Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO  uBio
uBio
Compartir
Phyton (Buenos Aires)
versión On-line ISSN 1851-5657
Phyton (B. Aires) vol.78 no.2 Vicente López jul./dic. 2009
ARTÍCULOS ORIGINALES
Toxicity and accumulation of arsenic in wheat (Triticum aestivum L.) varieties of China
Toxicidad y acumulación de arsénico en variedades de trigo (Triticum aestivum L.) en China
Zhang1 WD, DS Liu2, JC Tian3, FL He1
1 State Key Laboratory of Crop Biology, Agricultural College, Shandong Agricultural University, Taian City, 271018, Shandong Province, P. R. China.
2 Shandong Institute of Environmental Science, Shandong Environmental Protection Bureau, Lishan Street 50, Jinan 250013, Shandong Province, P. R. China.
3 Sub-Center of Cereal Quality Control and Test (Taian), Ministry of Agriculture of People's Republic of China, Taian City, 271018, Shandong Province, P. R. China.
Address Correspondence to: Jichun Tian, e-mail: jctian1954@yahoo.com.cn
Recibido- Received 27.04.2009.
Aceptado-Accepted 01.06.2009.
Abstract. Soil arsenic contamination becomes a potential agricultural and environmental hazard worldwide, and has been a serious problem for safe food production. A field experiment on soil contamination was conducted on four wheat varieties (Jimai, Gaoyou, Weimai and Wennong) in Eastern China, using 50 or 100 mg arsenic/kg soil. Biomass production and yield components were determined and arsenic concentrations were measured in plant tissues. Differential arsenic effects on wheat varieties were determined at maturity. Results showed that addition of arsenic significantly (p<0.05) reduced root, stem and spike dry weight and yield components, which resulted in the decrease of grain yield per plant. Arsenic concentrations in plant tissues increased significantly (p<0.05) with treatments, and its uptake varied considerably among wheat varieties, plant tissues and arsenic treatments. Arsenic concentrations in plant tissues were as follows: roots > stems > leaves and rachises > grains > glumes > awns. In the arsenic treatments, arsenic concentrations in bran were about 2-3 times higher than those in flour. Most of the arsenic contaminated flour exceeded the Chinese tolerance limit. Arsenic contents of grain parts were dependent on variety and treatment level in polluted soils. Weimai and Wennong showed highest amounts of arsenic in flour than the other varieties at 50 or 100 mg/ kg soil treatment, respectively. Weimai possessed significantly lower (p<0.05) amount of arsenic in bran than any other wheat variety. Results showed significant variety differences in arsenic concentration in polluted areas; it is of outstanding importance that wheat with the lowest possible arsenic concentration is used for food or fodder production. The present results provide scientific basis for revising the standards of wastewater discharges.
Key words: Arsenic; Wheat (Triticum aestivum L.); Biomass; Yield components; Arsenic toxicity.
Resumen. La contaminación del suelo por arsénico es un peligro potencial agrícola y ambiental en el mundo, y ha habido serios problemas para una producción de alimentos saludable. Se condujo un experimento a campo sobre la contaminación del suelo con arsénico en cuatro variedades de trigo (Jimai, Gaoyou, Weimai, Wennong) en China Oriental usando 50 ó 100 mg de arsénico/kg de suelo. Se determinaron la producción de biomasa y los componentes del rendimiento, y se midió la concentración de arsénico en tejidos vegetales. Los efectos del arsénico sobre las variedades de trigo se determinaron a la madurez del cultivo. Los resultados mostraron que la adición de arsénico redujo significativamente (p<0,05) el peso seco de raíces, tallos y espigas, y los componentes del rendimiento, lo que determinó una disminución en el rendimiento de grano por planta. La concentración de arsénico en los tejidos vegetales se incrementó significativamente (p<0,05) con los tratamientos, y su absorción varió considerablemente entre las variedades de trigo, tejidos vegetales y tratamientos de arsénico. La concentración de arsénico en los tejidos vegetales fue como sigue: raíces > tallos > hojas y raquis > granos> glumas > aristas. En los tratamientos, las concentraciones de arsénico en salvado fueron 2-3 veces más altas que en la harina. La mayor parte de la harina contaminada por arsénico excedió el límite de tolerancia permitido en China. El contenido de arsénico de las partes del grano dependió de la variedad y el tratamiento en las áreas contaminadas. Weimai o Wennong mostraron mayores cantidades de arsénico en la harina que las otras variedades a 50 ó 100 mg de arsénico/kg de suelo, respectivamente. Weimai tuvo una menor (p<0,05) cantidad de arsénico en el salvado que las otras variedades. Los resultados mostraron diferencias significativas entre las variedades en la concentración de arsénico en lasáreas contaminadas; es de fundamental importancia que se utilice trigo con la menor concentración posible de arsénico en la producción de alimentos y forraje. Los resultados presentados proveen una base científica para revisar los estándares de la descarga de aguas residuales.
