Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Salud colectiva
Print version ISSN 1669-2381On-line version ISSN 1851-8265
Salud colect. vol.12 no.3 Lanús Sept. 2016
http://dx.doi.org/10.18294/sc.2016.1073
Articles
Clinical trials in Latin America: implications for the sustainability and safety of pharmaceutical markets and the wellbeing of research subjects
1PhD in Public Health. Associate Professor, Houston Health Science Center, School of Public Health, University of Texas at Houston. Visiting professor, Georgetown University, USA.
2PhD in Law, PhD in Sociology. Professor Emeritus, Department of Sociology, University of Texas at Austin, USA.
ABSTRACT This study sought to verify whether drugs approved by the US Food and Drug Administration (FDA) were registered, commercialized and sold at affordable prices in the Latin American countries where they had been tested, as well as to ascertain their contribution to the quality of the pharmaceutical market. The list of New Molecular Entities (NMEs) approved by the FDA in 2011 and 2012 was consulted to determine the countries where pivotal trials were conducted. Affordability was assessed as a proportion of income and information on safety and efficacy was gathered from independent drug bulletins. In the study years, 33 medications were tested in 12 Latin American countries. Only 60% of the expected registrations had been completed by September 2014. With one exception, all products for which pricing information was obtained (n=18) cost more than one monthly minimum wage in all countries. Only five drugs were classified as "could be better than available treatments." Just one of the NMEs responds to the health care priorities in low and middle income countries.
KEY WORDS: Clinical Trial; Drug Industry; Drug Price; Pharmaceutical Trade; Latin America.
INTRODUCTION
The phenomenal cost of many novel treatments calls into question whether low- and middle-income countries will be able to access them.1 The issue becomes more poignant as an increasing number of pivotal[a] trials are carried out in these countries where patients are more easily recruited and retained,2,3 expediting the completion of clinical trials. Shorter trials allow the pharmaceutical industry to hasten the attainment of marketing approval for the new molecular entities (NMEs) and maximize the benefits they can accumulate during their market-exclusivity period.4 The inability to recruit enough research participants in high-income countries5 and the few regulatory hurdles in low- and middle-income countries reinforce this tendency.
There has been little scrutiny of the consequences that conducting clinical trials has on the availability and the appropriate and safe use of new pharmaceuticals, as well as on the private and public health budgets of the host countries. International ethical declarations require that approved NMEs be made available to the populations in which they have been tested.6 The Latin American regulatory agencies base their marketing decisions on the actions taken by their counterparts in "high sanitary surveillance countries" (United States, Japan, Australia, selected individual countries in Europe and the European Medicines Agency).
Additionally, Latin American patients and patient groups are increasingly using the judiciary system to exercise their constitutional right to health, including access to new and expensive pharmaceuticals. Vargas-Pélaez et al.7 conducted a scoping study of the literature on lawsuits for access to medicines and health services. They identified 65 articles, 80% of which involved a Latin American country (68% Brazil, 9% Colombia and 3% Argentina). The Latin American authors cited in this study mentioned that in some cases the courts decide without taking into consideration the evidence of drug efficacy and safety or the appropriateness of the treatment for a particular patient, possibly putting the plaintiff at risk of adverse effects and drug misuse. Moreover, some authors asserted that the pharmaceutical industry was interested in promoting access to medicines through the courts, because it resulted in the inclusion of medicines in the public formularies that might be useful for only a small group of patients rather than the needs of society.
In other words, current judiciary, ethical and regulatory conditions lead to NMEs being made available in the countries where tested. The final result is that those countries where the NMEs have been tested have to cover the costs of the NMEs, regardless of their safety profile and whether they offer any advantage over cheaper existing treatments. While the magnitude of the financial impact will differ across countries and will in part depend on the sales price of the NMEs in each country, public coverage of these new and expensive NMEs will strain public pharmaceutical budgets.
The health consequences of outsourcing clinical trials have been off the radar of researchers, possibly because it is assumed that the regulatory agencies of "high sanitary surveillance countries" only allow the commercialization of products that are safe and effective, and what is available to the residents of high income countries ideally should also be offered to the residents of less prosperous countries, especially if they have contributed to their development.
Using information on pricing and value of the NMEs approved by the US Food and Drug Administration (FDA) in 2011 and 2012 that were tested in Latin America, this article analyzes some of the health, financial and ethical consequences of outsourcing clinical trials to the region.[b]
This paper explores the following questions: 1) Are new molecular entities approved by the FDA in 2011 and 2012 available in the Latin American countries where the pivotal trials were conducted? 2) If registered, are they marketed at affordable prices? 3) Do these NMEs add therapeutic value to existing treatments, as reported in independent drug bulletins? A discussion on the implications of conducting clinical trials in Latin America under current judicial, regulatory and ethical conditions for the national pharmaceutical markets and research participants follows.
METHODS
This is a cross-sectional study. The list of NMEs approved by the Food and Drug Administration (FDA) in 2011 and 2012 was obtained from FDA publications.11,12 Gadobutrol (Gadovist(r)) was approved during the study period but excluded from the study because it is a contrast dye used in radiology, not a pharmaceutical treatment. The FDA's medical reviews of the NMEs, included in the FDA's drug approval history, provided the names of countries where the trials had been conducted. If this information was not available in the medical reviews, we obtained it from the trial sponsors. The drug approval histories can be found in Drugs@FDA.13
Obtaining the regulatory and marketing status of NMEs
To obtain the regulatory status of the NME in each country, we searched the pharmaceutical registers. The information included in the registers varies slightly by country. Brazil, Chile and Colombia maintain a register of approved pharmaceuticals; Argentina has a register of marketed products; Mexico publishes a list of the products approved per time period; and Peru catalogues products available in pharmacies. Table 1 offers a list of the websites consulted. For the countries without registers or with incomplete registers (Costa Rica, Ecuador, Panama, Peru, Uruguay) we approached the regulatory agencies. All attempts to contact the regulators in Dominican Republic and Venezuela failed. Using the information provided on the websites of the pharmaceutical companies we contacted the USA headquarters to gather information on the marketing status of their products in the selected countries.
Determining the price of NMEs
In Latin America, about 78% of the medicines are paid out-of-pocket in retail pharmacies.27 Since the products of interest were not included in the World Health Organization-Health Action International (WHO/HAI) medicine prices database, we obtained the price of the unit dose of each product from the countries' price observatories, which report the maximum price to consumers (Brazil, Mexico) or the observed consumer prices (Argentina, Chile, Colombia, Ecuador, Peru) (Table 1).
The consumer prices in Costa Rica, where there is no observatory, and of Argentine products not tracked by the observatory were provided by pharmacological experts who obtained them from local distributors. The quantities needed to complete a course or a year of treatment was calculated using the FDA-approved product label. The pricing information was gathered between August 25 and September 20, 2014.
Measuring affordability
Wealth was measured using: 1) the monthly minimum wage in 2014, obtained from public announcements in the media; 2) the monthly income per capita, from the 2013 World Bank database of Gross Domestic Product (GDP) per capita;28 3) the monthly household net adjusted disposable income;29 and 4) the monthly household financial wealth as reported by the Organisation of Economic Co-operation and Development (OECD),30 which was only available for 2011 and for Brazil, Chile and Mexico (Table 2).
Table 2 Monthly minimum wage, monthly income per capita, household net-adjusted disposable income, and household financial wealth (in USD), by country.
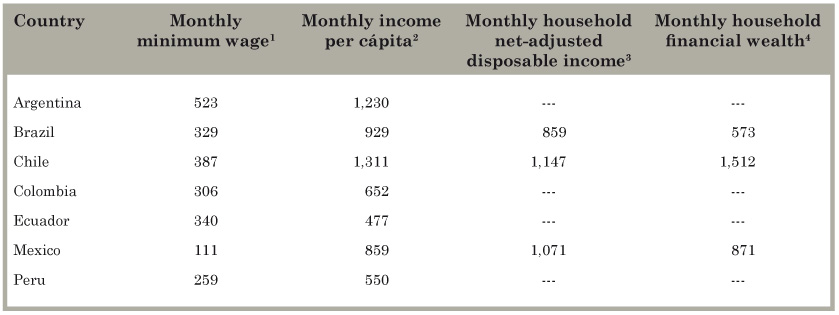
Source: Homedes and Ugalde.8) --- No data available at the time of the study. 1The minimum wage per country was obtained from public announcements in the media. For Argentina, the monthly minimum wage is as of September 2014; for Brazil and Chile, as of July 2014; for Colombia, Ecuador, and Mexico, as of January 2014; for Peru, as of June 2012. Local currencies were exchanged into dollars according to the official exchange rate of September 1, 2014. 2Income per capita comes from the 2013 database of the World Bank28 adjusted by authors to the median value of the year. 3Household net-adjusted disposable income from the Better Life Index of the Organisation for Economic Co-operation and Development for the year 2014.29 It is defined as "the amount of money that a household earns, or gains, each year after taxes and transfers. It represents the money available to a household for spending on goods or services."30) 4Household financial wealth is from the Better Life Index of the Organisation for Economic Co-operation and Development for the year 2014.29 It is defined as "the total value of a household's financial worth, or the sum of their overall financial assets minus liabilities. Financial wealth takes into account: savings, monetary gold, currency and deposits, stocks, securities and loans."30
These measurements have a number of limitations, due to aspects related to the difficulty of measuring individual and household income and wealth using aggregate data:
Income distribution is very unequal in most countries. As can be seen in Table 3 the Gini index of wealth distribution shows a very large concentration of wealth. The table also shows that for practically all countries except Argentina and Venezuela, the wealthiest ten percent of the population receives more than 35% of the GNP.
