Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Acta Odontológica Latinoamericana
versão On-line ISSN 1852-4834
Acta odontol. latinoam. vol.23 no.3 Buenos Aires dez. 2010
ARTÍCULOS ORIGINALES
Physicochemical salivary properties, Lactobacillus, Mutans Streptococci counts and early childhood caries in preschool children of Colombia
María C. Martínez-Pabón1, Blanca S. Ramírez-Puerta2, Gloria M. Escobar-Paucar2, Ángela M. Franco-Cortés2
1 Laboratory of Immunodetection and Bioanalysis, Faculty of Dentistry, University of Antioquia. Medellín, Colombia.
2 Salud Bucal y Bienestar Research Group, Faculty of Dentistry. University of Antioquia. Medellín, Colombia.
CORRESPONDENCE Dra. Maria Cecilia Martinez-Pabon Calle 64 N° 52-59. Medellin, Colombia. S.A. macemapa@hotmail.com
ABSTRACT
Dental caries is a multifactorial disease, in which saliva has an important role. Saliva properties affect the growth of cariogenic microorganisms, so variations below threshold levels are considered risk factors for the development of dental caries. Salivary tests and microbiological counts of cariogenic bacteria are often used for clinical purposes in order to reveal problems that could explain caries development and have been evaluated in multiple studies, but these studies have mainly been carried out on adults. Thus, the aim of this study was to identify physicochemical salivary properties, mutans streptococci and Lactobacillus counts among preschool children, and their relationship with ECC in a cross-sectional study in Medellin, Colombia. Caries was determined using ICDAS criteria for a sample of 201 preschool children. Stimulated saliva samples were processed for bacterial detection, mean flow rate, pH and buffer capacity calculation. Risk variables for ECC were assessed individually and adjusted using a binary logistic regression model. The results showed that 71.14% of study subjects had ECC. Flow rate, final pH and Buffer capacity increased with age, but none of them were related to the presence of ECC. Although OR analysis detected correlations between ECC, gender, age, dental eruption stage and Lactobacillus presence, binary logistic regression analysis only showed gender and Lactobacillus as strongly associated with ECC. It can be concluded that gender and Lactobacillus presence are key variables for the presence of ECC in these children; whilst age and dental eruption stage play a minor role. Further studies are required to clarify the role of mutans streptococci counts in this population and to establish normal salivary parameters as well as threshold levels for hyposalivation in preschool children, as results showed differences with normal parameters used for adults.
Key words: Dental caries; Saliva; Streptococcus mutans; Lactobacillus; Preschool children.
RESUMEN
Caries de la infancia temprana, propiedades fisicoquímicas de la saliva y recuento de Estreptococos del grupo Mutans y Lactobacillus en niños pre-escolares de Colombia
La caries dental es una enfermedad multifactorial en la cual la saliva tiene un impacto importante. Las propiedades de la saliva afectan el crecimiento de microorganismos cariogenicos y por lo tanto las variaciones por debajo de los niveles umbral son consideradas factor de riesgo para el desarrollo de caries. Las pruebas salivares y los recuentos de microorganismos cariogenicos son utilizadas con frecuencia en la clinica para identificar factores que puedan explicar el desarrollo de lesiones cariosas y han sido evaluados en multiples estudios, pero estos han sido desarrollados principalmente en adultos. El objetivo de este trabajo fue identificar propiedades fisicoquimicas de la saliva y recuentos de estreptococos del grupo mutans y Lactobacillus, en relacion con la Caries de la Infancia Temprana (CIT). Para esto, se realizo un estudio descriptivo transversal. Se registraron las lesiones de caries dental con el sistema ICDAS en 201 ninos pre-escolares de hogares infantiles de estrato socioeconomico medio-bajo. Se obtuvieron muestras de saliva estimulada, que fueron procesadas para detectar estreptococos del grupo mutans y Lactobacillus; se calculo la tasa de secrecion salivar, pH y capacidad Buffer. Se analizaron posibles variables indicadoras de riesgo para CIT, mediante el calculo de OR individuales por variable y OR ajustadas mediante un modelo de regresion logistica binario. Los resultados indicaron que el 71.14% de los ninos presentaban CIT. Las propiedades de la saliva mostraron un mejor comportamiento con la edad, con valores diferentes a los parametros definidos para poblaciones adultas, pero ninguna tuvo relacion con la enfermedad. El genero y la presencia de Lactobacillus mostraron asociacion fuerte en el analisis de regresion sugiriendolos como principales indicadores de riesgo para CIT en esta poblacion. Se requieren estudios prospectivos y analizar variaciones biologicas, para clarificar el papel de los estreptococos del grupo mutans y revisar los valores umbral para la evaluacion de la secrecion salivar en preescolares.
