Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO  uBio
uBio
Compartilhar
Phyton (Buenos Aires)
versão On-line ISSN 1851-5657
Phyton (B. Aires) vol.85 no.1 Vicente López jun. 2016
ARTÍCULOS ORIGINALES
Total aboveground plant biomass is more strongly affected by climate than species diversity on a grassland in Liaoning, China
La biomasa aérea total es más afectada por el clima que por la diversidad de especies en un pastizal natural de Liaoning, China.
Zhou C1,2, C Busso3, J Liu1, YG Yang1, Y Sun1, YZ Fang1, QQ Zhang1, YB Zhou1,4, YN Wang1,4, Z Zhang4,5, ZW Wang6, YF Yang2
1 School of Life Sciences, Liaoning University, Shenyang, 110036, China.
2 Key Laboratory for Vegetation Ecology, Ministry of Education, Institute of Grassland Science, Northeast Normal University, Changchun, 130024, China.
3 Departamento de Agronomía-CERZOS (CONICET), Universidad Nacional del Sur, 8000 Bahía Blanca, Argentina.
4 College of Life Science and Bioengineering, Shenyang University, Shenyang, 110044, China.
5 Hulunber Grassland Ecosystem Research Station of Chinese Academy of Agricultural Sciences.
6 State Key Laboratory of Forest and Soil Ecology, Institute of Applied Ecology, Chinese Academy of Science, Shenyang, 110016, China.
Address correspondence to: Zhuo Zhang, College of Life Science and Bioengineering, Shenyang University, Shenyang 110044, P.R. China. E-mail: zhangzhuonet@163.com
Received 23.V.2016.
Accepted 27.V.2016.
Abstract. The objective of this study was to analyze the relationships between total aboveground plant biomass versus altitude, latitude, longitude, mean annual temperature, mean annual precipitation, and species diversity. Simple linear regression analysis was used to study these relationships. Results showed that altitude was significantly, positively (R2 = 0.038, P<0.01) related with total plant aboveground biomass. Meanwhile, when longitude (R2 = 0.124, P<0.001) and latitude (R2 = 0.221, P<0.001) increased, total aboveground biomass decreased. The relationship between biomass and mean annual precipitation was significantly, positively linear (R2 = 0.149, P<0.001). Mean annual temperature was signifcantly, negatively correlated with biomass (R2 = 0.145, P<0.001). The relationship between species diversity and biomass was significant, but relatively weaker (i.e., R2<0.063, P<0.05) compared with those for the climatic variables. This is, species diversity explained less than 7% of the total variability in total aboveground plant biomass. This result agrees with the idiosyncratic response hypothesis which suggests that ecosystem function changes when species diversity changes, but the magnitude and direction of these changes are unpredictable because the roles of individual plant species are complex and varied.
Keywords: Productivity; Species diversity; Climate; Grasslands.
Resumen. El objetivo de este estudio fue analizar las relaciones entre la biomasa vegetal aérea total versus altitud, latitud, longitud, temperatura promedio anual, precipitación promedio anual, y diversidad de especies. Se utilizó regresión lineal simple para estudiar estas relaciones. Los resultados mostraron que la altitud se relacionó positivamente, significativamente (R2 = 0,038, P<0,01) con la biomasa aérea total. Al mismo tiempo, cuando la longitud (R2 = 0,124, P<0,001) y la latitud (R2 = 0,221, P<0,001) se incrementaron, la biomasa aérea total disminuyó. La biomasa aérea total y la precipitación promedio anual estuvieron positivamente, significativamente (R2 = 0,149, P<0,001) relacionadas. La temperatura promedio anual estuvo negativamente, significativamente (R2 = 0,145, P<0,001) relacionada con la biomasa aérea total. Sin embargo, y si bien la diversidad de especies y la biomasa aérea total se relacionaron negativamente, significativamente (i.e., R2 <0,063, P<0,05), dicha relación fue comparativamente más débil que aquella obtenida para las variables climáticas. Es decir, la diversidad de especies explicó menos de 7% de la variación total en la biomasa aérea total. Este resultado concuerda con la hipótesis de respuesta idiosincrática la cual sugiere que la función del ecosistema cambia cuando la diversidad de especies cambia, pero la magnitud y dirección de estos cambios son impredecibles debido a que los roles individuales de las especies son complejos y variados.
