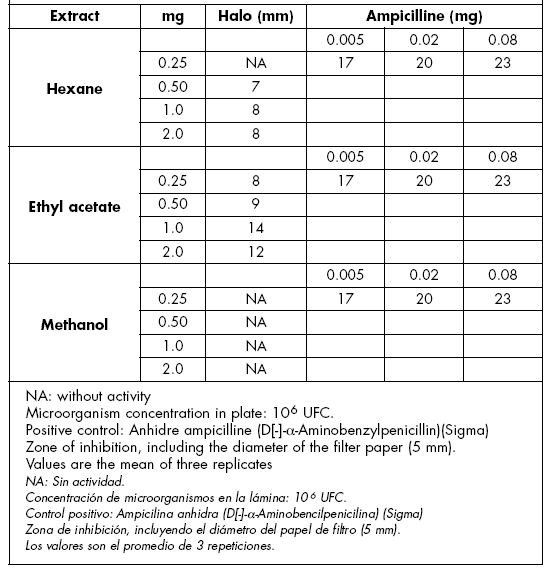
Antibacterial activity. The 3 leaf extracts (hexane, ethyl acetate or methanol) were tested against Bacillus subtilis and Escherichia coli. Four concentrations were used: 0.25, 0.50, 0.1 and 2.0 mg, and the activity was compared with ampicilline (0.02 mg).
The ethyl acetate extract showed no activity with 0.25 and 0.5 mg, and activity was low with 1 and 2 mg in E. coli. The methanolic extract showed a higher activity; only the lowest concentration was inactive. The growth inhibition halos with the methanolic and ethyl acetate extracts on E. coli are indicated in Table 3. The hexane extract was inactive.
Table 3. Antibacterial activity of E. morifolium extracts against Escherichia coli.
Tabla 3. Actividad antibacteriana de extractos de E. morifolium contra Escherichia coli.

The methanolic extract was inactive, and the ethyl acetate extract had a lower activity than the control in B. subtilis (Table 4). There was no activity with 0.25 mg of the hexane extract, and activity was low when using the remaining concentrations. The methanolic extract showed no activity and the ethyl acetate extract was active with 1 and 2 mg in B. subtilis (Table 4). These results validate treatments of diarrhea and other afflictions.
Table 4. Antibacterial activity of E. morifolium extracts against Bacillus subtilis.
Tabla 4. Actividad antibacteriana de extractos de E. morifolium contra Bacillus subtilis.
Anti-inflammatory activity. The 3 extracts showed activity. They were more effective than activity of indomethacin in the TPA (0.25 mg / ml). The major anti-inflammatory activity was found with the ethyl acetate extract (90.89% of inhibition). Percentage inhibition with the other extracts were 66.18% (hexane) and 58.81% (methanol). Values of the three extracts were analyzed by the Student's t-test. Their activity was significant in comparison to that in the controls (p ≤ 0.01) (Table 5). This explains their curative properties for muscle pain, tracheitis, cough and headache.
Table 5. Anti-inflammatory activity of E. morifolium extracts.
Tabla 5. Actividad antiinflamatoria de extractos de E. morifolium.

Eupatorium morifolium is used as a medicinal plant in the South and Southeast of Mexico. Results of this work confirm the appropriate use of this plant species for medical purposes.
REFERENCES
1. Argueta, V.A., A.L.M. Cano & M.E. Rodarte (1994). Atlas: Plantas Medicinales Tradicionales Mexicanas III. Instituto Nacional Indigenísta. México D.F. 1511 p. [ Links ]
2. Cavalieri, S. J. (2005). Drug Resistance, Bacterial-Laboratory Manuals. Departments of Laboratory Medicine and Microbiology. University of Washington, Seattle. 248 p. [ Links ]
3. Daniels, F.J. (1965). A simple microbiological method for demonstrating phototoxic compounds. Journal of Investigative Dermatology 44: 259-263. [ Links ]
4. De Young, L.M., J.B. Kheifets, S.J. Ballaron & J.M. Young (1989). Edema and cell infiltration in the phorbol ester-treated mouse ear are temporaly separate and can be differentially modulated by pharmacological agents. Agents and Action 26: 335-341. [ Links ]
5. Jones, S. (1987). Sistemática Vegetal. Mc Graw-Hill, México. 536 p. [ Links ]
6. Martínez, A.M., O.V. Evangelista, C.M. Mendoza, G.G. Morales, O.G. Toledo & L.A. Wong (2001). Catálogo de Plantas Útiles de la Sierra Norte de Puebla, México. Jardín Botánico, Instituto de Biología. UNAM, México D.F. 303 p. [ Links ]