Palabras clave: Arsénico; Trigo (Triticum aestivum L.); Biomasa; Componentes del rendimiento; Toxicidad con arsénico.
INTRODUCTION
The adverse effects of toxic chemicals on flora are of paramount importance for making soil ecotoxicological assessments. Arsenic (As) is a metalloid ubiquitously present in soils, normally at trace quantities (Matschullat, 2000). Worldwide, natural soil concentrations are around 5mg/kg, and this varies depending on the origin of the soil (Mandal & Suzuki, 2002). The arsenic content of agricultural soils (Andersson, 1992; Notter, 1993) and of natural waters (Hallberg, 1991) has increased during recent decades. Its levels were highly increased by industrial activities such as metal smelting, coal combustion and glass manufacture (Smith et al., 1998), and the present use of As compounds as fertilizers, pesticides, desiccants (Mahimairaja et al., 2005) and growth promoters for poultry and pigs (Christen, 2001).
High levels of arsenic in soils have been phytotoxic in plants: decreases in plant growth and fruit yields (Carbonell- Barrachina et al., 1995); discolored and stunted roots; withered and yellow leaves (Machlis, 1941); reductions in chlorophyll and protein contents, and in photosynthetic capacity (Marin et al., 1993).
Wheat is one of the most important cereals in the world. Inappropriate arsenic concentrations can be harmful to wheat seedling at early developmental stages. Seed germination and seedling growth were stimulated at low and inhibited at high As concentrations (5-20 mg/kg soil). Physiological activities of wheat seedlings were also changed under As stress (Li et al., 2007). Seed germination, biomass, root length and shoot height decreased, and As accumulation increased on early seedlings of six wheat varieties as concentrations increased (Liu & Zhang, 2007a). Further experiments showed that seedlings uptake, which followed Michaeli-Menten kinetics, increased with increasing As concentrations in the test solution. Average total amylolytic, α-amylase and β-amylase activities seemed to decrease with increasing As in wheat seedlings of different varieties (Liu et al., 2007b).
In China, arsenic is the first rate contaminant, and many wheat-planted areas have been polluted by human or industrious arsenate-containing wastes (Liu et al., 2002; Yang et al., 2002). Arsenic promotes initiation of cancer cells in a broad array of tissues (Kayajanian, 2000). Threats that arsenic imposes to human and animal health is aggravated by its long-term existence in the environment (Hall, 2002). Arsenic may accumulate in agricultural soils and plants, and it is a cumulative poison that affects all body systems (Berman, 1980; González et al., 2001). However, most works on arsenic toxicity in wheat have been short-term studies. Our working hypothesis was that arsenic reduces plant biomass and yield components in chronically-exposed wheat plants, and that high As concentrations affect arsenic distribution within the plant, especially in grains and flour. The objectives of this study were to determine the (1) arsenic effects on plant part biomass and yield components, and (2) arsenic content of different plant parts at wheat maturity. These results will be beneficial to predict and reduce the risk of arsenic entrance into the food chain.
MATERIALS AND METHODS
Plant material and soil preparation. Four varieties of winter wheat (Triticum aestivum L.: Jimai, Gaoyou, Weimai and Wennong) were supplied for the study by the Sub-Center of Cereal Quality Control and Test (Taian), Ministry of Agriculture of People's Republic of China (Taian, Shandong Province). Soil was a coarse-silty loam, collected from a local farm at 0-15 cm depth. It was crushed, mixed thoroughly and sieved through a 2mm mesh. A composed sample from this soil was collected for physico-chemical analysis. Some soil properties are presented in Table 1. A solution of As (Na3AsO4.12H2O) was mixed thoroughly with the soil at a rate of 0 (control), 50 and 100 mg As/kg soil. The soil was then air dried, passed through a 2 mm sieve again, and aged for 1 week.
Table 1. Physicochemical properties of the silty loam soil at the start of the study
Tabla 1. Propiedades fisicoquímicas del suelo arcilloso-limoso al comienzo del estudio.