Palabras clave: Productividad; Diversidad de especies; Clima; Pastizales naturales.
INTRODUCTION
Diversity-productivity relationships play an important role in research related to sustainability of ecosystems (Tilman et al., 1996). Many studies have shown that environmental factors play an important role in determining productivity (i.e., change in biomass during a time period/time period) and species diversity on plant communities (Mittelbach et al., 2001; Tilman et al., 2001; Maestre et al., 2006). Species diversity at a local scale may afect both production and stability of any given ecosystem (Weigelt et al., 2008). Many researches have hypothesized that the loss of species can influence vital ecological processes and functions, such as the loss of trophic levels and resilience (Begossi, 2000; Hooper et al., 2005; Fischer et al., 2006).The underlying mechanisms of this relationship have been greatly debated including measures (Shannon-Wiener, Simpson, evenness), study scales and types (genetic, species) of diversity (Wardle & Zackrisson, 2005; Costanza et al., 2007). Therefore, diversity-total aboveground plant biomass relationships are a current important issue for studying in ecology.
Much research has studied fve productivity-diversity patterns: (1) no relationship (Lawton, 1994), (2) hump-shaped (Guo & Berry, 1998), (3) U-shaped (Gillman & Wright, 2006), (4) positive (Bai et al., 2007), or (5) negative (Oksanen, 1996). It is then of interest to know the pattern between total aboveground plant biomass and species diversity. A meta-analysis by Mittelbach et al. (2001) showed that unimodal shapes are found mainly at local to landscape scales, whereas the pattern of humped-back usually happens at finer scales. Many ecologists have found that productivity-diversity relationships depend on the extent of the study area. Thus, different relationship patterns can be found at diferent spatial scales. Different area extents are different from the effects of historical and ecological processes (Fukami & Morin, 2003; Ma et al., 2010), productivity gradients (Virtanen et al., 2013) and sampling grains (Hector et al., 2010). However, surveys on the influences of plant diversity on its biomass or productivity have not been consistent possibly because of the intricacy in ecological features among investigations (e.g., Wu, 1980).
In nature, the diversity-productivity relationships have changed with spatial scales. At the within-community scale, the modal is dominated by unimodal relationships (Wu et al., 2014). No significant relationships have always occurred at the across-community scale (Postor et al., 1996). At the continental-to-global scale, the relationship between productivity and species diversity has often been positive (Waide et al., 1999). References about negative patterns between these variables are scarce. Even more rare are reports on the relationship between total aboveground plant biomass and species diversity.
There are typical meadow-, desert-, mountain-, and alpine-steppes, and upland meadows in China (Ma et al., 2010). Upland meadows are a special grassland type located at arid and semi-arid areas, below 150 m elevation. Different regional species composition and different dominant environmental factors may result in different relationships between total aboveground plant biomass and species diversity. In recent years, the meadow resources have been damaged by rodents, pests, and overgrazing because of a lack of any scientific control (Smith, 1985). Moreover, the most likely explanation for the potential upland meadow degradation and changes in species diversity on the plant communities has been the result of the combined impact of global warming and human activities (Rodwell et al., 1992). Species diversity and aboveground biomass of upland meadows are influenced by a range of ecological factors, including altitude, longitude, latitude, mean annual precipitation and mean annual temperature (Waide et al., 1999; Schmid, 2002a; Schmid et al., 2002b). However, and especially in the upland meadow, the relationship between species diversity and aboveground biomass is not clear.
This research was set up in an upland meadow area. The relationships between total aboveground biomass and latitude, longitude, altitude, mean annual precipitation and mean annual temperature were analyzed. Also, we analyzed the relationship between species diversity and total aboveground biomass. This study provides a major reference for an eficient protection and use of the natural meadow resources (Pre´vosto et al., 2004; Grace et al., 2007). Our results are also helpful for knowing the upland meadow, and recovering of the degraded meadow by using natural vegetation.
