Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Acta Odontológica Latinoamericana
versión On-line ISSN 1852-4834
Acta odontol. latinoam. vol.25 no.2 Buenos Aires oct. 2012
ARTÍCULOS ORIGINALES
Effect of water storage on microtensile bond strength of a two-step self-etch adhesive and a two-step etch-and-rinse adhesive
Juan C. Mendez1, Gloria E. Pabon1, Sergio P. Hilgenberg2, Eugenio J. Garcia3, Beatriz Arana-Correa1
1 Department of Biomaterials, Operative and Esthetic Dentistry, School of Dentistry, University of Santiago de Cali, Cali, Colombia.
2 School of Dentistry, University Estadual de Ponta Grossa, Ponta Grossa, PR, Brazil.
3 School of Dentistry, UNORP, São Jose do Rio Preto, SP, Brazil.
CORRESPONDENCE Eugenio Jose Garcia Departamento de Materiais Dentarios, Faculdade de Odontologia, Universidade de Sao Paulo. Av. Prof. Lineu Prestes 2227 Sao Paulo, SP. Brasil e-mail: eugarcia@usp.br
ABSTRACT
This study evaluated the effect of 24 h (1d) and one-year (1yr) water storage on the microtensile dentin bond strength of a twostep total-etch and a two-step self-etch adhesive system. Ten extracted human third molars were sectioned perpendicularly to their long axis to expose flat occlusal dentin surfaces. Teeth were divided into two groups (n=5) according to the adhesive used: a two-step etch-and-rinse adhesive (Adper Single Bond 2) and two-step self-etching adhesive (Adper Scotchbond SE). Composite resin (Z350) build-ups were incrementally constructed on the bonded surfaces. Specimens were sectioned into sticks (cross-sectional mean area 0.8 mm2) and after 1d and 1yr of storage in distilled water at 37°C, the sticks were stressed to failure by microtensile test (1 mm/min). Interfacial observation of silver nanoleakage was performed using scanning electron microscopy (SEM). Data (MPa) were analyzed by two-way ANOVA and Bonferroni's test (p = 0.05). SB2 showed the highest bond strength values after 1d of water storage. After 1yr, SB2 values significantly decreased and were similar to ASE, independently of water storage period. Both adhesives, independently of storage time, showed silver nitrate uptake within the hybrid layer and the adhesive layer. One-year of water storage only affected the bond strength of the two-step etch-and-rinse adhesive.
Keywords: Dentin bonding agent; Tensile strength; Scanning electron microscopy.
RESUMEN
Efecto del almacenamiento en agua en la resistencia adhesiva de un adhesivo auto-acondicionante de dos pasos y de un adhesivo de grabado ácido independiente de dos pasos
Evaluar el efecto del almacenamiento en agua por 24 h (1d) y un ano (1a) sobre la resistencia de union de un adhesivo de grabado acido independiene de dos pasos y un adhesivo autoacodicionante de dos pasos. Diez terceros molares fueron cortados perpendiculares a su eje longitudinal para exponer superficies planas de dentina. Los dientes fueron divididos en dos grupos segun el adhesivo a ser usado: uno de acondicionamiento acido independiente de dos pasos (Adper Single Bond 2) y uno autoacondicionante de dos pasos (Adper Scotchbond SE). Incrementos en resina compuesta (Z350) fueron construidas sobre las superficies tratadas con adhesivo. Los especimenes fueron cortados para obtener probetas (area de seccion transversal = 0,8 mm2) y despues de 1d y 1a de almacenamiento en agua destilada a 37° C, fueron sometidas a la prueba de microtraccion (1 mm/min). La captacion de nitrato de plata en las interfaces fue analizada con microscopia electronica de barrido (MEB). Los datos (MPa) fueron analizados con ANOVA de dos factores y la prueba de Bonferroni (p = 0,05). Despues de 1d de almacenamiento SB2 presento los mayores valores de resistencia de union. No hubo diferencias entre los adhesivos despues de 1a, y para ASE estos valores fueron similares al tiempo 1d. Independientemente del tiempo de almacenamiento, ambos adhesivos presentaron captacion de nitrato de plata dentro de la capa hibrida y la capa de adhesivo. Un ano de almacenamiento en agua solo afecto los valores de resistencia de union del adhesivo de grabado acido independiente de dos pasos.
Palabras clave: Agentes de adhesion a dentina; Resistencia a la traccion; Microscopia electronica de barrido.
INTRODUCTION
Self-etching adhesives were developed to provide some advantages over etch-and-rinse adhesives such as simplification of the bonding technique and reduction of postoperative sensitivity. Independently of the bonding strategy, i.e. the use or not of a preliminary separate etching step, the stability of the bonded interfaces of both approaches depends on the creation of a compact, homogeneous hybrid layer1. Even though most current adhesives systems have shown excellent immediate bond strength values, they do not have the same behavior in long-term studies2.