The non-monetized activities, which are highly prevalent in Latin America, and the remittances sent through informal channels - that in some countries such as Mexico and Ecuador are significant - can only be estimated and we do not know the accuracy of the estimates.
Income generated from illegal activities such as contraband, drugs or prostitution is not considered when estimating the GNP of a nation.
A relatively large number of breadwinners do not receive a monthly check and have difficulties in determining the yearly income. Frequently, agricultural workers and those in the informal sector do not receive the minimum wage.
Databases from international organizations convert local currencies into dollars at a given moment which does not coincide with the time of purchase of the medication, and currency exchanges may vary significantly throughout the year. Some currencies are relatively stable as is the case of Mexico, or Ecuador whose currency is the US dollar, but others for example the Brazilian real fluctuated against the dollar in 2014 almost 9%, and the Argentine peso has also experienced severe depreciations. A second problem is related with the value of the currency. The official and the real value are not always the same, and in some cases the difference can be large. During the years of our analysis the real value (in the black market) of the peso in Argentina was about 40% higher than the official.
Table 3 Gini index and income distribution in the lowest and highest deciles, by country.
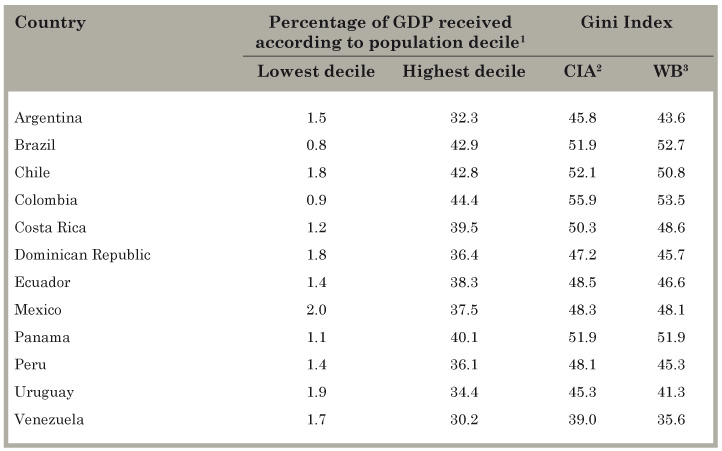
Source: Homedes and Ugalde.8) 1Data correspond to the following years: Argentina, 2010; Brazil, 2008; Chile, 2009; Colombia, 2010; Costa Rica, 2009; Dominican Republic, 2010; Ecuador, 2010; Mexico, 2010; Panama, 2010; Peru, 2010; Uruguay, 2010; Venezuela, 2006. As clarified in the database, "data come from household surveys, the results adjusted for household size. Nations use different standards and procedures in collecting and adjusting the data. Surveys based on income will normally show a more unequal distribution than surveys based on consumption. The quality of surveys is improving with time, yet caution is still necessary in making inter-country comparisons."31) 2Data from the Central Intelligence Agency (CIA)31 corresponds to the following years: Argentina, 2009; Brazil, 2012; Chile, 2009; Colombia, 2010; Costa Rica, 2008; Dominican Republic, 2010; Ecuador, 2013; Mexico, 2008; Panama, 2010; Peru, 2010; Uruguay, 2010; Venezuela, 2011. 3World Bank (WB) information is for all countries for the year 2012, except for Argentina and Chile, which corresponde to the year 2011. The World Bank data "are based on primary household survey data obtained from government statistical agencies and World Bank country departments.32 For more information and specificity, see the methodology used by the World Bank.33
Similarly, problems related to household definitions and estimates of number of persons and incomes can also be highlighted. As anthropologists are well aware, the concept of a household/family is not as clearly defined as many may think. Studies of marginal neighborhoods, where very large number of Latin Americans reside, show many varieties of household formations. We can talk about blood and ritual families, and of two or more unrelated persons/families residing under the same roof, sharing some expenditures and having difficulties quantifying the amount that each contributes. We have observed that when household surveys take place, respondents have difficulties answering the questions about the number of persons who live in the household. Migrants can come and go and may have two places of residence but do not always define which the primary one is. Dual nationalities are common, with significant numbers in several countries (Argentina, Bolivia, Ecuador and Mexico) and migrants may or may not be counted in the national censuses. These circumstances are difficult for census workers to handle, and the official figures on number of persons per household may not always be correct. These uncertainties influence the quality of the data on household disposable income presented by national and international agencies. Ritual families are common in Latin America. Godfathers and mothers may not live in the household but the ritual family knows that additional income is available in important cases.
Niëns and Brouwer discuss the challenges in gathering information related to the price of medicines and incomes, and in determining affordability thresholds.34) Niëns et al. decided that a threshold of 5% of total expenditures for the purchase of medicines would classify them as unaffordable in countries such as India and Indonesia.35 O'Donnell et al. considered health care expenditures catastrophic if they surpassed 10% of yearly household income.36
In this study, the affordability of each course of treatment of one medicine is presented in relation to the monthly per capita income or household wealth and income, which is equivalent to 8.3% of the yearly income. Whether or not the calculated cost is affordable or results in a catastrophic expenditure depends on the specific socioeconomic status of the individual or the household. Given the high income inequalities of the region (Table 3), the use of average income measures will overestimate the purchasing power of those in the lowest income deciles.
Appraising the therapeutic value of NMEs
Data bases from two reputable independent drug bulletins - Revue Prescrire of the French organization Prescrire and Worst Pills, Best Pills of the Health Research Group of the US organization Public Citizen - were consulted for evidence of the added value of the NMEs to existing treatments. These reports incorporated safety information and Prescrire often included information from other bulletins. In total, we have information from 15 independent pharmacology bulletins from 11 countries. Information from Fojo et al.37 was also used to assess the value of new cancer treatments.
FINDINGS
Registration and availability
The 33 products included in this study are shown in Table 4. Obtaining information on the commercialization status of each product from pharmaceutical companies was difficult. The headquarters of some companies responded quickly (Vertex, Exelixis, GSK, BMS, Sanofi, AstraZeneca), while others referred us to their country subsidiaries or to the companies responsible for commercializing their products outside the USA, and we often had to contact them several times. The accuracy of the information provided depended on the familiarity of the respondent with the company's practices and databases. Two companies provided contradictory information, and one referred us back and forth between the innovative company and the licensee. With few exceptions (Pfizer Brazil, Colombia and Mexico; Janssen Argentina; Novartis Argentina and Colombia; Takeda-Brazil, and Boehringer-Mexico), the Latin American offices were less willing to share information than the respondents at USA headquarters. The Vice President of one of the companies wrote "In response to your question below, we have a policy of restricting the disclosure of proprietary business information/strategies unless we have a formal business relationship protected by a confidential disclosure agreement" (September 5, 2014).
Table 4 Products approved by the FDA in 2011 and 2012 that were tested in pivotal trials in Latin America.

Information on registration status and pricing helped resolve some of the inaccuracies reported by industry. For example, the conclusion was reached that bosutinib was not marketed in Argentina or Peru since it was not included on the list of marketed products (Argentina) or in the catalogue of products available in pharmacies (Peru), and price information was not available in either country. Similarly, pricing information indicated the probable availability of pertuzumab in Mexico, rivaroxaban in Colombia and Mexico, and ticagrelor in Argentina. It was impossible to confirm the NME's marketing status in ten cases: pasireotide in Brazil; rilpivirine in Argentina, Chile and Mexico; pertuzumab in Peru; teriflunomide in Chile and Mexico; tofacinitib in Costa Rica and Peru; and vandetanib in Mexico.
Combining information on registration and availability indicated that ten of the 33 products (30%) were not registered nor commercialized in any of the countries where they had been tested (aclidium bromide, axitinib, bedaquiline, bosutinib, carbozantinib, elvitegravir-cobicistat-emtricitabine-tenofovir disoproxil fumarate, lucinactant, perampanel, tbofilgastrim, ziv-aflibercept); eight (25%) were registered and commercialized in all countries where tested (aflibercept, indacaterol maleate, ipilimumab, linagliptin, regorafenib, roflumilast, taliglucerase alfa, telaprevir); and two products were registered but not marketed in any of the countries where tested (enzalutamide and ezogabine).
