INTRODUCTION
In recent years, studies have been carried out where different wastes are incorporated into red ceramics as a viable option for the environment and for the recovery of some wastes (thus avoiding landfill disposal) 1. This is because products made from red clays have a great potential to absorb a large amount of materials that are incorporated into the raw material due to the huge production volume, long lifetime of these products, inertisation of waste and the tolerance of red ceramic products and their processing to variations in raw material composition making them attractive for encapsulation of solid waste 2. This allows the incorporation of reasonable amounts of waste into the composition of pastes used in the manufacture of ceramic building products 3.
Glass used by the general population in the form of containers is relatively inert and non-biodegradable 4. Despite being a 100% recyclable material, there is a large volume of material generated causing environmental problems with regard to its storage in the appropriate place 5. This encourages studies and research to reuse this waste in the different manufacturing processes 6,7. The incorporation of waste glass in products made from clay is a natural alternative considering the compatibility of the chemical composition of these materials and glass which is mainly composed of silica (SiO2), sodium oxides (Na2O) and calcium oxide (CaO) 8,9.
Although there are different studies showing the addition of the residues in red ceramics, many of these lack information pointing to the compatibility of the materials 10,11. It is extremely important that there is an interaction between the incorporated residue and the clay, so that it is not just an aggregate in the composition, thus becoming a filler.
This study aims to evaluate the compactibility and characterise the microstructure of ceramic pieces with the addition of glass through microscopic analysis. The effects of firing temperature on the mixture of clay and glass residue were discussed in terms of physico-mechanical properties and microstructure.
MATERIALS AND METHODS
The raw material used as the basis for the study was provided by a ceramic company in Santa Catarina, Brazil. The standard paste consisting of a mixture of different clay materials was taken after the maturation and mixing process, where 100 kg of ceramic paste were collected from four different points. Then, they were mixed and a sufficient quantity was left for laboratory tests, obtaining at the end approximately 30 kg.
Approximately 10 kg of glass bottles were collected and passed through the beneficiation process. The bottles were washed, aggregated and transformed into fragments. In sequence they were dried in an electric dryer (DeLeo brand n° 2211) and passed in a laboratory laminator (Bertan brand) for the crushing of the grains. Finally, the crushed material was passed through a 10 mesh (2 mm) sieve.
The red clay and glass residue were chemically characterised using a Philips wavelength dispersive X-ray fluorescence spectrometer (WDXRF) model PW2400. Mineralogical composition was carried out on a Shimadzu X-ray diffractometer model XRD 6000.
Based on published studies 12,13, 20 % (by weight) of waste glass was used in the ceramic paste. The preparation was carried out through a helical screw mixer. Subsequently, the paste was put to rest for 24 hours for moisture homogenisation. The samples were prepared by piston extrusion (without vacuum): 250 mm reduction with a 27 mm funnel with 45° angle. With a constant pressure recorded by the manometer, 30 samples were made. All the pieces were then placed in an electric dryer (DeLeo brand No 2211) at a temperature of 65 ± 5 °C for 24 h plus 12 h at a temperature of 100 ± 10 °C for complete elimination of moisture. The last step of the heat treatment process consists of firing, where the formulation was sintered in an electric muffle furnace (Jung, model 7012), with a heating rate of 2 °C/min and a holding time of 120 min, at three temperatures: 800, 900 and 1000 °C.
The technological properties of compressive strength and water absorption of the samples were performed according to ABNT NBR 15270. The mechanical test was carried out on EMIC DL-20000 equipment 14,15.
A scanning electron microscope (SEM) (Zeiss, EVO MA10) and an optical microscope (Olympus, BX41M-LED) were used for grain boundary analysis and to verify the interaction between the ceramic and the glass residue.
RESULTS AND DISCUSSION
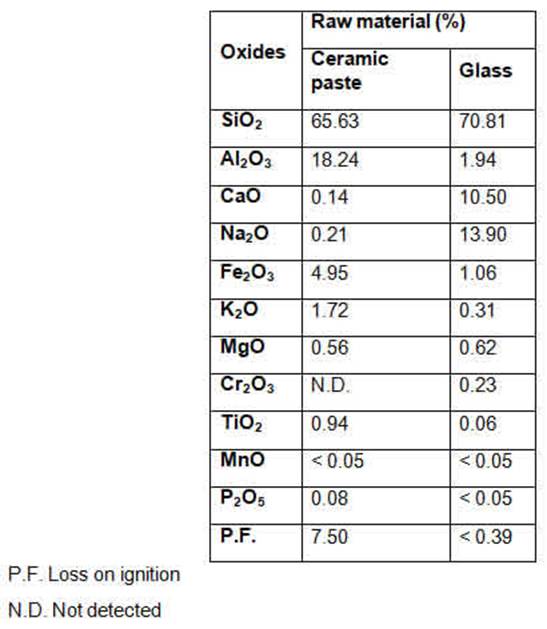
Table 1 shows the chemical composition of the glass containers, used as an alternative raw material in the study, and of the ceramic paste.
It is possible to observe that silica (SiO2) is predominant in the composition of glass, and is the basic raw material with the function of vitrification. Sodium oxide (Na2O) is the second most important oxide in the glass composition, with the function of increasing mechanical strength, as well as alumina (Al2O3). The oxides of calcium (CaO), magnesium (MgO) and potassium (K2O) are other compounds that make up the glass, and have functions such as increasing resistance to chemical attack and increasing resistance to thermal shock. The analysis confirmed that the glass is of the sodium-calcium type. The relevance of the study, the presence of silica (SiO2) and sodium oxide (Na2O) in significant levels helps in the formation of liquid phases improving the quality of the piece, especially in relation to mechanical strength, as they are fluxing oxides and help in the vitrification of the material 8,16.
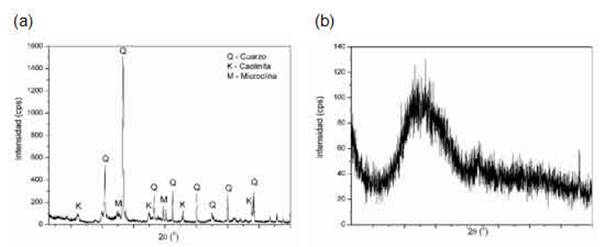
The oxides of the chemical elements presented in Table 1 are present in the crystalline phases identified in the X-ray diffractogram in Figure 1(a) for the clay. The crystalline phases of quartz (SiO2, JCPDS no. 46-1045), microcline (KAlSi3O8, JCPDS no. 19-932) and kaolinite (CaAl2Si2O8.4H2O,JCPDS no. 20-0452), characteristic of a clay body used for the manufacture of red ceramics, were identified. Figure 1(b) shows the diffractogram for the glass residue, an amorphous structure is observed.
The technological analysis of the sample with 20 % glass incorporated into the clay mass (Figure 2) indicates that the increase in temperature densifies the final piece. This behaviour is a consequence of the formation of liquid phases caused by the high temperature where the softening point of the glass is exceeded. This causes the glass to fill the porosity sites contributing to the decrease of water absorption of the ceramic pieces 17-19.
It should be emphasised that the use of solid wastes with fluxing characteristics, such as glass, has the tendency to present a significant increase in the compressive strength of the ceramic piece 14-15. It is also necessary to take into consideration that the increase of temperature improves the mechanical behaviour of the ceramic piece as a consequence of the sintering of the grains causing a higher density. In relation to the particle size of the glass residue, the larger the particle size, the higher the energy required to generate the liquid phase 16, making higher temperatures more effective in this study.