Palabras clave: Caries dental; Saliva; Streptococcus mutans; Lactobacillus; Niños preescolares.
INTRODUCTION
Early Childhood Caries (ECC)1 constitutes a burden for the dental profession and public health administrators, as many authors report that the situation in primary dentition has not improved over the last decades, impacting children’s quality of life and future oral health2-4. Biological as well as socioeconomic factors are related to the development of dental caries, and are both relevant in understanding the disease process and designing individual or population- oriented preventive approaches. The cariogenic capacity of oral microorganisms is essential, but does not alone determine the presence of dental caries. It is necessary to have appropriate substrates and host-related physiological conditions to enable colonization and survival, creating an imbalance that facilitates the development of caries lesions5. Saliva has an important impact, through functions relying on physicochemical characteristics such as flow rate, pH and buffering capacity, so variations under threshold levels are considered risk factors for the development of dental caries6.
Salivary tests and microbiological counts of cariogenic bacteria are used for clinical and research purposes7. However, studies of salivary flow rate, pH and buffer capacity have mainly been carried out on adults and there are limited data on stimulated salivary flow rates in children, especially in younger children, although there is a clear relationship with dental caries8-10. In Colombia, there are no available data on dental caries in preschool children, although the reduction of dental caries levels is more evident in permanent than in primary dentition. The latest National Oral Health Survey showed that 60.4% of five-year old children had experienced dental caries11, and recent studies show no improvement12. The aims of this study were: (1) to identify stimulated salivary flow rate, buffer capacity, pH and, bacterial counts of mutans streptococci and Lactobacillus, among a preschool children population and (2) to establish their correlation with ECC.
MATERIAL AND METHODS
Population and sample
The sample was taken from children at five kindergartens selected from among a total of twelve centers located in northeastern zone of the city of Medellin, Colombia, South America. These private institutions receive government aid for attending to children living in an urban area with middle and middle-low socioeconomic conditions, according to the classification by the Mayor of Medellin, taking into account housing characteristics. Saliva samples were taken and clinical evaluations performed on 201 preschool children. This study was approved by the Ethics Committee at Faculty of Dentistry of Antioquia University, and children’s parents submitted written informed consent.
Clinical examination
Caries levels were recorded using the International Caries Detection and Assessment System (ICDAS) criteria. A single examiner was trained by a pediatric dentist not belonging to ICDAS Committee, who was trained by an ICDAS Coordinating Committee member. The training included theoretical and clinical sessions. Cohen’s Kappa were 0.72 (IC 0.65 – 0.78) inter-examiners and 0.77 (IC 0.70 – 0.83) intra-examiner. The examination took place in a portable dental chair under halogen light, installed at the selected centers. Teeth were cleaned, dried, and cotton rolls were placed to isolate the examination area; visual inspection was performed with a plain mouth mirror and WHO 11.5 probe to check for surface discontinuity13. For classification of the study group according to the diagnosis of caries, levels 1 and 2 of ICDAS were taken together and included in the group of children with ECC. In other words, children diagnosed without dental caries had to be free of restorations, cavitated carious lesions and any white spot lesion suggestive of dental caries. ECC and severe caries were diagnosed following the criteria of the American Academy of Pediatric Dentistry. Tooth eruption stages were classified as incomplete in children with less than 20 temporary teeth present in mouth.