MATERIALS AND METHODS
Study area. The study was conducted at twelve sites across an upland meadow transect in the Liaoning Province of China. These sites represented a range of vegetation types .The transect covered latitudes from 41.33° to 42.71° N, and longitudes from 119.47° to 122.54° E. Altitude ranged from 218 to 800 m a.s.l. Along the transect, the mean annual temperature ranged from 5.6 to 7.9 °C, the mean annual precipitation from 391.9 to 510.4 mm. Each selected site was protected by an enclosure. The main vegetation types corresponded to a semiarid upland meadow. The main species at this site are Bothriochloa ischaemum, Cleistogenes polyphylla, Leymus chinensis, Ostryopsis davidiana, Lespedeza daurica, Phragmites adans, Arundinella anomala, Carex callitrichos, Zoysia japonica, and Stipa grandis.
Experimental design. The 12 field sites along the transect were sampled in July and August (i.e., mid-summer) of 2011, 2012 and 2013. Their geographical positions are shown in Fig. 1B and C. At each site, we established 24-30 quadrats (1 x 1 m) and followed the method of the transect line (Mueller-Dombois & Ellenberg, 1974).
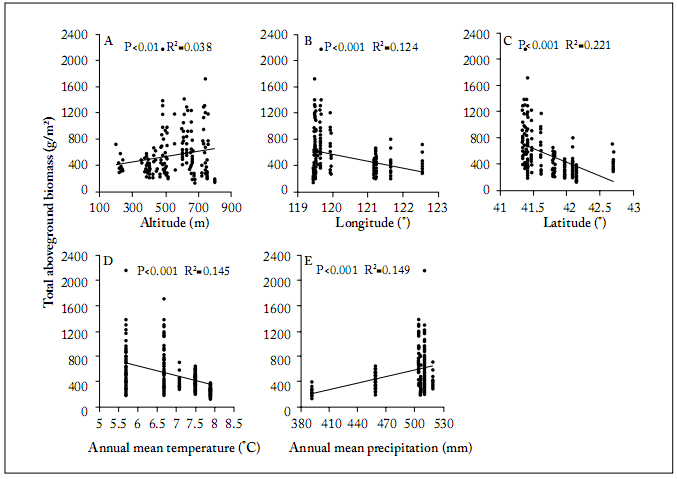
Fig. 1. Linear regressions for the effects of altitude, longitude, latitude, mean annual temperature and mean annual precipitation on total aboveground biomass. Panels B and C show geographical positions of the study sites on the X axis.
Fig. 1. Regresiones lineales de los efectos de altitud, longitud, latitud, temperatura anual promedio y precipitación anual promedio versus la biomasa aérea total. Los paneles B y C muestran la posición geográfica de los sitios de estudio en el eje X.
Measurements. In each quadrat, the numbers of plant species was first counted for calculating the diversity indices (i.e., Shannon-Wiener and Simpson indexes). Thereafter, the aboveground plant material was harvested, oven-dried at 60 °C for 48 h, and then weighed to determine total aboveground biomass. Mean annual precipitation (MAP) and mean annual temperature (MAT) were calculated from the World-Clim database (Hijmans et al., 2005).
Data processing and analysis. The Shannon-Wiener and Simpson indexes were analyzed by the R software package. Regression analysis was used to analyze the relationships between the diversity indexes and biomass of the community, and between the diversity indexes and environmental factors. Data analysis was performed using the SPSS software 17.0.
RESULTS AND ANALYSIS
The relationship of biomass and ecological factors. The linear regressions between biomass versus longitude, latitude and mean annual temperature showed significantly negative correlations (P<0.001, Fig. 1 B, C, D). The linear relationships of biomass versus altitude (P<0.01, R2 =0.038) and mean annual precipitation (P<0.001) were significantly positive (Fig. 1 A, E).
Relationship of species diversity and geographical factors. The Simpson´s index was negatively correlated with MAP (r= -0.158, n=185, P<0.05). However, it was not the case for other environmental factors (-0.041=r≤0.070, n=185, P>0.05). The Shannon-Wiener index was not significantly correlated with any environmental factor (P>0.05). Mean annual temperature was significantly, negatively correlated with altitude (r= -0.337, n=185, P<0.05), but not with MAP (r= -0.0060, n=185, P>0.05).