Wheat cultivation. Seeds were surfaced-sterilized with hypochlorite for 3 min and washed with tap water. They were then submerged in distilled water, and cultured in an incubator for 24 h set at 37°C. After the radicle appeared, seeds were planted in plastic cells (5×5×5 cm) containing moist vermiculite. Fourteen-day-old seedlings with two leaves were carefully transplanted to concrete pools. Pools were under a rainproof shelter in the farm. They were 3 (wide) x 3 (long) x 3 (deep) meters. Pools were filled with soil from local farm. This soil was taken from 50 cm below the surface soil. Pools were filled up to 2 meters depth. The As contaminated soil filled up the remaining 1 m of the pools. Urea, triple super phosphate (TSP) and muriate of potash (MP) were applied at the rate of 10, 40 and 20 kg per hectare for nitrogen, phosphorous and potassium, respectively, in the first 30 cm of the contaminated soil. Fertilizers were incorporated into the soil by hand, and applied before transplantation. Ten kilograms of urea per hectare were applied at the tillering and panicle initiation stages as a field protocol. The experiment was laid out as a completely randomized block design with four replicates. There was one pool for each level of arsenic, so that 12 pools were used in this study. Two hundred seedlings were planted per square meter at equal spacing. Pools were watered periodically by dripping to approximate field capacity. Representative mature wheat plants were randomly sampled in each replicate at the end of the wheat growing season. Each replicate pool consisted of 30~40 plants.
Plant measurements and arsenic determination. Mature plants were harvested and separated into roots, stems (including straw and leaves) and spikes. Roots were washed completely using distilled water. Dry weight (biomass) of plant parts was recorded after drying them in an oven for 60 h at 80°C, Yield components were investigated according to the"Rules of registration for crop variety (wheat)" (AMPRC, 2006).
For arsenic determination, spikes were partitioned into awn, grain, glume and rachis. Wheat grains were milled into flour using a Bühler Laboratory Mill (Bühler-Miag, Uzwil, Switzerland). Plant (straw, roots, and leaves) and spike parts (awns, glumes, grains and rachises) were first washed thoroughly with tap water and then with de-ionized water, and dried at 60-70°C for 72 h. The bigger dried materials, such as straws, were cut into pieces with stainless steel scissors. All samples were ground into powder using a mortar. These samples were ashed in a muffle furnace at 550°C for 10 h, and 0.2 g of each sample was dissolved in a 1:1 (v/v) hydrochloric acid/NANOpure water solution. Arsenic concentration was determined in the extraction solution by ICP-AES (IRIS/AP, Inductively coupled plasma atomic emission spectroscope, TJA, USA). This was conducted in the Analysis and Test Center (Jinan, Shandong Province). All values reported in this study are the mean of four replicates; at least three independent measurements were made on each sample.
Statistical analysis. All experimental data were subjected to analysis of variance (ANOVA) using SPSS, version 11.0 (SPSS, 2001). Mean values were compared by the LSD method or Duncan's multiple range test at the 5% level.
RESULTS
Plant biomass of roots, stems (straw and leaves) and spikes in four wheat varieties under two arsenic treatments. Addition of arsenic to the soil at concentrations of 50 mg/kg or 100 mg/kg significantly (p<0.05) decreased the biomass of roots, stems and spikes (Table 2). The greatest biomass reduction was found in roots, and the lowest in spikes. Reductions in biomass differed among varieties within a same plant part.
Table 2. Biomass (gr/plant) of roots, stems (straw and leaves) and spikes in four wheat varieties exposed to two arsenic treatments.
Tabla 2. Biomasa (gr/planta) de raíces, tallos (residuos de cosecha y hojas) y espigas en cuatro variedades de trigo expuestos a dos niveles de arsénico.
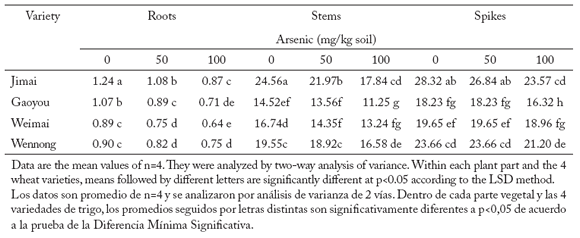
Dry weight of roots was significantly (p<0.050) decreased in the four varieties at the 50 mg/kg arsenate treatment. Stem biomasses of Gaoyou and Wennong, and spike biomasses of Jimai, Weimai and Wennong were all reduced by arsenic addition, but they remained below the limits of significance.
Dry weight of shoots, stems and spikes were all significantly (p<0.050) decreased in all four varieties at 100 mg As/kg soil. However, the reduction differed among varieties and plant parts. For example, root, stem and spike biomasses were reduced by 30.0, 27.5 and 16.9% in Jimai, respectively, and by 16.7, 15.3 and 13.8% in Wennong, respectively, compared with values in the control. Root biomasses of Weimai and Wennong were not significantly different to controls, but they were significantly different (p<0.010) to the 100 mg/kg soil arsenic treatment.