Figure 2: Analysis of the mechanical compressive strength versus water absorption of the samples with 20% glass incorporated.
SEM analysis of the mixture of the components at 800 °C (Figure 3(a)) shows the presence of separated particles, where the glass at this temperature only appears as an aggregate, i.e. substantially inert. After firing at 900 °C (Figure 3(b)) of the sample surface, a stronger interaction between the glass and the ceramic body was observed, forming a glass base very different from the sample at 800 °C. At 1000 °C (Figure 3(c)) it can be observed that there is a stronger interaction during firing of the glass particles with the matrix. That is, as the firing temperature was increased, the glass phases become more fluid allowing a better contact and interaction with the solid phase of the clay and thus a decrease in porosity occurs resulting in a lower water absorption which changes from 17.3% at low temperature to 12.9 % for firing at 1000 °C 20. Another point observed is the geometry of the particles, which, as the temperature increases, change from being particles with more spiny ends to more oval, this phenomenon is explained by the increase of the energy applied to sintering which, from the same principle as highlighted above, changes the morphology of the glass particles 21.

Figure 3: SEM micrographs of red ceramics with a glass content of 20 wt.% as a function of firing temperatures: (a) 800 °C, (b) 900 °C and (c) 1000 °C.

Figure 4: Optical microscopy images of red ceramics with a glass content of 20 wt.% as a function of firing temperatures: (a) 800 °C, (b) 900 °C and (c) 1000 °C.
It is possible to observe that from 900 °C onwards there is a release of volatile substances derived from the high temperatures, which caused the generation of bubbles in the formation of the liquid phase and left pores in the sample as observed in some points of Figure 4. The porosity at higher temperatures may result from the compaction stage and be generated in the firing process itself by loss of mass caused by the release of volatiles or as a consequence of the different coefficients of thermal expansion of the phases present in the ceramic, generating cracks. This behaviour is explained by a phenomenon called overfiring porosity, which occurs when a material is fired at a temperature above that required to produce a liquid phase. The result of this phenomenon is the appearance of deformations, bubbles or pores 18,22,23.
CONCLUSION
From the evaluation of the microscopy results of the samples, it was possible to observe that there is a large interaction of the glass composite-based ceramic clay matrix.
At 800 °C the glass remains inert, and at higher temperatures (900 and 1000 °C) there were physical changes resulting in vitrification and pore generation as a product of gas release.
Despite these phenomena, it is concluded that glass is a material that is suitable for the manufacture of clay-based products, giving these wastes a sustainable environmental destination.