Relationship of biomass and species diversity. Total aboveground biomass and species diversity (calculated with the Shannon-Wiener and Simpson indexes) were negatively, significantly correlated (Fig. 2, P<0.05). However, it has to be recognized that the determination coeficients (R2 <0.07) were low (Fig. 2).

Fig. 2. Relationships between the Shannon-Wiener and Simpson indexes versus total aboveground biomass.
Fig. 2. Relaciones entre los índices de Shannon-Wiener y Simpson y la biomasa aérea total.
DISCUSSION
In grasslands, productivity is significantly influenced by the geographic position which decides the effect on the climatic variation (Briggs et al., 1995). Bruun et al. (2006) observed a significantly negative quadratic function between longitude and total productivity, and a positive correlation between latitude and species diversity. Their results are not consistent with the linear, decreasing biomass with increasing latitude in our research. Our findings agree with those reported by Begon et al. (1986). Longitude and latitude afect biomass because they determine differences in micro-climates (Begon et al., 1986; Asaeda et al., 2005).
In the alpine meadow, the relationship between productivity and altitude has been shown to be linear, significantly negative because of the decreases in soil nutrients and temperature with increasing altitude (Roem et al., 2000; Rastetter et al., 2004). Even though the coeficient of determination between altitude and biomass was significant, altitude only explained less of 4% of the variation in total aboveground plant biomass (Fig. 2).
Precipitation is regarded as a critical factor for controlling productivity in most steppes, because it affects soil water availability (O’Connor et al., 2001; Huxman et al., 2004; Hu et al., 2010). This is mostly why the relationship between mean annual precipitation and biomass was positive in our study. This result is consistent with that of previous studies (Oberbauer& Billings, 1981; Begon et al., 1986; Giorgetti et al., 1997; Yang et al., 2009). Another reason for the plant response to increasing precipitation is the result of a long-term vegetation adaptation to local habitats in the whole study research area. It is well known that plants have several avoidance and/or tolerance mechanisms to deal with various levels of soil water availability (Brown, 1995).
The relationship between productivity and species diversity is a basic but controversial theme, which is gaining increasing attention in recent years (Wu et al., 2014). However, reports on the relationship between species diversity and biomass have received much less attention. Our study showed that statistically, the relationship between biomass and species diversity in upland meadow was significantly, negatively linear. However, the coeficient of determination was less than 7%. This is, the high number of data points (n=185) made significant the relationship (P<0.05), although less than 7% of the total variation in total aboveground biomass was explained by species diversity (Fig. 2). Distribution of data points in Fig. 2 are similar to those reported by Lawton in 1994 when defning the idiosyncractic response hypothesis. This hypothesis suggests that ecosystem function changes when species diversity changes. However, the magnitude and direction of these changes is unpredictable because the roles of individual species are complex and varied (Lawton, 1994). The upland meadow is always from semi-arid to arid type, which are therefore nonequilibrium grazing systems. Density-independent, stochastic biotic conditions and variable plant growing conditions are characteristics of the upland meadow (Ellis & Swift, 1988). Rangeland communities in semi-arid and arid sites can show low biomass and are species poor (Giorgetti et al., 1997). This is why species diversity is so important for keeping ecosystem resilience at these sites (Bai et al., 2004).
In natural plant communities, species diversity is always correlated with geographic factors (altitude, longitude, latitude: Begon et al., 1986). Previous research proposes that species diversity is positively related to heterogeneity (Lundholm & Larson, 2003). Without the inclusion of species at the same trophic level and extreme habitats, it does not appear that the unimodal altitude-diversity curve will show (Grime, 1973a,b; Grime, 1979). These views can illustrate why some findings of diversitygeographic factor patterns are not always unimodal, and often change with taxonomic groups and the extent of the investigation. To restore and keep a high level of species diversity and ecosystem functioning, it will be necessary to maintain a moderate herbivory stocking rate in the study upland meadow. Wu et al. (2006) already reported that social, ecological and economic dimensions must be integrated to a landscape-level for restoration and management of the Inner Mongolia grassland.