Saliva sampling
Stimulated salivary samples were collected between 8:00 a.m. and 11:00 a.m., at least one hour after a meal. Each child was seated in a school chair and the procedure was carried out under close supervision. Stimulation was induced by chewing 1 gr. of wax for one minute until soft. Then the child spit the saliva intermittently into a sterile 50-mL graduated plastic centrifuge tube (Greiner Bio-oneR, Frickenhausen, Germany) for 5 minutes. After collection, samples were transported to the laboratory in refrigerated conditions, within the following 3 hours.
Saliva analysis
The total volume of saliva was calculated avoiding foam, with a sterile micropipette. Mean flow rate was calculated taking into account the total time of sample collection and the volume of saliva. The pH of the samples was measured in a digital pHmeter (Metrohm Ion Analysis, Sweden). One experienced examiner performed all the observations according to Ericsson’s method14. Briefly, 1 mL of saliva was mixed with 3 mL of 0.005 N hydrochloric acid, subject to bubbling, and after 20 minutes, final pH values were determined again. Buffer capacity was calculated by the difference between initial and final pH. For mutans streptococci and Lactobacillus counts, samples were vortexed for 30 sec (IKAR VibroFix, Staufen, Germany) at maximal setting. Then the samples were serially diluted from 10-1 to 10-6 with Brain Heart Infusion (Difco Laboratories, Detroit, MI). The 10-4, 10-5, and 10-6 dilutions were plated on Mitis Salivarius Agar (Difco, USA) supplemented with 0.001% potassium tellurite, 0.2 U/mL bacitracin (Sigma, USA), and 10% (w/v) sucrose (CJ Co., Korea) (MSB agar). To determine mutans streptococci, four typical colonies were randomly selected from the plate cultures of each child exhibiting bacterial growth after 48 hours of anaerobic incubation, based on their morphology, size, and color. Then they were identified using the API-20 STREP system (bioM’erieux Vitec, Inc., Hazelwood, MO). Rogosa agar was used to culture Lactobacillus spp15. The 10-3 dilution was plated and incubated anaerobically at 37°C for at least 48 hours. After incubation, agar plates were screened at 10-50X magnification using a Stemi 2000R stereomicroscope (Carl Zeiss, Oberkochen, Germany). CFU/mL of individual species were calculated by a single trained investigator (MM).
Statistical Analysis
Data collected were analyzed using Statistical Package for the Social Sciences (SPSS) 15.0R (SPSS, Chicago, IL, USA). The proportion of children with ECC and Severe Early Childhood Caries condition (S-ECC) was calculated taking into account the evidence of dental caries experience16. Demographic characteristics and cultured microorganisms were analyzed based on the frequency distributions and were compared to identify differences in the prevalence of ECC using two-sided Fisher’s exact test for variables with two categories and Pearson’s Chi square test (χ2) for variables with more than 2 categories. To identify differences between age and levels of salivary factors such as salivary flow rate, initial salivary pH, final salivary pH and buffer capacity, Pearson’s Chi square test was applied. Median values and range were obtained for salivary flow rate, initial pH, final pH and buffer capacity according to the clinical groups for all subjects. The Kolmogorov-Smirnov test for large sample size did not show normal distribution, so variables were analyzed using a nonparametric Mann-Whitney U-test to compare between groups with and without ECC. Cultured microorganisms were categorized into four groups for mutans streptococci, according to CFU/mL, and two groups for presence or absence of Lactobacillus, to be entered in a logistic regression model. Odds ratio (OR) with 95% confidence interval was calculated for variables related to dental caries development. Variables with OR values greater than 2, a confidence interval not including 1.0, and a P-value < 0.05, were considered risk indicator variables. Finally, a binary logistic regression model was used; each candidate risk variable was evaluated after adjusting for the other candidate risk variable and for potential confounding variables. In the stepwise logistic regression analysis, P < 0.05 was used as the entry criterion while P > 0.10 was the removal criterion. The Hosmer-Lemeshow goodness-of-fit test statistic was set at P > 0.05. Significance for all tests was established at a P-value < 0.05.