Biomasses in wheat roots, stems and spikes decreased (p<0.001) with increasing As soil concentrations, and there were significant differences (p<0.001) among varieties.
Yield components in four wheat varieties under two arsenic treatments. Arsenic at 50 mg/kg or 100 mg/kg soil significantly reduced (p<0.050) the number of spikes per plant and the thousand-kernel weight (Table 3). However, it showed little significant effect on the number of spikelets or grains per ear. In the 50 mg/kg treatment, the greatest reduction in spike number per plant and grain weight was 40.9% and 15.5% in Gaoyou and Jimai, respectively, when compared with values in the control. In the 100 mg/kg treatment, the number of spikelets and grains per ear were also lower as a result of arsenic addition. However, such decreases seldom reached significant levels.
Table 3. Yield components in four wheat varieties exposed to two arsenic treatments.
Tabla 3. Componentes de rendimiento en cuatro variedades de trigo expuestos a dos niveles de arsénico.
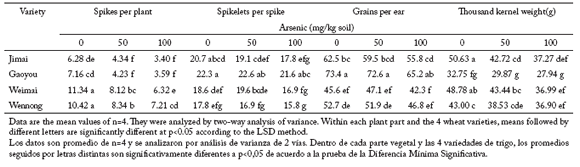
All four yield components showed significant reductions as As increased in the soil (p<0.05), and there were also significant differences (p<0.001) among varieties. Arsenic concentration in different plant parts in plants exposed to two arsenic treatments. Arsenic concentration varied considerably between wheat varieties, plant parts and arsenic treatments (Table 4). Generally, arsenic concentration in roots was about 10 times higher than that in shoots, which in turn contained nearly as much As as that in leaves under the two arsenic treatments.
Table 4. Arsenic accumulation (mg/kg dry weight) in root, straw and leaf of four wheat varieties exposed to two arsenic treatments.
Tabla 4. Acumulación de arsénico (mg/kg peso seco) en raíces, residuos de cosecha y hojas en cuatro variedades de trigo expuestos a dos niveles de arsénico.
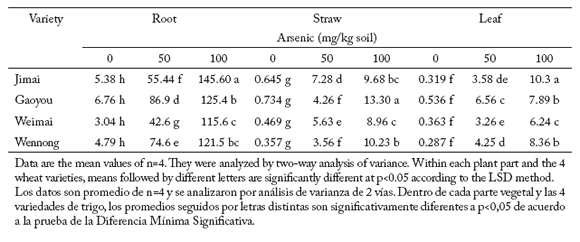
A little amount of arsenic was found in roots, straw or leaves of all four wheat varieties growing in control soils. In the 50 mg/kg treatment, As concentration and distribution in various plant parts varied among varieties. Gaoyou and Weimai had the highest and lowest root arsenic concentrations, respectively. They differed significantly with respect to the other two varieties. Concentrations of As were the highest in straw for Jimai and in leaves for Gaoyou. Wennong and Weimai had significantly lower As concentrations in straw and leaves, respectively, than the other varieties.
In the 100 mg As/kg soil treatment, arsenic concentrations in roots, straw and leaves significantly increased (p<0.010) with respect to the other two treatments (Table 4). Arsenic concentrations in roots, straw or leaves did not follow the same pattern, neither in all study varieties nor in the different As treatments. For example, in straw the highest arsenic concentration in the 50 mg/kg treatment (a) was found in Jimai, where arsenic concentrations reached values of 7.28 mg As/kg dry weight, and (b) 100 mg/kg treatment was found in Gaoyou, where arsenic concentrations reached 13.3 mg As/kg dry weight. In the 100 mg/kg treatment, the lowest arsenic concentrations in both straw and leaves were found in Weimai, but in the 50 mg/kg treatment, these concentrations were found in Wennong instead.
Arsenic concentrations in the different plant parts in the four wheat varieties increased (p<0.001) with increasing As concentrations in the soil. However, there were not significant differences (p< 0.05) among varieties.
Arsenic concentration in different spike parts on plants exposed to two arsenic treatments. Very little arsenic amount was detected in all spike parts in the four wheat varieties growing in control soil. Similar to the arsenic accumulation trends observed in wheat plant parts, arsenic concentrations increased with external arsenic levels in each part of spikes (Table 5). Arsenic concentrations in various spike parts on both arsenic addition levels at maturity were in the order: rachis> grain > glume > awn.