ACKNOWLEDGEMENTS
We acknowledge support from the National Science Foundation of China (Grant 31570332, 31472134, 31360574), Outstanding Young Scholars Foundation of Higher University in Liaoning Province (LJQ2014004), Open Foundation of Key Laboratory for Vegetation Ecology, Ministry of Education, Northeast Normal University, Open foundation of Hulunber Grassland Ecosystem Research Station of Chinese Academy of Agricultural Sciences, and College Students Innovation and Entrepreneurship Project of Liaoning Province (201511035000005), Students’ Innovation and Entrepreneurship Training Project of Liaoning University (x201510140161). We thank constructive comments and criticisms of anonymous referees on an earlier version of this manuscript.
REFERENCES
1. Asaeda, T., D.N. Hai, J. Manatunge, D. Williams & J. Roberts (2005). Latitudinal characteristics of below- and above-ground productivity of typha: a modelling approach. Annals of Botany 96: 299-312. [ Links ]
2. Bai, Y.F., J.G. Wu, Q.M. Pan, J.H. Huang, Q.B. Wang, F.S. Li, A. Buyantuyev & X.G. Han (2007). Positive linear relationship between productivity and diversity: evidence from the Eurasian steppe. Journal of Applied Ecology 44: 1023-1034. [ Links ]
3. Bai, Y.F., X.G. Han, J.G. Wu, Z.Z.Chen & L.H. Li (2004). Ecosystem stability and compensatory efects in the Inner Mongolia grassland. Nature 431(7005): 181-184. [ Links ]
4. Begon M., J.L. Harper & C.R. Townsend (1986). Ecology. Individuals, Populations, and Communities. Sinauer Associates, Inc. Sunderland. Massachusetts. [ Links ]
5. Begossi, A. (2000). Conservation Ecology, Human Ecology and the management of the commons. IASCP. [ Links ]
6. Briggs, J.M. & A.K. Knapp (1995). Inter annual variability in primary production in tall grass prairie: climate, soil moisture, topographic position and fre as determinants of aboveground productivity. American Journal of Botany 82: 1024-1030. [ Links ]
7. Brown, R.W. (1995). The water relations of range plants: Adaptations to water deficits. In: D.J. Bedunah and R.E. Sosebee (eds.), pp. 291-413. Wildland plants: Physiological ecology and developmental morphology. Society for Range Management, Denver, Colorado. 710 p. [ Links ]
8. Bruun, H.H., J. Moen, R. Virtanen, J.A. Grytnes, L. Oksanen & A. Angerborn (2006). Effects of altitude and topography on species richness of vascular plants, bryophytes and lichens in alpine communities. Journal of Vegetation Science 17: 37-46. [ Links ]
9. Costanza, R., B. Fisher, K. Mulder, S. Liu & T. Christopher (2007). Biodiversity and ecosystem services: A multi-scale empirical study of the relationship between species richness and net primary production. Ecological Economics 61: 478-491. [ Links ]
10. Ellis, J.E. & D.M. Swift (1988). Stability of African pastoral ecosystems: alternate paradigms and implications for development. Journal of Range Management 41: 450-459. [ Links ]
11. Fischer, J., D.B. Lindenmayer & A.D. Manning (2006). Biodiversity, ecosystem function, and resilience: ten guiding principles for commodity production landscapes. Frontiers in Ecology & the Environment 4: 80-86. [ Links ]
12. Fukami, T. & P.J. Morin (2003). Productivity–biodiversity relationships depend on the history of community assembly. Nature 424(6947): 423-426.