Table 5 presents the approval and commercialization status in September 2014 of the products included in the study that were registered and commercialized in some countries but not in others (excluding the ten NMEs that were not registered in any country and the eight that were marketed in all the countries in which they were tested). Of an expected 121 registrations, if the 33 products had been registered in all the countries where tested, only 67 (55%) were completed. Registration did not lead to commercialization on at least eight occasions, and we could not determine the marketing status of seven registered products. In total, we confirmed that 42% of the tested products were marketed where tested. In two cases the pharmaceutical company stated that a product was available when the regulatory agency said it had not been approved.
Table 5 Approval and marketing status in September 2014 of selected new molecular entities tested in Latin American countries and approved by the FDA in 2011 and 2012, by country.1

Source: Homedes and Ugalde.8 R = Registered; NR = Not registered; C = Commercialized; NC = Not commercialized; NOR = No response; CI = Contradictory information. ---- No clinical trials with the new molecular entity were conducted in the country. 1The table does not include the eight NMEs that have been registered and commercialized in all the countries, nor the ten NME that have not been registered nor commercialized in any country. 2The company BMS said that apixaban was not marketed in Mexico but we found its price. It could only be available for compassionate use. 3The Caja Costarricense de Seguridad Social (CCSS), the social security agency that provides health care including free medications to 90% of the Costa Rica's population, has not bought it and it is not available in major pharmacies.
Prices of new molecular entities in Latin America
The prices of the 21 NMEs marketed in Latin America, except three that were unavailable, are displayed in Table 6. Prices varied widely by country, both in absolute and in relative terms. Argentina had the highest absolute price for many of the drugs included in this study (aflibercept, apixaban, belatacept, ipilimumab, pasireotide, telaprevir, ticagrelor, tofacitinib, vandetanib), on occasion even doubling the second highest price (aflibercept, belatacept, tofacitinib). Brazil had the lowest prices for apixaban, belatacept, pertuzumab, rivaroxaban and ticagrelor, but the price of belimumab was more than twice that in Chile. The price of belimumab, indacaterol and ipilimumab was lowest in Chile; Colombia had the lowest price for aflibercept and tofacitinib, and the highest for rivaroxaban.
Table 6 Price of commercialized medicines by countries where tested and by number of months of income needed to pay for a course or a year of treatment according to the local price (measured in monthly minimum wage and monthly income per capita in USD). Selected countries of Latin America.1
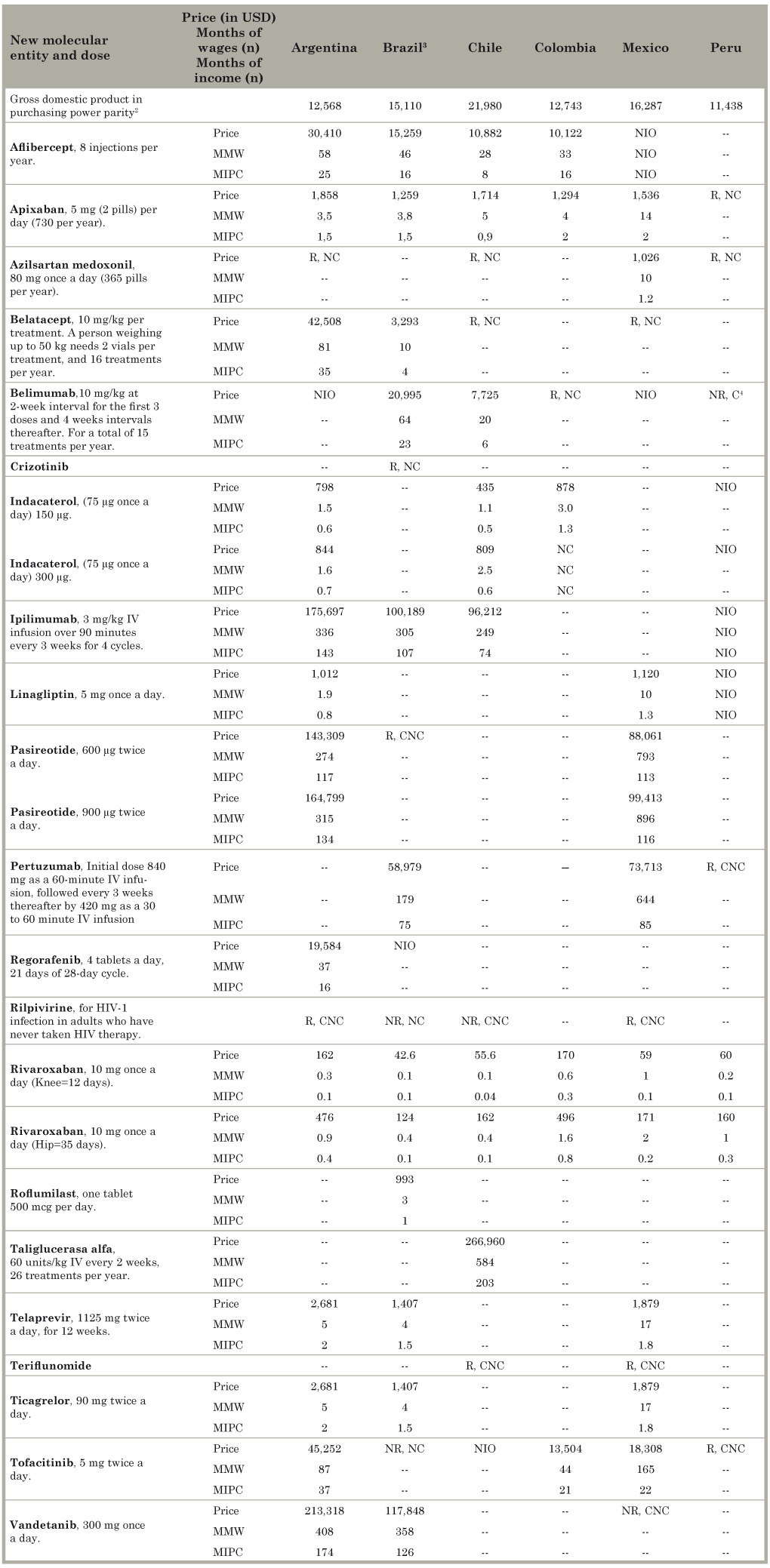
Source: Homedes and Ugalde.9 MMW = Monthly minimum wage; MIPC = Monthly income per capita; R = Registered: NR = Not registered; C = Commercialized; NC = Not commercialized; CNC = Commercialization not confirmed (it could not be confirmed whether the product was available in the country); NIO = No information obtained. 1Prices are for a complete course of treatment, or, in the case of chronic disease, for a year of treatment. All MNW and MIPC figures above 3 months have been rounded. For cells with no information (--), no clinical trials were conducted in the country or the product is not available. In Ecuador there was information only for indacaterol, and to simplify the table we have not included the information, but it is available from corresponding author. 2Information from the World Bank for the year 2014. All gross domestic product (GDP) data are reported in purchasing power parity in US dollars based on Atlas methodology used by the International Monetary Fund and the World Bank. The Argentina GDP per capita is based on data officially reported by the National Institute of Statistics and Censuses of Argentina that do not estimate the purchasing power parity. The International Monetary Fund has called on Argentina to adopt measures to address the quality of official GDP and consumer price index data. For more information please refer to the World Bank.(38) 3In Brazil, ANVISA publishes a list of maximum prices for the consumer that does not include taxes. The tax rate varies by state from 0-19%. We have included the maximum tax rate to the ANVISA prices. 4GlaxoSmithKline says it is available.
We could not find any relationship between prices and the GDP per capita or the minimum wages in these countries. In Brazil aflibercept costs US$15,259 per course of treatment; in Argentina, which has a slightly lower GDP per capita than Brazil, the course of treatment of the same medication is US$30,410. Brazilians pay 46 times the monthly minimum wage and Argentines 58, making the treatment unaffordable in both countries but considerably more in Argentina. If we compare aflibercept in Colombia and Chile - the latter has a considerably higher GDP per capita than the former - the drug is slightly cheaper in Colombia, but patients have to pay twice as many monthly minimum wages as Chileans. In the case of indacaterol, in Colombia the cost of the drug is twice that in Chile, and the same is true for rivaroxaban. Many other significant price differences can be found in Table 6.
Table 7 indicates the number of monthly minimum wages (MMW) necessary to purchase one course of treatment or a year of treatment for chronic conditions. In all countries, the cost of all products except one was greater than one MMW. Five medications required more than one and up to four MMW; and six NMEs (39%) cost between 100 and 896 MMW. Using the monthly income per capita (MIPC), Argentineans and Chileans could purchase three products with less than one MIPC, and Brazilians one (see Table 8). One product (rivaroxaban) costs less than one MIPC in all countries, but four products (two for cancer) cost more than 100 MIPC.