Table 5. Arsenic accumulation (mg/kg dry weight) in rachis, grain, glume and awn of four wheat varieties exposed to two arsenic treatments.
Tabla 5. Acumulación de arsénico (mg/kg peso seco) en raquis, grano, gluma y arista en cuatro variedades de trigo expuestos a dos niveles de arsénico.
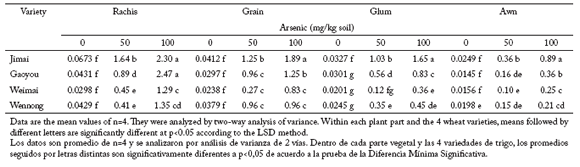
Arsenic concentrations in the spike parts were different among the four varieties when As was added in the plant soil. Jimai had significantly higher arsenic concentrations than Weimai in all four spike parts (Table 5). Arsenic concentrations in the different spike parts increased (p<0.05) with increasing As concentration in the soil in the four wheat varieties. Except in glumes, there were no significant differences (p<0.05) in arsenic concentration among varieties.
Arsenic concentration in flour and bran on plants exposed to two arsenic treatments. Arsenic concentrations were about 2-3 times higher in bran than in flour in the arsenic treatments (Table 6).
Table 6. Arsenic accumulation (mg/kg dry weight) in flour and bran of four wheat varieties exposed to two arsenic treatments.
Tabla 6. Acumulación de arsénico (mg/kg peso seco) en harina y salvado en cuatro variedades de trigo expuestos a dos niveles de arsénico.

The overall As concentration interval in flour from the four varieties of wheat was 0.29-0.78 mg/kg dry weight in all wheat varieties at the 50 mg/kg soil treatment. Jimai had the highest As concentration, while it was half of it in Weimai. In the 100 mg/kg treatment, however, the overall As concentration interval was 0.59-1.49 mg/kg dry weight flour in all four wheat varieties. Arsenic concentrations in Jimai and Gaoyou were significantly higher than those in Weimai and Wennong.
The overall concentration interval was 0.59 (Weimai) -1.49 (Gaoyou) mg/kg dry weight bran in the 50 mg/kg treatment, and 1.98 (Weimai) -3.04 ( Jimai) mg/kg dry weight bran in the 100 mg/kg treatment, in the four varieties of wheat (Table 6). The arsenic concentration in bran was about 2-3 times higher than that in flour, depending on the variety.
The ratio of bran/flour changed with both the variety and the arsenic treatment. For example, the ratio of bran/ flour was higher in Jimai than in Gaoyou, in the 0 and 50 mg/kg treatments. However, in the 100 mg/kg treatment, the situation was just opposite. Among the four varieties in this study, Wennong showed the largest relative difference between the bran and flour arsenic concentrations; the average bran/flour concentration ratio was 3.56. The smallest average bran/flour concentration ratio was found in Gaoyou (2.14).
DISCUSSION
The two As treatments used in this study represent either moderate or serious contamination levels in China. A Copper Smeltery located in Changle County in the Hubei Province in China polluted the surrounding villages to about 40-70 mg As/kg soil; it was considered that the area was polluted by As to above average levels: 50 mg As/kg soil (Xie et al., 2005). Heavily As-contaminated soils can reach 238.4 and 302.2 mg/kg soil close to ore deposits in Xingren county, Guizhou province, and Binzhou county, Hunan province in China, respectively (Zhong & Tang., 2006). The range of As contamination in soils of central India (i.e., Ambagarh Chauki, Chhattisgarh) is 9-105 mg/kg soil (Patel et al., 2005). Arsenic concentrations in agricultural soils of As-affected areas in Bangladesh range from 20 to 90 mg/kg soil (Ullah, 1998). Our previous laboratory experiments also showed that As levels of 100 mg/kg soil reduced wheat seed germination rates and inhibited seedling growth. However, wheat seedlings transplanted into high As-polluted soils reached the mature phenological period (data not shown).
Although varieties tested in this study differed greatly in their response to As addition in soil, they all followed the same pattern: when As was added, a negative response by plant part biomass and yield components, and a positive response by plant parts at wheat maturity were obtained.
Toxic symptoms included wilting and necrosis of leaf margins, and stunt root and stems. Increased As concentrations in the soil led to more pronounced reductions in root biomass than in any other plant part. Biomass inhibition followed the order: roots > stems > spikes. Large numbers of studies have indicated that excessive As did harm plant seedling biomass and growth, and that this harm could be attributed to a series of physiological activity alterations (Han et al., 2002; Liu& Zhang, 2007; Liu et al., 2007). It is not surprising that biomass decreases were obtained in mature plants. Seth et al. (2007) reported fresh weight reductions in giant duckweed after long-term exposures to As. Biomass reductions were lower on spikes than roots or stems, and this might result in lower As accumulation in spikes.