RESULTS
This study obtained information about salivary properties, cariogenic bacteria and ECC in 201 (97 female and 104 male) preschool children living in a middle-low and low income urban area in Medellin, Colombia. Demographic and clinical characteristics according to presence or absence of ECC in the study subjects are shown in Table 1.
Table 1: Demographic characteristics of the children and clinical findings (n=201). *

ECC was detected in 71.14% of study subjects, whereas 56.7% had severe forms of the disease. Categories established for gender and dental eruption stage showed significant difference (P < 0.05) in relation to presence/absence of ECC. The proportion of children with dental caries was higher for males than for females, and there was also a significant difference between age groups and ECC (P < 0.05), with 5-year-old children having the highest proportion of affected subjects (39.8%). Salivary properties related to flow rate, final pH and buffer capacity showed significant differences by age, whereas initial salivary pH showed no statistical difference (Table 2). It was found that salivary flow rate increased with age; nevertheless, a high proportion of children (63.2%) were in the low rate group, defined as < 0.5 ml/min according to accepted threshold. Similarly, final pH increased with age, children reaching ≥ 6 value varying from 42.9 % by age two, to 73.8% at five years. Buffer capacity, defined as the difference between initial and final pH, showed an improvement with age, reflected in lower variations.
Table 2: Physicochemical salivary properties of the children by age (n=201).
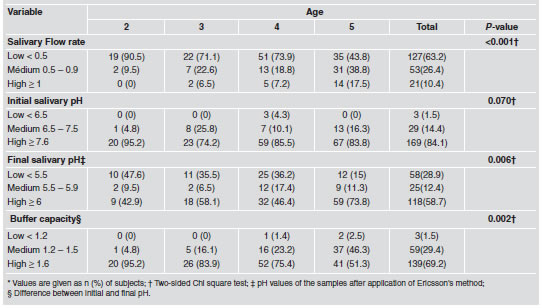
Table 3 presents microorganism counts and salivary properties, including variations between initial and final pH (buffer capacity) in relation to ECC presence. None of the salivary factors analyzed was significantly related to caries presence, showing similar mean values in both groups. In contrast, significant relationship with mutans streptococci levels (P-0.048) and Lactobacillus presence (P-0.00) were found.
Table 3: Counts (cfu/ml) of microorganisms and salivary properties in stratified clinical groups (n=201)
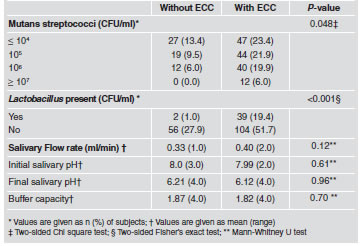
Table 4 shows frequency, percentage and estimated odds ratios (OR) and 95% confidence intervals (CI) for gender, age, eruption stage, mutans streptococci levels and Lactobacillus presence. OR for development of dental caries was significantly greater (P < 0.05) for male children older than 3 years and complete primary dentition. Although mutans streptococci appeared to be a positive indicator of caries risk in the previous analysis (Table 3), when each category is compared in the OR analysis to the reference group (mutans streptococci ≤ 104), the mutans streptococci loses strength as risk indicator for the development of dental caries. Calculation of OR with ≥ 107 category was not possible because of the absence of children without ECC with this amount of bacteria. The OR of development of dental caries was significantly greater (P < 0.05) for children with presence of Lactobacillus. Based on the OR analysis, four variables were considered as candidate risk variables: gender, age, dental eruption stage and presence of Lactobacillus. These variables were evaluated in a binary logistic regression analysis using a stepwise variable selection; the model with adjusted OR is shown in Table 5. The age and denture eruption stage changed and became non-significant, only gender and presence of Lactobacillus remained significant in the model, adjusting for the other variables; although they presented only a slight increase of the adjusted OR, the model indicated that male gender and presence of Lactobacillus were 2.09 and 11.7 times more likely to have ECC. None of the variables included in the regression model presented interacting relationships (Wald test. P value > 0.05).