Table 7 Number of 2014 monthly minimum wages (MMW) needed to purchase one course of treatment or one year of treatment for chronic conditions of the new molecular entities tested in Latin America in 2011 and 2012.
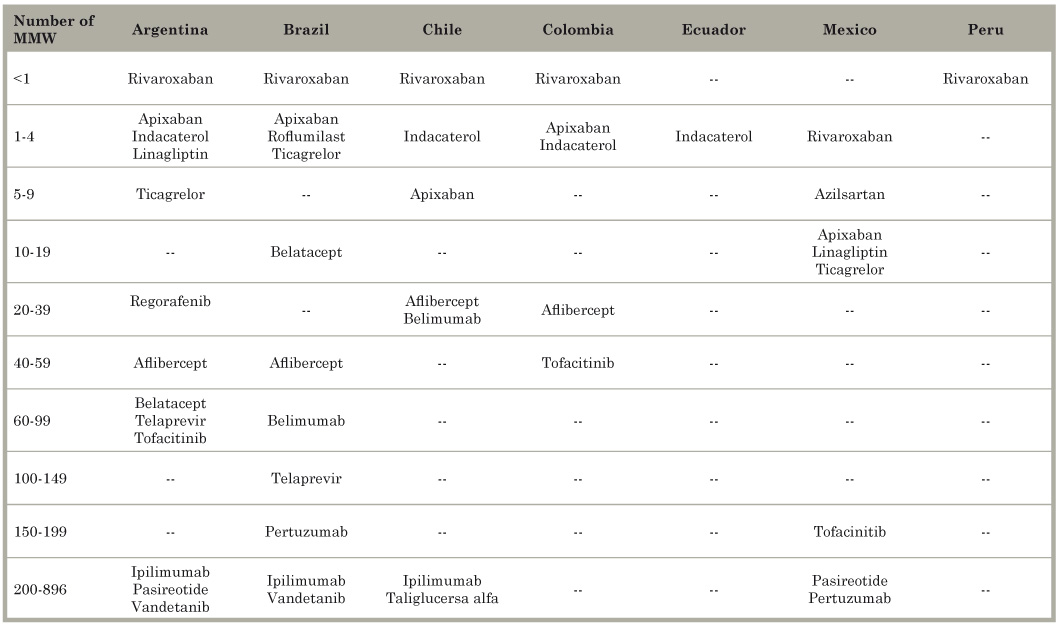
Source: Own elaboration using information from Homedes and Ugalde.9
Table 8 Number of 2013 monthly income per capita (MIPC) needed to purchase new molecular entities that were tested in Latin American countries in 2011 and 2012.
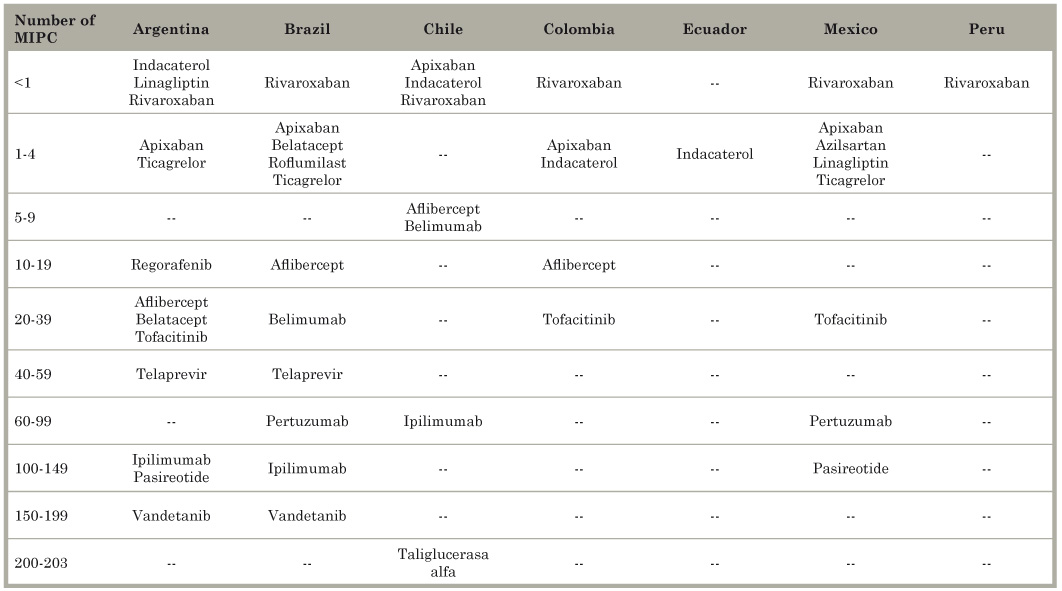
Source: Own elaboration using information from Homedes and Ugalde.9
The results using monthly household net adjusted disposable income and monthly household financial wealth were very similar to MIPC findings. Comparisons between MMW and MIPC measurements in the seven Latin American countries housing over 85% of all clinical trials conducted in the region are shown in Table 6. The hypothesis that the large majority of the population cannot afford the drugs tested in their countries is categorically confirmed.
Therapeutic advantage of NMEs
Prescrire and Health Research Group evaluated 26 of the 33 NMEs included in this study, and determined that 21 of the 26 (80%) offered no therapeutic advantage over existing treatments and had significant side effects, advising against the use of ten of them (Table 9). According to these sources and the independent bulletins cited by Prescrire, the remaining five products (crizotinib, enzalutamide, ipilimumab, pasireotide, and telaprevir) could offer some advantage to a subset of patients, but the risk-benefit ratio remained uncertain. Only three of these five products were available in the countries where tested.
Table 9 Summary of the assessments carried out by independent drug bulletins regarding new molecular entities tested in Latin American countries and approved by the US Food and Drug Administration in 2011 and 2012.

Source: Homedes and Ugalde;9 Prescrire;39,40,41 Public Citizen;42 Prescrire;43,44,45,46,47,48,49,50 Public Citizen;51 Prescrire;52,53 Public Citizen;54 Prescrire.55,56,57,58,59,60,61,62,63,64,65,66,67) Note: Neither Prescrire nor the Health Research Group of Public Citizen had evaluated azilsartan medoxonil, taliglucerasa alfa, tofacitinib, cabozantinib, ezogabine, lucinactant or tbo-filgastrim.
Eight of the 33 products included in our study (25%) were included in Fojo et al.'s evaluation.37 Only one of them (enzalutamide) increased overall survival significantly (by 4.8 months) in patients with castration-refractory prostate cancer; four increased the progression-free survival period (vandetanib, pertuzumab, carbozantinib, crizotinib), two did not fulfill the American Society of Clinical Oncology (ASCO) criteria to determine clinical relevance (ziv-aflibercept, regorafenib), and the authors were uncertain about ipilimumab (Table 10).
Table 10 Efficacy of oncological treatments approved by the US Food and Drug Administration in 2011 and 2012 and tested in countries of Latin America.

Source: Own elaboration using information from Fojo et al.37 *Estimated. NIO = No information obtained. ASCO = American Society of Clinical Oncology.
In contrast with the methodology used by the independent drug bulletins mentioned above, Fojo et al.37 assessed the value of each NME without comparing it with other treatment options. Two NMEs that qualified as useful in their publication (vandetanib and pertuzumab) were questioned by the independent drug bulletins. Vandetanib was considered more dangerous than beneficial, and the benefit-risk ratio of pertuzumab was judged to be insufficiently known. While Australian Prescriber and Medical Letter thought that it appeared to increase survival without worsening the condition of human epidermal growth factor receptor 2 (HER-2) positive women with metastasis of breast cancer, Medical Letter thought that the effect on overall survival had not been determined and others considered that it increased the side-effects, the benefits were uncertain, and there was insufficient information to recommend its commercialization. At a price of more than US$50,000 (pertuzumab) and US$100,000 (vandetanib) per treatment in Brazil and US$200,000 in Argentina (vandetanib), these NMEs are not affordable.
DISCUSSION
The pharmaceutical industry claims that the implementation of clinical trials in Latin America strengthens research capacity in biomedical science and transfers highly desirable foreign exchange to the region.68 However, there are also negative consequences for the financial sustainability and safety of the pharmaceutical market and for the wellbeing of research participants.
Financial sustainability and safety of pharmaceutical market
There seems to be little communication between the research and development units of the pharmaceutical companies and those responsible for marketing the final products. Clinical trials are outsourced when the countries meet the conditions of the sponsor or of the contract research organizations managing the trial, such as expedited approval of protocols, large urban centers with quality hospitals, and an abundance of easy-to-recruit patients.4 On the other hand, the registration and marketing of new products are business decisions based on a country's regulatory conditions, the presence of a business affiliate or partner, the willingness of the public health care system to include a product in its formulary, the number of patients who can afford the treatment, and estimates of drug profitability for the company.