13. Gillman, L.N. & S.D. Wright (2006). The influence of productivity on the species richness of plants: a critical assessment. Ecology 87: 1234-1243. [ Links ]
14. Giorgetti, H., O.A. Montenegro, G. Rodríguez, C.A. Busso, T. Montani, M.A. Burgos, A.C. Flemmer, M.B. Toribio y S.S. Horvitz. (1997). The comparative influence of past management and rainfall on range herbaceous standing crop in east-central Argentina: 14 years of observations. Journal of Arid Environments 36: 623-637. [ Links ]
15. Grace, J.B., T.M. Anderson, M.D. Smith, E. Seabloom, S.J. Andelman, Meche, G. Weiher, E. Allain, L. Jutila, H. Sankaran, M. Knops, J. Ritchie & M. Willig (2007). Does species diversity limit productivity in natural grassland communities. Ecology Letters 10: 680-689. [ Links ]
16. Grime, J.P. (1973a). Competitive exclusion in herbaceous vegetation. Nature 242: 344-347. [ Links ]
17. Grime, J.P. (1973b). Control of species density in herbaceous vegetation. Journal of Environmental Management 1: 151-167. [ Links ]
18. Grime, J.P. (1979). Plant strategies and vegetation processes. Wiley, UK. [ Links ]
19. Guo, Q. & W.L. Berry (1998). Species richness and productivity: dissection of the hump-shaped relationships. Ecology 79: 2555-2559. [ Links ]
20. Hector. A., Y. Hautier, P. Saner, L. Wacker, R. Bagchi, J. Joshi, M. Scherer-Lorenzen, E.M. Spehn, E. Bazeley-White, M. Weilenmann, M.C. Caldeira, P.G. Dimitrakopoulos, J.A. Finn, K. HussDanell, A. Jumpponen, C.P. Mulder, C. Palmborg, J.S. Pereira, A.S. Siamantziouras, A.C. Terry, A.Y. Troumbis, B. Schmid & M. Loreau (2010). General stabilizing effects of plant diversity on grassland productivity through population asynchrony and overyielding. Ecology 91: 2213-2220. [ Links ]
21. Hijmans, R. J., S.E. Cameron., J.L. Parra., P.G. Jones & A, Jarvis (2005). Very high resolution interpolated climate surfaces for global land areas. International Journal of Climatology 25: 1965-1978. [ Links ]
22. Hooper, D.U., F.S. Chapin, J.J. Ewel, A. Hector, P. Inchausti, S. Lavorel, J.H. Lawton, D.M. Lodge, M. Loreau & S. Naeem, et al. (2005). Effects of Biodiversity on Ecosystem Functioning: A Consensus of Current Knowledge. Ecological Monographs 75: 3-35. [ Links ]
23. Hu, Z.M., G.R. Yu, J.W. Fan, H.P. Zhong, S.Q. Wang & S.G. Li (2010). Precipitation-use efciency along a 4500-km grassland transect. Global Ecology & Biogeography 19(6): 842-851. [ Links ]
24. Huxman, T.E., M.D. Smith, P.A. Fay, A.K. Knapp, M.R. Shaw, M.E. Loik, S.D. Smith, D.T. Tissue, J.C. Zak, J.F. Weltzin, W.T. Pockman, O.E. Sala, B.M. Haddad, J. Harte, G.W. Koch, S. Schwinning, E.E. Small & D.G. Williams (2004). Convergence across biomes to a common rain-use eficiency. Nature 429(6992): 651-654. [ Links ]
25. Lawton, J.H. (1994). What do species do in ecosystems. Oikos 71: 367-374. [ Links ]
26. Lundholm, J.T. & D.W. Larson (2003). Relationships between spatial environmental heterogeneity and plant species diversity on a limestone pavement. Ecography 26: 715-722. [ Links ]
27. Ma, W.H., J.S. He, Y.H. Yang, X.P. Wang, C.Z. Liang, M. Anwar, H. Zeng, J.Y. Fang & B. Schmid (2010). Environmental factors covary with plant diversity-productivity relationships among Chi nese grassland sites. Global Ecology & Biogeography 19: 233-243. [ Links ]
28. Maestre, F.T., M.A. Bradford & J.F. Reynolds (2006). Soil heterogeneityand community composition jointly infuence grasslands biomass. Journal of Vegetation Science 17: 261-270. [ Links ]
29. Mittelbach, G.G., C.F. Steiner, S.M. Scheiner, K.L. Gross, H.L. Reynolds, R.B. Waide, M.R. Willig, S.I. Dodson & L. Gough (2001). What is the observed relationship between species richness and productivity? Ecology 82: 2381-2396. [ Links ]
30. Mueller-Dombois, D. & H. Ellenberg (1974). Aims and methods of vegetation ecology. John Wiley & Sons, Inc. New York. [ Links ]
31. O’Connor, T.G., L.M. Haines & H.A. Snyman (2001). Influence of precipitation and species composition on phytomass of a semiarid African grassland. Journal of Ecology 89: 850-860.