Table 4: Univariate analysis of indicative factors for ecc in children.

Table 5: Binary logistic regression analysis: significant risk factors for development of ECC‡.

DISCUSSION
Using ICDAS criteria, the children studied showed a high prevalence of dental caries (71.14%), considering their early age and the high proportion of subjects with incomplete dental eruption (84.7%). Although dental caries is more prevalent in developing countries and low-income communities17, it is a complex disease with multiple contributing factors, some of which may present variations from one population to another, creating unique environments. In fact, some demographic and clinical characteristics, as well as laboratory findings, showed statistically significant differences between children with and without ECC in a population sharing other characteristics.
It is well known that dental caries has cumulative characteristics, so lesions are more easily identified in older age groups. In this study, the relationship between age and tooth eruption stage with dental caries was consistent with other studies showing that dental caries severity increases with age16,18,19, as well as the finding that Early Childhood Caries was more frequent in males20. However, results related to stimulated saliva properties in the children examined requires detailed analysis, considering the conflicting results among authors and the lack of inclusion of values that appear frequently in samples from children when compared to established thresholds for adults, which makes the clinical use of these variables as clinical risk indicators a complex field. Saliva properties showed statistically significant differences by age, reflecting a tendency to increase flow rate as well as mean values of final pH and buffer capacity, constituting potential protective factors. However, no statistically significant differences were found in salivary flow rate, initial and final pH, and buffer capacity between children with and without dental caries, contrary to some previous studies8, 21, a finding supporting the ambiguous evidence available in relation to salivary properties and dental caries in children. While some authors have found that chronically low salivary flow rate (<0.8-1.0 ml/min stimulated whole saliva) is a strong indicator of an increased risk of caries21-25, others were not able to demonstrate a relationship or predictive value for individuals with normal salivary flow rate26-30. Similarly, some studies report a correlation between low levels of salivary pH and buffer capacity and caries21,23,31, but others conclude that salivary factors are poor indicators of caries risk and caries experience28,32-35. An additional problem for concluding studies about saliva and ECC is the fact that there are few reports on stimulated salivary properties such as flow rate in young children27,36 so normality parameters or threshold limits are not totally defined in scientific literature. While analyzing the relationship between salivary variables, age and dental caries, we noted that it was necessary to adjust threshold levels for salivary flow rate, as 174 children (79.8%) presented values below 0.7 ml/minute, considered as the reference value for hyposalivation in adults37,38. This finding shows the need to establish proper reference values for preschool children through large population samples and analytical studies in this specific age group. Determining the role of mutans streptococci and Lactobacillus as caries risk indicators is a complex matter, as they can also be part of normal oral flora in healthy people, or in contrast, people with carious lesions may have no detectable counts of these bacteria in their mouths39-42. Nevertheless, the evidence indicates both microorganisms as important cariogenic agents, promoting their usefulness in risk assessment, as they are associated with a considerable increase in caries risk39,43.