Latin American regulatory agencies do not deny market authorizations when the product has already been commercialized in high surveillance countries. The fact that the FDA and the European Medicines Agency69,70,71,72,73,74,75,76,77 tend to approve NMEs without ensuring that they are more effective and/or safer than existing treatments78 results in the presence of the same products in Latin America, regardless of how they affect the financial sustainability and safety of the corresponding markets. Moreover, Brazil's ethical guidelines require that all drugs tested in the country be registered when found to be safe and effective. Resolution 446 of 2012 of the regulatory agency (ANVISA) reads: "When developing new drugs, if safety and effectiveness is proven, its registration is obligatory in Brazil."79 However, it appears that ANVISA is not enforcing the regulation.
As shown, the price of marketed products was unaffordable for the large majority of the Latin American population. By definition, unaffordability implies that a person or household cannot redistribute resources to obtain the product. This is also becoming a problem for high-income countries. Physicians and health authorities in the United States and the United Kingdom are reluctant to prescribe and pay expensive medications offering little advantage over existing cheaper alternatives with better-known safety and efficacy profiles.80,81,82,83)
Medicine prices are unrelated to product development costs84,85 and our data indicate that pharmaceutical manufacturers are not abiding by WHO's recommendations to set prices according to countries' wealth or tiered pricing.86 We have not identified any articles discussing the large price differentials among neighboring Latin American countries. Why the same drug represents a higher financial burden for Argentines than Brazilians, Mexicans or other Latin Americans needs to be better understood. Exploring these differences will require a detailed analysis of all the drug pricing components in each country, including the manufacturer's sale price, transportation costs, importation tariffs, the margin of benefits for distributors and dispensers, sales taxes and others. This analysis might demonstrate that governments can adopt policies to increase the affordability of NMEs, but it is also likely to show large differences in the manufacturer's sale price, which tends to be based on the industries' assessment of what each country is willing to pay. More collaboration among pharmaceutical policy makers and procurement experts across Latin American countries could lead to improved pricing structures for the region.
The judicialization process described in the introduction may encourage the pharmaceutical industry to maintain high prices with the aggravated consequence of exposing patients to NMEs that, according to independent drug bulletins, should not be used. Latin American patients and patients' groups-often financially supported by the innovative pharmaceutical companies-sue the governments87,88,89,90,91 to gain access to the newest treatments, including those that have not been included in national formularies.92,93,94 Judges tend to base their decisions on individual needs instead of societal priorities; if this trend continues, health care systems will be severely strained and many could go bankrupt.95,96,97,98,99,100 Without denying the difficulties, it would be useful to assess the NME affordability threshold for the public health sector. Our hypothesis is that most of the NMEs discussed in this paper are not affordable for the ministries of health or the central governments.
The wellbeing of research participants
If the 26 products included in our study for which we obtained efficacy and safety information from independent sources are a sample representative of the NMEs tested in Latin America, we need to conclude that the current research and development models of NMEs are faulty. Two questions need to be pondered: 1) Did ethics committees appropriately protect human research participants, considering that to consciously expose research participants to unnecessary risks would translate into a violation of the ethical principle of beneficence?; and 2) Did the clinical trials have to be conducted in the vulnerable populations of Latin America who, given current prices, are unlikely to benefit from the discoveries?
The fact that a large number of NMEs failed to add therapeutic value to existing treatments leads us to conclude that the patients enrolled in the experimental arm of the clinical trial were in fact worst off than if they had not participated in the study and received the standard treatment. Similarly, the patients included in the control arm, except those who received the best available treatment, were also incurring unnecessary risks, especially if they were enrolled in a placebo-controlled or non-inferiority trial. Some of these risks could potentially have been avoided if research sponsors, regulatory agencies and ethics committees had conducted a more in-depth analysis of the results of pre-clinical studies and of earlier phases of the clinical trials101,102 and if the NMEs were always tested against the best available treatment.
According to article 20 of the Helsinki Declaration, vulnerable populations should not be enrolled in clinical trials when the products can be tested in non-vulnerable populations.103 Latin American trial subjects tend to be of low socioeconomic status, are often medically illiterate, and according to some authors should be considered vulnerable.104,105,106 All products included in this study, except bedaquiline, which is used in the treatment of multidrug-resistant tuberculosis, could have been tested in non-vulnerable populations.
Another ill consequence of conducting clinical trials sponsored by industry that needs to be further explored is how they displace research that would be more relevant for the region. The rewards offered to principal investigators, including but not limited to payments, lure some of them away from developing other products needed in the region, such as treatments for dengue, malaria and leishmaniasis or from the increasingly urgent need of developing biological generics that could save lives and dollars.107,108
Complying with ethical and regulatory premises
In a broken research and development system with many unnecessary clinical trials that do not result in better therapies and render the NMEs unaffordable for the private and public sectors, some ethical, judicial and regulatory conditions designed to protect the population have ironically had the opposite effect. Allowing the exploitation of vulnerable populations also has negative consequences for the sustainability and safety of the pharmaceutical markets.
Most ethical guidelines such as those of Council for International Organizations of Medical Sciences (CIOMS),6 the Universal Declaration on Bioethics and Human Rights,109 or the Declaration of Helsinki103 assert that post trial access to treatment should be ensured. According to Guideline 10 of the CIOMS, which refers to "Research in populations and communities with limited resources":
Before undertaking research in a population or community with limited resources, the sponsor and the investigator must make every effort to ensure that:
the research is responsive to the health needs and the priorities of the population or community in which it is to be carried out; and
any intervention or product developed, or knowledge generated, will be made reasonably available for the benefit of that population or community. (6
The document also includes commentaries regarding the guidelines. Some of the most crucial aspects of the contents of the commentary on Guideline 10 are as follows:
[...] This is applicable especially to research conducted in countries where governments lack the resources to make such products or benefits widely available. Even when a product to be tested in a particular country is much cheaper than the standard treatment in some other countries, the government or individuals in that country may still be unable to afford it. If the knowledge gained from the research in such a country is used primarily for the benefit of populations that can afford the tested product, the research may rightly be characterized as exploitative and, therefore, unethical. [...] The negotiation should cover the health-care infrastructure required for safe and rational use of the intervention, the likelihood of authorization for distribution, and decisions regarding payments, royalties, subsidies, technology and intellectual property, as well as distribution costs, when this economic information is not proprietary. [...] In general, if there is good reason to believe that a product developed or knowledge generated by research is unlikely to be reasonably available to, or applied to the benefit of, the population of a proposed host country or community after the conclusion of the research, it is unethical to conduct the research in that country or community.6 [italics added]
Similarly, article 15 of the Universal Declaration on Bioethics and Human Rights, regarding the "Sharing of benefits" of clinical trials, states that the "Benefits resulting from any scientific research and its applications should be shared with society as a whole and within the international community, in particular with developing countries."109 Principle 22 of the Declaration of Helsinki highlights that "in clinical trials, the protocol must also describe appropriate arrangements for post-trial provisions."103
Nevertheless, in contrast with the commentary of Guideline 10 of the CIOMS, neither the sponsors of the clinical trials, or the regulatory agencies, or any of the bodies that approved ethical declarations regarding clinical trials have suggested pre-trial mechanisms to ensure that NMEs will be made available at affordable prices. Without such mechanisms, Latin American Research Ethics Committees and regulatory agencies approve the implementation of many clinical trials that should not be authorized, facilitating the violation of the justice principle and the exploitation of trial participants.
In January 2014, a new Consensus Framework for Ethical Collaboration between Patients' Organizations, Health Care Professionals and the Pharmaceutical Industry was published, the third point of which states:
Clinical Research. Continuing to advocate and support the principle that all human subject research must have legitimate scientific purpose, aims to improve health outcomes, and be ethically conducted...110
If the industry wants to be congruent with its own consensus framework, and with universally accepted ethical principles6,103,109 it will have to:
Include in clinical trial protocols the projected price of the potential NME so that the regulatory agencies and research ethics committees can take into consideration the affordability of the NME before authorizing the research.
Establish a mechanism to guarantee the registration and availability of the new NMEs that prove to be safe and effective, in coordination with the regulatory agencies of the countries where the NMEs are tested.
Reconsider their research and commercial strategies to ensure that NMEs add therapeutic value to the existing therapeutic arsenal at an affordable price.
Discontinue sponsorship non-inferiority clinical trials, unless they are strictly necessary.
Until these conditions are met, it might be better to forego compliance with the requirement that NMEs be made available in the countries where tested. Similarly, given the proclivity of reputable regulatory agencies to approve NMEs that according to independent pharmacology experts should not be approved, Latin American regulatory agencies might want to consider delinking their market authorization decisions from those made by the regulatory agencies of high sanitary surveillance countries and instead use the information provided independent drug bulletins. Given the dearth of true innovation, delaying the approval of NMEs until independent assessments are available will not be detrimental for the Latin American residents. Exceptions could be made for true breakthrough NMEs.