Alteration of some wheat yield components by toxic elements has also been reported with cadmium treatments (Zhang et al., 2002). Results obtained by these authors were similar to ours. Reduction in biomass and yield components might be due to direct toxic effects of As on biochemistry and physiological processes in the wheat plant. Our experiment showed that As had an adverse effect on spike number per plant, which resulted from less tillering and weak tillers. Additionally, all four wheat varieties showed late tiller initiation. Grain number per ear and thousand-kernel weight were significantly reduced under As treatments. Damaging effects of As on plants might also be due to effects on phosphate uptake and utilization. Phosphate level is known to control plant growth and development, and arsenate is a phosphate analogue. Increasing arsenate in soil results in enhanced competition between phosphate and arsenate for sorption sites. This result in a decrease in the amount of phosphate sorbed (Meharg & Hartley-Whitaker, 2002). Furthermore, after entering a plant, arsenate may replace phosphate in various phosphorolysis reactions, thus interfering with phosphate metabolism and causing plant toxicity (Dixon, 1997).
Irrespective of the As treatment, roots contained higher concentrations of As than straw and leaves, and As concentrations in straw were slightly higher than those in leaves. Highest As accumulation in roots than in any other plant part was also reported in rice (Marin et al., 1992, 1993), maize, English ryegrass, rape and sunflower (Gulz et al., 2005). The present study showed that the ranges of As concentrations in straw and leaves were 3.56-7.28 and 3.26-6.56 mg/kg at 50 mg/kg soil treatment, respectively, and 8.96-11.30 and 6.24-10.30 mg/kg at 100 mg/kg soil, respectively. The maximum As concentration allowed in fodder plants by the Chinese law is 4 mg/kg on a dry weight basis. Wheat varieties investigated in this study accumulated relatively high As concentrations in their edible parts; this indicates that wheat might represent a risk for animal and human health when this crop grows on As polluted soils.
The distribution of poisonous elements in spike parts was seldom documented in the literature. Chen et al. (2007) reported that cadmium (Cd) concentrations in each part of detached spikes increased with external Cd levels in developing barley grains; final Cd concentrations in various organs were in the order: awn > grain > rachis > glume. They also reported that awns, rachises and glumes may be involved in Cd transport into developing grains. The present study demonstrated
that As concentrations increased in each part of spikes at increasing As levels. Arsenic concentration order, however, was rachis > grain > glume > awn in all four wheat varieties. More detailed information is necessary about the physiological processes responsible for As translocation and accumulation in grains to understand the influences of other spike parts on As grain concentration.
Flour is the main edible portion in wheat grains. Arsenic content in flour is critical for human health. Our results showed that As concentrations in bran were much higher than those in flour. He et al. (2001) reported that comparing various nourish components in wheat, the percentage of As in protein was the highest (74% of total As). This indicated that As in wheat seed mainly exists associated to protein parts. Bran is mainly constituted by the embryo and capsule of wheat seeds, and is richer in protein than flour processed from the endosperm. Our study showed that the concentrations in flour ranged from 0.29-0.78 mg/kg and 0.59-1.49 mg/kg at 50mg/kg soil and 100mg/kg soil, respectively, in the tested wheat varieties. Most of the As contaminated flour exceeded the Chinese tolerance limit: 0.5 mg/kg flour (Li et al., 2006). Wheat bran and flour have been mainly used as fodder and staple food for animals and humans, respectively, in China. It is necessary to monitor the As content in wheat yielded from As polluted areas; it is of major importance that wheat with the lowest As content is provided to the peasants. Rahman et al. (2007) and Cheng et al. (2006) also demonstrated significant variety differences in As content on rice grain at higher As concentrations in the soil; low As rice varieties were recommended for planting in Bangladesh and China. Arsenic intake by humans from rice and cattle could be potentially important; there exists possible food chain pathways through which As may enter into the human body (Rahman et al., 2008). Except Wennong which concentrated the lowest amounts of As in flour at 100 mg/kg soil treatment, Weimai showed significantly lower As amounts in bran and flour compared to the other wheat varieties. There was a dependence of As content in grain parts with variety and treatment level. This implies that it is necessary and possible to select or breed wheat varieties with low As content in wheat grains for peasants in polluted areas.