In this study both, high levels of mutans streptococci and presence of Lactobacillus showed significant statistical differences between children with and without ECC (p < 0.05). However, OR analysis showed no association between ECC and mutans streptococci counts, in agreement with some authors39,44, although they are recognized in the literature as an important variable in caries risk assesment45. So we can hypothesize that the high counts of this group of microorganisms found in children without ECC in this study may indicate that a carious process may be present at a subclinical level, a limitation of this cross-sectional study in comparison with prospective ones, or biological variations within bacteria present in this population that need to be explored in through more specific tests. In contrast, presence of Lactobacillus proved to be a good candidate risk variable, as did complete temporary teething, age above four years and male gender, but in the binary logistic regression analysis, tooth eruption stage and age showed a reduction in their effect when adjusted. So Lactobacillus and gender were the only variables analyzed considered to be strong risk factors for ECC, in agreement with other studies46,47.
Our findings show limitations for using conventional risk assessment models for this preschool population, and that some key variables show no relationship with ECC, suggesting that further studies are needed on aspects such as salivary properties in children under six. This initial information may provide guidelines for future studies seeking a more complete understanding of risk factors related to the physicochemical properties of saliva affecting the development of ECC in children living in developing countries.
CONCLUSIONS
• Only male gender and Lactobacillus presence could be considered strong indicators for ECC presence in the population studied.
• Physicochemical salivary properties showed no relationship with ECC presence or absence, but results showed the need to conduct studies leading to the establishment of normal parameters as well as threshold levels for hyposalivation and saliva alteration in preschool children.
• Prospective studies, as well as genetic microbiological analyses, are required to elucidate controversial results for mutans streptococci counts in relation to ECC in this population.
ACKNOWLEDGEMENTS
This research was supported by the Technical Research Council of the Faculty of Dentistry (University of Antioquia, CIFO-Code 017-2006). The authors would like to thank Lida Zamaida Chica and Catalina Londono for their valuable participation.
1. American Academy of Pediatric Dentistry Clinical Affairs Committee-Restorative Dentistry Subcommittee, American Academy on Pediatric Dentistry Council on Clinical Affairs. Guideline on pediatric restorative dentistry. Pediatr Dent 2008 -2009;30 Suppl 7:163-169. [ Links ]
2. Bonecker M, Cleaton-Jones P. Trends in dental caries in Latin American and Caribbean 5-6- and 11-13-year-old children: a systematic review. Community Dent Oral Epidemiol 2003;31:152-157. [ Links ]
3. Truin GJ, van Rijkom HM, Mulder J, van’t Hof MA. Caries trends 1996-2002 among 6- and 12-year-old children and erosive wear prevalence among 12-year-old children in The Hague. Caries Res 2005;39:2-8.
4. Haugejorden O, Birkeland JM. Analysis of the ups and downs of caries experience among Norwegian children aged five years between 1997 and 2003. Acta Odontol Scand 2005;63:115-122. [ Links ]
5. Marsh PD. Dental plaque as a microbial biofilm. Caries Res 2004;38:204-211. [ Links ]
6. Axelsson P. Internal Modifying Factors Involved in Dental Caries. In: Anderson C, editor. Diagnosis and Risk Prediction of Dental Caries. 1st ed. Germany: Quintessence Publishing Co, Inc.; 2000;91-146. [ Links ]
7. Larmas M. Saliva and dental caries: diagnostic tests for normal dental practice. Int Dent J 1992;42:199-208. [ Links ]
8. Holbrook WP, de Soet JJ, de Graaff J. Prediction of dental caries in preschool children. Caries Res 1993;27:424-430. [ Links ]
9. Holbrook WP. Dental caries and cariogenic factors in preschool urban Icelandic children. Caries Res 1993;27:431-437. [ Links ]
10. Pajari U, Lahtela P, Lanning M, Larmas M. Effect of antineoplastic therapy on dental maturity and tooth development. J Pedod 1988;12:266-274. [ Links ]
11. Colombia, Ministerio de Salud. Centro Nacional de Consultoria. III Estudio Nacional de Salud Bucal – ENSAB III, II Estudio Nacional de Factores de Riesgo de Enfermedades Cronicas – ENFREC II. 1999 2a ed. Bogota: Ministerio de Salud; 1999.