Limitations
Some FDA reviews of NMEs did not specify which of the clinical trials were pivotal. Even though we also gathered information from trial sponsors, we may have included trials that technically might not be considered pivotal. We were unable to avoid the limitations for determining the affordability thresholds that other researchers had previously encountered. What humans are willing to sacrifice and the risks they are ready to confront cannot be easily defined by others; it is a personal decision that is heavily influenced by personal values and culture.
To determine the price of drugs continues to be complex, and currently there is no gold-standard methodology. At this point, despite their shortcomings, the country observatories are probably the best and most reliable source of information, which tends to be based on variations of the WHO-HAI methodology. Currency variations add to the complexity of reporting pricing information across countries. We priced the drugs in September of 2014, but the data used to determine the MIPC is from 2013. During this time differential, some currencies depreciated while others appreciated.
Moreover, in the Latin American countries included in this study, income is very poorly distributed (Table 3). If we were to remove the highest two income deciles, the income per capita for the rest of the population would be drastically reduced, in most countries halved, and therefore the affordability threshold would have to be lowered.
The information on registration and commercialization status of NMEs may contain inaccuracies. Using triangulation methods we identified and corrected some errors, but others may not have been detected.
CONCLUSIONS
This is the first study that questions the health benefits of the clinical trials implemented in Latin America not only for trial subjects but for the health systems and residents of those countries.
Many of the products tested are unavailable and/or unaffordable to the large majority of Latin Americans, and only a few proved to be more efficacious for a select group of patients but with significant side effects. The balance between the benefits and the negative economic and health consequences of conducting clinical trials in Latin America lead us to suggest that the number of trials should be drastically reduced.
The current drug research and development model is very questionable, and perhaps until the model is improved clinical trials should only be carried out in the countries where the corporations are headquartered. We conclude that:
There is an urgent need to determine the public sector affordability of NMEs. Since the pricing of NMEs is unrelated to research and development and production costs, the industry has wide pricing margins.
The risk of having to register and commercialize very expensive products, endangering the budget of the ministries of health without improving the health of the patients is a reason to reduce the implementation of clinical trials in Latin America.
There is a need to strengthen ethics committees so that in their evaluation of clinical trials they can pay significant attention to the affordability and pertinence of the NME.
The products included in this study did not respond to the most pressing therapeutic needs of the region, and they may divert scientific resources from addressing issues of higher relevance. While governments more than welcome the investments that accompany foreign trials, it is important to document their opportunity costs.
The Latin American regulatory agencies should use the information provided by independent pharmaceutical experts and bulletins when they evaluate the request to market NMEs. Exceptions could be made for the few true breakthrough NMEs.
It is important to uncover the reasons for drug price differentials across the different countries of the region.
ACKNOWLEDGEMENTS
We are indebted to all employees of the regulatory agencies and pharmaceutical industries who facilitated access to the information reported in this article. Our task could not have been completed without the assistance of the experts who helped us confirm the registration of status and prices of some NMEs: Martín Cañas (Argentina), Corina Bontempo Duca de Freitas (Brazil), Andrea Carolina Reyes Rojas and Oscar Andía (Colombia), Marvín Gómez (Costa Rica), Belén Mena (Ecuador), Rogelio Fernández (Mexico), Alarico Rodríguez (Uruguay). We would like to thank Iain Chalmers, Donald Light, Pauline Rosenau and Bruno Schlemper Junior for their comments to earlier versions of this document
REFERENCES
1. Express Scripts. Drug trend report [Internet]. 2010 [citado 10 may 2015]. Disponible en: http://goo.gl/kOSQvQ. [ Links ]
2. Glickman SW, McHutchison JG, Peterson ED, Cairns CB, Harrigton RA, Califf RM, Schulman K. Ethical and scientific implications of the globalization of clinical research. New England Journal of Medicine. 2009;360(8):816-823. [ Links ]
3. Wenner DM. The social value of knowledge and international clinical research. Developing World Bioethics. 2015;15(2):76-84. [ Links ]
4. Homedes N, Ugalde A. Globalization and clinical research in Latin America. In: Homedes N, Ugalde A, (eds). Clinical trials in Latin America: where ethics and business clash. Dordrecht: Springer; 2014. p 55-78. [ Links ]
5. Durivage HJ, Bridges KD. Clinical trial metrics: Protocol performance and resource utilization from 14 cancer centers. Journal of Clinical Oncology. 2009;27(Suppl 15):6557. [ Links ]
6. Council for International Organizations of Medical Sciences. International Ethical Guidelines for Biomedical Research involving human subjects. Geneva: CIOMS; 2002. [ Links ]
7. Vargas-Pélaez CM, Mattozo Rover MR, Leite SN, Rossi Buenaventura F, Rocha Farias M. Right to health, essential medicines, and lawsuits for access to medicines - A scoping study. Social Science and Medicine 2014;121:48-55. [ Links ]
8. Homedes N, Ugalde A. Availability and affordability of new medicines in Latin American countries where pivotal clinical trials were conducted. Bulletin of the World Health Organization. 2015;93(10):674-683. [ Links ]
9. Homedes N, Ugalde A. Health and ethical consequences of outsourcing pivotal clinical trials to Latin America: A cross-sectional, descriptive study. PLoS One. 2016;11(6):e0157756. [ Links ]
10. Homedes N, Ugalde A. Are private interests clouding the peer-review process of the WHO Bulletin?: A case study. Accountability in Research. 2016;23(5):309-317. [ Links ]
11. Food and Drug Administration. 2011 novel drug [Internet]. FDA; 2012 [citado 10 may 2016]. Disponible en: http://goo.gl/dPZ0tI. [ Links ]
12. Food and Drug Administration. Novel drug approvals for 2012 [Internet]. 2016 [citado 10 may 2016]. Disponible en: http://goo.gl/OCd2Fk. [ Links ]
13. Drugs@FDA [Internet]. Silver Spring: Food and Drug Administration; 2015 [citado 15 may 2015]. Disponible en: http://goo.gl/aM8sbK. [ Links ]
14. Administración Nacional de Medicamentos, Alimentos y Tecnología Médica. Vademecum Nacional de Medicamentos [Internet]. Buenos Aires: ANMAT; 2014 [citado 27 may 2015]. Disponible en: http://goo.gl/QIF6v4. [ Links ]
15. Administración Nacional de Medicamentos, Alimentos y Tecnología Médica. Listado oficial de medicamentos comercializados [Internet]. Buenos Aires: ANMAT; 2014. Disponible en: http://goo.gl/Gnrx0E. [ Links ]
16. Agência Nacional de Vigilância Sanitária. Listas de Preços de Medicamentos [Internet]. Brasília: ANVISA; 2015 [citado 15 may 2015]. Disponible en: http://tinyurl.com/nwe9go4. [ Links ]
17. Agência Nacional de Vigilância Sanitária. Medicamentos Analisados [Internet]. Brasília: ANVISA; 2015 [citado 10 may 2015]. Disponible en: http://goo.gl/M2tnEf. [ Links ]
18. Instituto Nacional de Vigilancia de Medicamentos y Alimentos. Consulta datos de productos [Internet]. Bogotá: INVIMA; 2014 [citado 15 may 2015]. Disponible en: http://goo.gl/VhICMI. [ Links ]
19. Instituto Nacional de Vigilancia de Medicamentos y Alimentos. Listado de entidades químicas con información no divulgada protegida según el Decreto 2085 de 2002 [Internet]. Bogotá: INVIMA; 2014 [citado 15 may 2015]. Disponible en: http://goo.gl/kY1vDu. [ Links ]