ACKNOWLEDGEMENTS
We thank our co-workers Cailing Sun, Yongxiang Zhang, and Xiansheng Chen for their assistance to this research. This research is a part of the Program for Growing Superior Grain Cultivators in Shandong Province (2005), and the National Technology Supporting Program of China (2006BAD01A02-15). The Program of Water Monitoring and Protection in Shandong Province also provided financial support for this research.
REFERENCES
1. AMPRC (Agricultural Ministry of People's Republic of China) (2006). Rules of registration for crop variety (wheat). Agricultural Standards of People's Republic of China. NY/T 967-2006. [ Links ]
2. Andersson, A. (1992). Trace Elements in Agricultural Soils: Fluxes, Balances and Background Values. Swedish Environment Protection Agency Report 4077. Naturvas rdsverket, Solna. [ Links ]
3. Berman, E. (1980). Toxic Metals and Their Analysis, Ch. 4. Heyden and Son Ltd., London, UK. [ Links ]
4. Carbonell-Barrachina, A., F. Burlo-Carbonell & J. Mataix-Beneyto (1995). Arsenic uptake, distribution, and accumulation in tomato plants: effects of arsenate on plant growth and yield. Journal of Plant Nutrition 18: 1237-1250. [ Links ]
5. Chen, F., F. Wu, J. Dong, E. Vincze, G. Zhang, F. Wang, Y. Huang & K. Wei (2007). Cadmium translocation and accumulation in developing barley grains. Planta 227: 223-232. [ Links ]
6. Cheng, W., G. Zhang, H. Yao, W. Wu & M. Xu (2006). Genotypic and environmental variation in cadmium, chromium, arsenic, nickel, and lead concentrations in rice grains. Journal of Zhejiang University, Science B 7: 565-571. [ Links ]
7. Christen, K. (2001). Chickens, manure, and arsenic. Environmental Science and Technology 5: 184-185. [ Links ]
8. Dixon, H.B.F. (1997). The biochemical action of arsenic acids especially as phosphate analogues. In: Sykes, A.G. (ed). pp. 191-228. Advances in Inorganic Chemistry. Academic Press, Inc., San Diego, CA. [ Links ]
9. González, M.M., M. Gallego & M Valcárcel (2001). Determination of arsenic in wheat flour by electrothermal atomic absorption spectrometry using a continuous precipitation-dissolution flow system. Talanta 55: 135-142. [ Links ]
10. Gulz, P.A., S. Gupta & R. Schulin (2005). Arsenic accumulation of common plants from contaminated soils. Plant Soil 272: 337-347. [ Links ]
11. Hall, A. (2002). Chronic arsenic poisoning. Toxicological Letters 128: 69-72. [ Links ]
12. Hallberg, R.O. (1991). Environmental implications of metal distribution in Baltic Sea sediments. Ambio 20: 309-316. [ Links ]
13. Han, Z.X, G.Y. Feng & W.Z. Lu (2002). Study on effects of As(III) in environment on wheat sprout and the original researcher of prevention and treatment of arsenic toxicant. Acta Botanica Boreali-Occidentalia Sinica 18: 123-128. [ Links ]
14. He, M., J. Yang, P. Zhang & W. Song (2001). The main existing forms of arsenic in polluted wheat seed and its stability. Journal of Beijing Normal University 37: 126-130. [ Links ]
15. Kayajanian, G..M. (2000). Arsenic, dioxin, and the promotional step in cancer creation. Ecotoxicology and Environmental Safety 45: 195-197. [ Links ]
16. Li, C., S. Feng, Y. Shao, L. Jiang, X. Lu & X. Hou (2007). Effects of arsenic on seed germination and physiological activities of wheat seedlings. Journal of Environmental Sciences 19: 725-732. [ Links ]
17. Li, X.W., J.Q. Gao, Y.F. Wang & J.S. Chen (2006). Chinese total dietary study in 2000-the dietary arsenic intakes. Journal of Hygiene Reserches 35: 63-67. [ Links ]
18. Liu, J., B. Zheng, H.V. Aposhian, Y. Zhou, M.L. Chen, A. Zhang& M.P Waalkes (2002). Chronic arsenic poisoning from burning high-arsenic-containing coal in Guizhou, China. Environmental Health Perspectives 110: 119-122. [ Links ]
19. Liu, X. & S. Zhang (2007). Intraspecific differences in effects of cocontamination of cadmium and arsenate on early seedling growth and metal uptake by wheat. Journal of Environmental Sciences 19: 1221-1227. [ Links ]