12. Franco AM, Ochoa EM, Ramirez BS, Segura AM, Tamayo A, Garcia C. Situacion de salud bucal de los escolares de Medellin: VI monitoreo, ano 2006. Rev Salud Publica Med 2007;1:58-69. [ Links ]
13. Ismail AI, Sohn W, Tellez M, et al. The International Caries Detection and Assessment System (ICDAS): an integrated system for measuring dental caries. Community Dent Oral Epidemiol 2007;35:170-178. [ Links ]
14. Ericsson Y, Hellstrom I, Jared B, Stjernstrom L. Investigations into the relationship between saliva and dental caries. Acta Odontol Scand 1954;11:179-194. [ Links ]
15. Rogosa M, Mitchell JA, Wiseman RF. A selective medium for the isolation and enumeration of oral lactobacilli. J Dent Res 1951;30:682-689. [ Links ]
16. Ismail AI, Lim S, Sohn W, Willem JM. Determinants of early childhood caries in low-income African American young children. Pediatr Dent 2008;30:289-296. [ Links ]
17. Selwitz RH, Ismail AI, Pitts NB. Dental caries. Lancet 2007 Jan 6;369(9555):51-59. [ Links ]
18. Segovia-Villanueva A, Estrella-Rodriguez R, Medina-Solis CE, Maupome G. Caries severity and associated factors in preschool children aged 3-6 years old in Campeche City, Mexico. Rev Salud Publica (Bogota) 2005;7:56-69. [ Links ]
19. Escobar GM, Ramirez BS, Franco AM, Tamayo AM, Castro JF. Experiencia de caries dental en ninos de 1- 5 anos de bajos ingresos. Rev CES Odont 2009;22:21-28. [ Links ]
20. Beltran-Aguilar ED, Barker LK, Canto MT, et al. Surveillance for dental caries, dental sealants, tooth retention, edentulism, and enamel fluorosis—United States, 1988- 1994 and 1999-2002. MMWR Surveill Summ 2005;54:1-43.
21. Tukia-Kulmala H, Tenovuo J. Intra- and inter-individual variation in salivary flow rate, buffer effect, lactobacilli, and mutans streptococci among 11- to 12-year-old schoolchildren. Acta Odontol Scand 1993;51:31-37. [ Links ]
22. Kirstila V, Hakkinen P, Jentsch H, Vilja P, Tenovuo J. Longitudinal analysis of the association of human salivary antimicrobial agents with caries increment and cariogenic micro-organisms: a two-year cohort study. J Dent Res 1998; 77:73-80. [ Links ]
23. Vehkalahti M, Nikula-Sarakorpi E, Paunio I. Evaluation of salivary tests and dental status in the prediction of caries increment in caries-susceptible teenagers. Caries Res 1996;30:22-28. [ Links ]
24. Woltgens JH, Gruythuysen RJ, Geraets WG. Relationship between cariogenic events and salivary tests in boys and girls: oral examination. J Biol Buccale 1992;20:145-149. [ Links ]
25. Woltgens JH, Gruythuysen RJ, van der Linden LW, Geraets WG. Cariogenic changes in dental enamel of boys and girls in relation to salivary properties. II. Radiological examination. J Biol Buccale 1992;20:235-240. [ Links ]
26. Dodds MW, Johnson DA, Mobley CC, Hattaway KM. Parotid saliva protein profiles in caries-free and cariesactive adults. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 1997;83:244-251. [ Links ]