32. Oberbauer, S.F. & W.D. Billings (1981). Drought tolerance and water use by plants along an alpine topographic gradient. Oecologia 50: 325-331. [ Links ]
33. Oksanen, J. (1996). Is the humped relationship between species richness and productivity an artefact due to plot size? Clinical & Experimental Allergy 84: 293-295. [ Links ]
34. Postor, J., A. Downing & H.E. Erickson (1996). Species-area curves and diversity-productivity relationships in beaver meadows of Voyageurs National Park, Minnesota, USA. Oikos 77: 399-406. [ Links ]
35. Prévosto, B., L. Kuiters, Bernhardt-Römermann M, Dölle, M., W. Schmidt & M. Hofmann (2004). Impacts of land abandonment on vegetation: successional pathways in European habitats. Enabling Relationships in Health & Social Care 46: 167-179. [ Links ]
36. Rastetter, E.B., B.L. Kwiatkowski, S.L. Dizés & J.E. Hobbie (2004). The role of down-slope water and nutrient fuxes in the response of Arctic hill slopes to climate change. Biogeochemistry 69: 37-62. [ Links ]
37. Rodwell, J.S., C.D. Pig, D.A. Ratclife, A. J.C. Mallock, H.J.B. Birks & M.C.F. Proctor (1992). Grasslands and Montane Communities. Cambridge University Press, Cambridge. British Plant Communities. [ Links ]
38. Roem, W.J. & F. Berendse (2000). Soil acidity and nutrient supply ratio as possible factors determining changes in plant species diversity in grassland and heathland communities. Biological Conservation 92: 151-161. [ Links ]
39. Schmid, B (2002a). The species richness-productivity controversy. Trends in Ecology & Evolution 17: 113-114. [ Links ]
40. Schmid, B., A. Hector, M. Huston, P. Inchausti, I. Nijs, P. Leadley & D. Tilman (2002b). The design and analysis of biodiversity experiments. Oxford University Press, Oxford. [ Links ]
41. Smith, R.S. (1985). Conservation of Northern Upland Meadows. Yorkshire Dales National Park Authority, Bainbridge. [ Links ]
42. Tilman, D., D. Wedin & J. Knops (1996). Productivity and sustainability infuenced by biodiversity in grassland ecosystems. Nature 379(6567): 718-720. [ Links ]
43. Tilman, D., P.B. Reich, J. Knops, D. Wedin, T. Mielke & C. Lehman (2001). Diversity and productivity in a long-term grassland experiment. Science 294(5543): 843-845. [ Links ]
44. Virtanen. R., J.A. Grytnes, J. Lenoir, M. Luoto, J. Oksanen, L. Oksanen & J.C. Svenning (2013). Productivity-diversity patterns in arctic tundra vegetation. Ecography 36: 331-341. [ Links ]
45. Waide, R.B., M.R. Willig, C.F. Steiner, G. Mittelbach, L. Gough, S.I. Dodson, G.P. Juday & R. Parmenter (1999). The relationship between productivity and species richness. Annual Review of Ecology & Systematics 30: 257-300. [ Links ]
46. Wardle, D.A. & O. Zackrisson (2005). Effects of species and functional group loss on island ecosystem properties. Nature 435(7043): 806-810. [ Links ]
47. Weigelt, A., J. Schumacher, C. Roscher & B. Schmid (2008). Does biodiversity increase spatial stability in plant community biomass? Ecology Letters 11: 338-347. [ Links ]
48. Wu, J.G., G.M. Jiang & X.G. Han (2006). Restoration and management of the Inner Mongolia Grassland require a sustainable strategy. AMBIO 35: 269-270. [ Links ]
49. Wu, J.S., Z.X. Shen & X.Z. Zhang (2014). Precipitation and species composition primarily determine the diversity-productivity relationship of alpine grasslands on the Northern Tibetan Plateau. Alpine Botany 124: 13-25. [ Links ]
50. Wu, Z.Y. (1980). Vegetation of China. Science Press, Beijing. [ Links ]
51. Yang, Y.H., J.Y. Fang & Y.D. Pan (2009). Aboveground productivity in Tibetan grasslands. Journal of Arid Environments 73: 91-95. [ Links ]