20. Liu, X., S. Zhang, X. Shan & P. Christie (2007). Combined toxicity of cadmium and arsenate to wheat seedlings and plant uptake and antioxidative enzyme responses to cadmium and arsenate cocontamination. Ecotoxicology and Environmental Safety 68: 305-313. [ Links ]
21. Machlis, L. (1941). Accumulation of arsenic in shoots of sudangrass and bushbean. Plant Physiology 16: 521-543. [ Links ]
22. Mahimairaja, S., N.S. Bolan, D.C. Adriano & B. Robinson. (2005). Arsenic contamination and its risk management in complex environmental settings. Advances in Agronomy 86: 1-82. [ Links ]
23. Mandal, B.K. & K.T. Suzuki (2002). Arsenic around the world: a review. Talanta 58: 201-235. [ Links ]
24. Marin, A.R., P.H. Masscheleyn & W.H. Patrick (1992). The influence of chemical form and concentration of arsenic on rice growth and tissue arsenic concentration. Plant Soil 139: 175-183. [ Links ]
25. Marin, A.R., S.R. Pezashki, P.H. Masschelen & H.S. Choi (1993). Effect of dimethylarsenic acid (DMAA) on growth, tissue arsenic, and photosynthesis of rice plants. Journal of Plant Nutrition 16: 865-880. [ Links ]
26. Matschullat, J. (2000). Arsenic in the geosphere: a review. Science of the Total Environment 249: 297-312. [ Links ]
27. Meharg, A.A. & J. Hartley-Whitaker (2002). Arsenic uptake and metabolism in arsenic resistant and non-resistant plant species. New Phytologist 154: 29-43. [ Links ]
28. Notter, M. Ed. (1993). Metals and the environment: The Environment in Sweden-Status and Trends (MIST), Swedish Environmental Protection Agency Report 4245. A.B. Fälths Tryckeri, Värnamo. [ Links ]
29. Patel, K.S., K. Shrivas, R. Brandt, N.J. Akubowski, W. Corns & P. Hoffmann (2005). Arsenic contamination in water, soil, sediment and rice of central India. Environmental Geochemistry and Health 27: 131-145. [ Links ]
30. Rahman, A.M., H. Hasegawa, M.M. Rahman, M.A. Mazid-Miah & A. Tasmin (2008). Arsenic accumulation in rice (Oryza sativa L.): human exposure through food chain. Ecotoxicology and Environmental Safety 69: 317-324. [ Links ]
31. Rahman, M.A., H. Hasegawa, M.M. Rahman, M.N. Islam, M.A.M. Miah & A. Tasmin (2007). Arsenic Accumulation in Rice (Oryza sativa L.) Varieties of Bangladesh: A Glass House Study. Water Air and Soil Pollution 185: 53-61. [ Links ]
32. Seth, C.S., P.K. Chaturvedi & V. Misra (2007). Toxic Effect of Arsenate and Cadmium Alone and in Combination on Giant Duckweed (Spirodela polyrrhiza L.) in Response to its Accumulation. Environmental Toxicology 22: 539-549. [ Links ]
33. Smith, E., R. Naidu & A.M. Alston (1998). Arsenic in the soil environment: a review. Advances in Agronomy 64: 149-195. [ Links ]
34. SPSS. (Statistical Product and Service Solutions) (2001). SPSS 11.0 for Windows Update. SPSS Inc., Chicago. IL. [ Links ]
35. Ullah, S.M. (1998). In: International Conference on Arsenic Pollution of Groundwater in Bangladesh: Causes, Effects and Remedies. Dhaka Community Hospital, Dhaka, Bangladesh, p. 133. [ Links ]
36. Xie, H., X. Liao, T. Chen & J. Lin (2005). Arsenic in plants of farmland and its healthy risk: a case study in an As-contaminated site in Dengjiatang, Chenzhou City, Hunan province. Geographical Research 24: 151-159. [ Links ]
37. Yang, L., W. Peterson, P.J.W. Peter, W. Wang, S. Hou & J. Tan (2002). The relationship between exposure to arsenic concentrations in drinking water and the development of skin lesions in farmers from Inner Mongolia, China. Environmental Geochemistry and Health 24: 293-303. [ Links ]
38. Zhang, G.P., M. Fukami & H. Sekimoto (2002). Influence of cadmium on mineral concentrations and yield components in wheat genotypes differing in Cd tolerance at seedling stage. Field Crops Research 77: 93-98. [ Links ]
39. Zhong, G.M. & Z.Z. Tang (2006). Environmental Cadmium, Lead and Arsenic Pollution and Health Impact-Research Progress in China. Journal of Environmental Health 23: 562-565. [ Links ]