27. O’Sullivan EA, Curzon ME. Salivary factors affecting dental erosion in children. Caries Res 2000;34:82-87.
28. Raitio M, Pienihakkinen K, Scheinin A. Assessment of single risk indicators in relation to caries increment in adolescents. Acta Odontol Scand 1996;54:113-117. [ Links ]
29. Sullivan A, Schroder U. Systematic analysis of gingival state and salivary variables as predictors of caries from 5 to 7 years of age. Scand J Dent Res 1989;97:25-32. [ Links ]
30. Sullivan A. Correlation between caries incidence and secretion rate/buffer capacity of stimulated whole saliva in 5-7-year-old children matched for lactobacillus count and gingival state. Swed Dent J 1990;14:131-135. [ Links ]
31. Pienihakkinen K. Caries prediction through combined use of incipient caries lesions, salivary buffering capacity, lactobacilli and yeasts in Hungary [corrected. Community Dent Oral Epidemiol 1987;15:325-328. [ Links ]
32. Almstahl A, Kroneld U, Tarkowski A, Wikstrom M. Oral microbial flora in Sjogren’s syndrome. J Rheumatol 1999; 26:110-114.
33. Kirstila V, Tenovuo J, Ruuskanen O, Nikoskelainen J, Irjala K, Vilja P. Salivary defense factors and oral health in patients with common variable immunodeficiency. J Clin Immunol 1994;14:229-236. [ Links ]
34. Pohjamo L, Knuuttila M, Tervonen T, Haukipuro K. Caries prevalence related to the control of diabetes. Proc Finn Dent Soc 1988;84:247-252. [ Links ]
35. Soderholm G, Birkhed D. Caries predicting factors in adult patients participating in a dental health program. Community Dent Oral Epidemiol 1988;16:374-377. [ Links ]
36. Crossner CG. Salivary flow rate in children and adolescents. Swed Dent J 1984;8:271-276. [ Links ]
37. Ericsson Y, Hardwick L. Individual diagnosis, prognosis and counselling for caries prevention. Caries Res 1978;12 Suppl 1:94-102. [ Links ]
38. Flink H, Bergdahl M, Tegelberg A, Rosenblad A, Lagerlof F. Prevalence of hyposalivation in relation to general health, body mass index and remaining teeth in different age groups of adults. Community Dent Oral Epidemiol 2008;36:523-531. [ Links ]
39. Thenisch NL, Bachmann LM, Imfeld T, Leisebach Minder T, Steurer J. Are mutans streptococci detected in preschool children a reliable predictive factor for dental caries risk? A systematic review. Caries Res 2006;40:366-374. [ Links ]
40. Snyder ML, Porter DR, Claycomb CK, Sims W, Macho FR. Evaluation of Laboratory Tests for the Estimation of Caries Activity. Arch Oral Biol 1963;168:541-547. [ Links ]
41. Davies GN, King RM, Collins AA. The relationship between Lactobacillus counts, Snyder tests and the subsequent incidence of dental caries. Arch Oral Biol 1959;1:62-73. [ Links ]
42. van Houte J. Bacterial specificity in the etiology of dental caries. Int Dent J 1980;30:305-326. [ Links ]
43. Bratthall D, Hansel Petersson G. Cariogram - a multifactorial risk assessment model for a multifactorial disease. Community Dent Oral Epidemiol 2005;33:256-64. [ Links ]
44. Acevedo AM, Ray MV, Socorro M, Rojas-Sanchez F. Frequency and distribution of Mutans Streptococci in dental plaque from caries-free and caries-affected Venezuelan children. Acta Odontol Latinoam 2009;22:15-20. [ Links ]
45. Bratthall D, Hansel Petersson G. Cariogram—a multifactorial risk assessment model for a multifactorial disease. Community Dent Oral Epidemiol 2005;33:256-264.
46. Teanpaisan R, Thitasomakul S, Piwat S, Thearmontree A, Pithpornchaiyakul W, Chankanka O. Longitudinal study of the presence of mutans streptococci and lactobacilli in relation to dental caries development in 3-24 month old Thai children. Int Dent J 2007;57:445-451. [ Links ]
47. Beighton D, Adamson A, Rugg-Gunn A. Associations between dietary intake, dental caries experience and salivary bacterial levels in 12-year-old English schoolchildren. Arch Oral Biol 1996;41:271-280. [ Links ]