20. Ministerio de Salud y Protección Social. Precios de Medicamentos - Circular 2 de 2012 Excel [Internet]. Bogotá: MINSALUD; 2013 [citado 15 may 2015]. Disponible en: http://goo.gl/WSaQ4C. [ Links ]
21. Ministerio de Salud. Sistema de consulta de productos registrados [Internet]. Santiago: Instituto de Salud Pública de Chile; 2015 [citado 15 may 2015]. Disponible en: http://goo.gl/N0tVVJ. [ Links ]
22. Precios de Remedios [Internet]. Chile [citado 18 ago 2014] Disponible en: https://goo.gl/Z9Bt4A. [ Links ]
23. Ministerio de Economía. Precios registrados de medicamentos con patente vigente [Internet]. México DF: Ministerio de Economía; 2014 [citado 15 may 2016]. Disponible en: http://goo.gl/aiuZnu. [ Links ]
24. Precios de Remedios [Internet]. México [citado 8 sep 2014]. Disponible en: https://goo.gl/rChQM1. [ Links ]
25. Dirección General de Medicamentos, Insumos y Drogas. Catálogo de Productos Farmacéuticos [Internet]. Lima: Ministerio de Salud; 2015 [citado 15 may 2015]. Disponible en: http://goo.gl/PIIS7W. [ Links ]
26. Dirección General de Medicamentos, Insumos y Drogas. Módulo de Consulta de Precios [Internet]. Lima: Ministerio de Salud; 2015 [citado 15 may 2015]. Disponible en: http://goo.gl/Z4FCts. [ Links ]
27. Panamerican Health Organization. Health systems and social protection in health. In: PAHO. Health in the Americas. Washington DC: PAHO; 2012. p. 205-251. [ Links ]
28. The World Bank. GDP per capita (current US$) [Internet]. c2016 [citado 10 may 2015]. Disponible en: http://goo.gl/MHMhCa. [ Links ]
29. Organisation for Economic Co-operation and Development. OECD Better Life Index [Internet]. c2016 [citado 10 may 2015]. Disponible en: http://goo.gl/GRl9QS. [ Links ]
30. Organisation for Economic Co-operation and Development. OECD Better Life Index: Income [Internet]. c2016 [citado 10 may 2015]. Disponible en: http://goo.gl/bVaDzi. [ Links ]
31. Central Intelligence Agency. The world factbook [Internet]. 2015 [citado 10 may 2015]. Disponible en: http://goo.gl/lYZWO2. [ Links ]
32. The World Bank. GINI index (World Bank estimate) [Internet]. c2016 [citado 10 may 2015]. Disponible en: http://goo.gl/uAt4tj. [ Links ]
33. The World Bank. PovcalNet: an online analysis tool for global poverty monitoring [Internet]. c2016 [citado 10 may 2015]. Disponible en: http://goo.gl/XTNVMH. [ Links ]
34. Niëns LM, Brouwer WBF. Measuring the affordability of medicines: importance and challenges. Health Policy. 2013;112:45-52. [ Links ]
35. Niëns LM, Van de Poel E, Ewen M, Laing R, Brour WBF. Practical measurements of affordability: an application to medicines. Bulletin of the World Health Organization. 2012;90:219-227. [ Links ]
36. O'Donnell O, van Doorslaer E, Wagstaff A, Lindelow M. Analyzing health equity using household survey data: a guide to techniques and their implementation [Internet]. Washington DC: World Bank; 2008 [citado 15 may 2015]. Disponible en: http://goo.gl/SSyar2. [ Links ]
37. Fojo T, Mailankody S, Lo A. Unintended consequences of expensive cancer therapeutics -the pursuit of marginal indications and a me-to mentality that stifles innovation and creativity. JAMA Otolaryngology-Head & Neck Surgery. 2014;140(12):1225-1236. [ Links ]
38. The World Bank. GDP per capita, PPP (constant 2011 international $) [Internet]. c2016 [citado 10 may 2015]. Disponible en: http://goo.gl/AenKjw. [ Links ]
39. Prescrire. Aclinidium: encore an atropinique inhale, peut être des effets cardivasculaires. Revue Prescrire. 2013;33(259):654-655. [ Links ]
40. Prescrire. Aflibercept: Dégénérescence maculaire liée al'âge: un autre anti-VEGF, sans plus. Revue Prescrire. 2013;33(353):170-173. [ Links ]
41. Prescrire. Apixaban et fibrillation auriculaire: Pas de preuves solides d'un progrès. Revue Prescrire. 2013;33(361):808-812. [ Links ]
42. Public Citizen. Emerging risks with new stroke prevention drugs. Worst Pills, Best Pills. Newsletter Articles; April 2013. [ Links ]
43. Prescrire. Axitinib: No better than sorafenib in kidney cancer. Revue Prescrire. 2013;22(143): 262-263. [ Links ]
44. Public Citizen. Harming Tuberculosis Patients Instead of Helping Them? WorstPills, Best Pills. Newsletter Articles; February 2013. [ Links ]
45. Prescrire. Bélatacept: Greffes de rein, plus de risque qu'avec la ciclosporine. Revue Prescrire. 2012;32(341):179-182. [ Links ]
46. Prescrire. Bélimumab: Des risques d'immuno-dépression, sans efficacité tangible établie. Revue Prescrire. 2013;33(354):258-261. [ Links ]
47. Prescrire. Bosutinib: Leucémie myéloïde chronique en situation d'échec: toxicité importante. Revue Prescrire. 2014;34(363):10. [ Links ]
48. Prescrire. Crizotinib: Effets indésirables graves avérés mais efficacité mal cernée dans les cancers bronchiques. Revue Prescrire. 2013;33 (357):498-501. [ Links ]
49. Prescrire. Stribild: deux nouvelles substances mais pas de progress. Revue Prescrire. 2013;33(256):408-411. [ Links ]
50. Prescrire. Enzalutamide: une alternative à l'abiratérone, après échec du docétaxel. Revue Prescrire. 2014;34(367):330-334. [ Links ]
51. Public Citizen. Letter to FDA on Indacaterol Maleate (ArcaptaNeohaler) [Internet]. 16 mar 2011 [citado 15 may 2015]. Disponible en: http://goo.gl/Taobep. [ Links ]
52. Prescrire. Ipilimumab: Immunostimulant à mieux évaluer dans le mélanome. Revue Prescrire. 2012;32(340):98-100. [ Links ]
53. Prescrire. Linagliptine: Non aux gliptines. Revue Prescrire. 2012;32(347):564-565. [ Links ]
54. Public Citizen. Diabetes drugs link to pancreas disease. Worst Pills, Best Pills. Newletter Articles, August 2013. [ Links ]
55. Prescrire. Pasiréotide: Faute de mieux. Revue Prescrire. 2013;33(356):415-416. [ Links ]
56. Prescrire. Pérampanel - Fycompa(r). Nième anticonvulsivant dans les épilepsies partielles, sans progrès. Revue Prescrire. 2014;34(365):171-172. [ Links ]
57. Prescrire. Pertuzumab: Un espoir à mieux évaluer pour certaines patientes atteintes de cancer du sein métastasé. Revue Prescrire. 2013;33(361):816-814. [ Links ]
58. Prescrire. Régorafénib: Cancer colorectal métastasé en échec: peut-être quelques semaines de survie en plus. Revue Prescrire. 2013,33(360):736-741. [ Links ]
59. Prescrire. Rilpivirine: En première ligne contre le HIV: l'éfavirenz est mieux connu. Revue Prescrire. 2012;32(345):494-497. [ Links ]
60. Prescrire. Anticoagulation après prothèse de hanche ou de genou: rivoraxaban pas mieux qu'énoxaparine. Revue Prescrire. 2009;29(314):926-927. [ Links ]
61. Prescrire. Roflumilast: Efficacité douteuse sur la BPCO, mais risques avérés. Revue Prescrire. 2012;32(343)328-333. [ Links ]
62. Prescrire. Telaprevir: Afterboceprevir, if necessary. Revue Prescrire. 2012;32(339):11-14. [ Links ]
63. Prescrire. Teriflunomide (Aubagio(r)): Sclérose en plaques: Seulement un métabolite du léflunomide. Revue Prescrire. 2014;34(373):808-812. [ Links ]
64. Prescrire. Le tériflunomide (Aubagio(r)) est commercialisé ou annoncé dans divers pays [Internet]. Prescrire; 2014 [citado 15 may 2015]. Disponible en: http://goo.gl/6JkDnp. [ Links ]
65. Prescrire. Ticagrelor: syndromes coronariens aigus: pas d'emballement. Revue Prescrire. 2011;31(333):488-493. [ Links ]
66. Prescrire. Vandétanib: Trop dangereux dans les cancers médullaires de la thyroïde. Revue Prescrire. 2012;32(342):256-259. [ Links ]
67. Prescrire. Aflibercept - Zaltrap(r): Cancer colorectal métastasé: au moins aussi mal toléré que le bévacizumab. Revue Prescrire. 2014;34(366):258. [ Links ]
68. Ugalde A, Homedes N. The regulatory framework and case studies from Argentina. In: Homedes N, Ugalde A, (eds). Clinical trials in Latin America: Where ethics and business clash. Dordrecht: Springer 2014. p. 79-114. [ Links ]
69. Naci H, Alexander W Carter AW, Mossialos E. Why drug development pipeline is not delivering better medicines. BMJ. 2015;351:h5542. [ Links ]
70. Goldacre B. Bad Pharma: How drug companies mislead doctors and harm patients. London: Fourth State; 2012. [ Links ]
71. Banzi R, Gerardi C, Bertele V, Garattini S. Approvals of drugs with uncertain benefit-risk profiles in Europe. European Journal of Internal Medicine. 2015;26(8):572-584. [ Links ]
72. Chalkidou K, Tunis S, Lopert R, Rochaix L, Sawicki PT, Nasser M, Xerri B. Comparative effectiveness research and evidence health policy: Experience from four countries. Milbank Quarterly. 2009;87(2):339-367. [ Links ]
73. Nicod E, Kanavos P. Commonalities and differences in HTA outcomes: A comparative analysis of five countries and implications for coverage decisions. Health Policy. 2012;108(2):167-177. [ Links ]
74. Prescrire. Évaluer le progrès thérapeutique: avec méthode, au service des patients. Revue Prescrire. 2015;35(382):565-569. [ Links ]
75. Garattini S, Chalmers I. Patients and the public deserve big changes in evaluation of drugs. BMJ. 2009;338:b1025. [ Links ]
76. Garattini S, Bertele V. Adjusting Europe's drug regulation to public health needs. Lancet. 2001;358(9275):64-67. [ Links ]
77. Light D, Lexchin J. The FDA's new clothes. BMJ. 2015;352:h4897. [ Links ]
78. Kim C, Prasad V. Strength of Validation for surrogate end points used in the US Food and Drug Administration's approval of oncology drugs. Mayo Clinic Proceedings. 2016;91(6):713-725. [ Links ]
79. Conselho Nacional de Saúde. Resolução Nº 466, de 12 de dezembro de 2012 [Internet]. 2012 [citado 10 may 2015]. Disponible en: http://goo.gl/z0qZEl. [ Links ]
80. Abboud C, Berman E, Cohen A, Cortes J, DeAngelo D, Deininger M, et al. The price of drugs for chronic myeloid leukemia (CML) is a reflection of the unsustainable prices of cancer drugs: from the perspective of a large group of CML experts. Blood. 2013; 121(22):4439-4442. [ Links ]
81. Ward A, Neville S. Drug cost watchdog chief calls for honesty with public. Financial Times [Internet]. 22 ago 2014 [citado 10 may 2015]. Disponible en: http://goo.gl/jOklBr. [ Links ]
82. Werth B. A tale of two drugs. MIT Technology Review [Internet]. 22 oct 2013 [citado 10 may 2015]. Disponible en: http://tinyurl.com/lnhmkfe. [ Links ]
83. Steenhuysen J. FDA cancer chief says 'escalating' drug prices can't continue. Reuters [Internet]. Health News; 1 jun 2014 [citado 15 may 2015]. Disponible en: http://tinyurl.com/hafwbd9. [ Links ]
84. Evaluate Pharma. Budget-busters: the shift to high-priced innovator drugs in the USA [Internet]. September 2014 [citado 15 may 2015]. Disponible en: http://tinyurl.com/mdgvv27. [ Links ]
85. Light DW, Warburton R. Demythologizing the high costs of pharmaceutical research. BioSocieties. 2011;6(1):34-50. [ Links ]
86. Yadav P. Differential pricing for pharmaceuticals [Internet]. WHO; 2010 [citado 15 may 2015]. Disponible en: http://tinyurl.com/h3hfpuw. [ Links ]
87. Sant'Ana JMB, Pepe VLE, Osório-de-Castro CGS, Ventura M. Essential drugs and pharmaceutical care: reflection on the access to drugs through lawsuits in Brazil. Pan American Journal of Public Health. 2011;29(2):138-144. [ Links ]
88. Vieira FS, Lopes LC, Barberato-Filho S, Marques DC, Pepe VLE. Pharmaceutical services and judicial decisions: proposals to improve access and rational use of medicines. Revista de Administração em Saúde. 2010;12(47):79-86. [ Links ]
89. Chieffi AL, Barata RCB. Legal suits: pharmaceutical industry strategies to introduce new drugs in the Brazilian public health care system. Revista de Saude Pública. 2010;44(3):421-429. [ Links ]
90. Pepe VLE, de-Aragao-Figueiredo T, Simas L, Osorio-de-Castro CGS, Ventura M. Health litigation and new challenges in the management of pharmaceutical services. Ciência & Saúde Colectiva. 2010;15(5):2405-2414. [ Links ]
91. Biehl J, Amon JJ, Socal MP, Petryna A. Between the court and the clinic: lawsuits for medicines and the right to health in Brazil. Health and Human Rights. 2012;14(1):36-52. [ Links ]
92. Andrade EIG, Machado CD, Faleiros DR, Szuster DAC, Guerra-Jr AA, Silva GD, et al. The judicialization of healthcare and the pharmaceutical care national policy in Brazil: the clinic management and the medicalization of the justice. Revista Médica de Minas Gerais. 2008;18(Suppl 4):S46-S50. [ Links ]
93. Cubillos L, Escobar ML, Pavlovic S, Iunes R. Universal health coverage and litigation in Latin America. Journal of Health Organisation and Management. 2012;26(3):390-406. [ Links ]
94. Reveiz L, Chapman E, Torres R, Fitzgerald JF, Mendoza A, Bolis M, Salgado O. Right to health litigation in three Latin American countries: a systematic literature review. Revista Panamericana de Salud Pública. 2013;33(3):213-222. [ Links ]
95. Diniz D, Medeiros M, Schwartz IVD. Consequences of the judicialization of health policies: the cost of medicines and mucopolysaccharidosis. Cadernos de Saúde Pública. 2012;28(3):479-489. [ Links ]
96. Tanaka OY. Juridical process of drug prescription in the Brazilian public health system or the challenge to ensuring the constitutional right to drugs access. Revista de Direito Sanitário. 2008;9(1):139-143. [ Links ]
97. Norheim OF, Wilson BM. Health rights litigation and access to medicines: priority classification of successful cases from Costa Rica's Constitutional Chamber of the Supreme Court. Health and Human Rights. 2014;16(2):47-61. [ Links ]
98. Hogerzeil HV, Samson M, Vidal Casanovas J, Rahmani-Ocora. Is access to essential medicines as part of the fulfillment of the right to health enforceable through the courts? Lancet. 2006;368(9532):305-311. [ Links ]
99. Yamin AE, Parra-Vera O. Judicial protection of the right to health in Colombia: from social demands to individual claims to public debates. Hastings International and Comparative Law Review. 2010;33(2):101-129. [ Links ]
100. Bergallo P. Courts and social change: lessons from the struggle to universalize access to HIV/AIDS treatment in Argentina. Texas Law Review. 2011;89(7):1611-1614. [ Links ]
101. Paul SM, Mytelka DS, Dunwiddie CT, Persinger CC, Munos BH, Lindborg SR, Schacht A. How to improve R&D productivity: the pharmaceutical industry's grand challenge. Nature Reviews Drug Discovery. 2010;9:203-2014. [ Links ]
102. Wokasch M. What really drives drug development inefficiencies-Part 1. Multi Briefs: Exclusive [Internet]. 5 may 2014 [citado 15 may 2015]. Disponible en: http://tinyurl.com/hvy5mn8. [ Links ]
103. World Medical Association. World Medical Association Declaration of Helsinki: Ethical Principles for Medical Research Involving Humans. JAMA 2013;310(20):2191-2194. [ Links ]
104. Okpechi IG, Swanepoel CR, Venter F. Access to medications and conducting clinical trials in LMICs. Nature Reviews Nephrology. 2015;11(3):189-194. [ Links ]
105. Pourrieux C. Ética de la investigación frente a intereses empresariales en la industria farmacéutica: Un caso en Argentina. Revista Redbioética UNESCO. 2014;5(10):94-99. [ Links ]
106. Verástegui EL. Consenting of the vulnerable: the informed consent procedure in advanced cancer patients in Mexico. BMC Medical Ethics. 2006;7:13. [ Links ]
107. ABIA, Alianza LAC, AIDAN, CIMUM, et at. Es el momento de actuar: aseguren el acceso a productos bio-terapéuticos asequibles. Declaración de la Sociedad Civil para pre-ICDRA e ICDRA. Boletín Fármacos. 2014;117(4):81-82. [ Links ]
108. Colombia. Carta abierta al Señor Presidente de la República de Colombia Doctor Juan Manuel Santos Calderón, 5 de septiembre de 2014. Boletín Fármacos. 2014;117(4):91-92. [ Links ]
109. UNESCO. Universal Declaration on Bioethics and Human Rights [Internet]. 19 oct 2005 [citado 15 may 2015]. Disponible en: http://tinyurl.com/39a6c9n. [ Links ]
110. International Alliance of Patients' Organizations, International Council of Nurses, IFPMA, International Pharmaceutical Federation, World Medical Association. Consensus framework for ethical collaboration between patients' organisations, healthcare professionals and the pharmaceutical industry [Internet]. 2014 [citado 15 may 2015]. Disponible en: http://tinyurl.com/htsq5nb. [ Links ]
ENDNOTES
[a] Pivotal clinical trials are those included in the applications for market authorization or New Drug Approval (NDA) documents that are submitted to the regulatory agencies.
[b] This article is based on the original version of an article that was divided in two and published by the same authors in the Bulletin of the World Health Organization8 and in PLoS One.9 The reasons for the division of the original article are explained in Accountability in Research. Policies and Quality Assurance.10
Received: June 07, 2016; Accepted: August 15, 2016











 text in
text in 