Several factors are involved in the reduction of the longevity of bonded interfaces, such as occlusal chewing forces, temperature, acid challenges, enzymes and microorganisms3. Water degradation may occur due to collagen or polymer hydrolysis. The former is commonly related to total-etch systems due to the poor resin infiltrated zone with naked collagen fibrils that are highly susceptible to hydrolysis. The latter was observed in both total-etch and self-etch adhesive systems and depends on factors such as the composition of the adhesive systems2. Microtensile test and aging by storage in water are two in vitro methodologies that provide important information about resin-tooth interface degradation after long storage periods. Therefore, the purpose of this study was to evaluate the effect of 24 h and one year of water storage on the μTBS using a twostep etch-and- rinse system as well as a two-step self-etching system. The null hypothesis tested is that no significant difference will be detected among the different bonding strategies in the oneday and one-year water storage period.
MATERIAL AND METHODS
Tooth Preparation
After approval of the Committee of Ethics in Research from the University of Santiago de Cali, ten intact, non-carious, non-restored, human third molars cleaned of gross debris were selected for this study. They were previously disinfected in 0.5% chloramine, stored in distilled water and used within six months after extraction. Flat coronal dentin surfaces were obtained by sectioning the occlusal enamel of all teeth with a diamond disc under water cooling (Isomet 1000, Buehler, Lake Bluff, IL, USA). The exposed dentin surfaces were further polished on wet # 600-grit silicon-carbide paper for 60 s to standardize the smear layer and randomly assigned to two groups (n=5) according to the adhesive system (Table 1): a two-step etch-and-rinse adhesive (Single Bond 2 [SB2], 3M ESPE, St. Paul, MN, USA) and a two-step self-etching adhesive (Adper SE Plus [ASE],3M ESPE, St. Paul, MN, USA).
Table 1: Composition (batch number) of materials used in this study.

Restorative Procedure
Adhesives were applied following manufacturer's instructions. Composite build-ups were made with three incremental layers (2 mm) of resin (Z350, 3M ESPE, St. Paul, MN, USA). Each layer of resin composite was light-activated for 40 s with a quartztungsten halogen-light unit set at 500 mW/cm2 (Optilux 501, Kerr Corporation, Orange, CA, USA).
Microtensile test
After storage in distilled water for 24 h at 37o <±> C each tooth was sectioned into sticks with a cross-sectional area of approximately 0.8 mm2 using a diamond disc under water cooling (Isomet 1000, Buehler, Lake Bluff, IL, USA). Specimens (25-30 per tooth) were randomly divided in two groups and subjected to microtensile bond strength test after 24 h (1d) or 1 year (1yr) of storage in distilled water at 37 oC. The remaining dentin thickness of specimens was not considered. The number of premature debonded sticks (D) per tooth during specimen preparation was recorded. The cross-sectional area of each stick was measured with the digital caliper (Absolute Digimatic, Mitutoyo, Tokyo, TYO, JAP) to the nearest 0.01 mm for calculation of the bond strength values (BS). Each bonded stick was attached to a device for microtensile testing with cyanoacrylate glue and stressed to failure in a universal testing machine (Model 5565, Instron Corp., Canton, MA, USA) at a crosshead speed of 1 mm/min. The failure modes were evaluated at 400X (HMV-2, Shimadzu; Tokyo, JAP) and classified as cohesive ([C] failure exclusive within dentin or resin composite, adhesive ([A] failure at resin/dentin interface), or adhesive/mixed ([A/M] failure at resin/dentin interface that included cohesive failure of the neighboring substrates).
Scanning electron microscopy for silver nitrate leakage evaluation
For scanning electron microscopy (SEM) analysis of the interface leakage two bonded sticks of each tooth were randomly selected and immersed in ammoniacal silver nitrate solution for 24 h. The silver- stained sticks were rinsed thoroughly in distilled water and placed in photodeveloping solution for 8 h under a fluorescent light to reduce the diamine silver ions into metallic silver grains within potential voids along the bonded interfaces.Then, stained sticks were embedded in epoxy resin (Epon- Thin™, Buheler Ltd., Illinois, USA) and polished to high gloss with SiC papers and diamond pastes of decreasing abrasiveness (Metadi IIR, Buehler Ltd, Lake Bluff, IL, USA) using polish cloths. After sonication in distilled water for 5 min they were mounted on aluminum stubs, then carbon coated (BAL-TEC SCD 050 Sputter Coater, Balzers, Liechtenstein) and observed by SEM (SS 440, LEO Electron Microscopy Ltd; Cambridge, UK).
Statistical analysis
Microtensile bond strength values were analyzed by two-way ANOVA, with the adhesive system and storage time as main factors. Bonferroni test for multiple comparisons was used when the F-factor was significant (α = 0.05). Representative SEM images at the resin–dentin interfaces were only evaluated qualitatively.
RESULTS
Microtensile bond strength
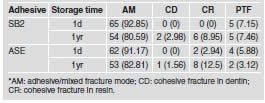
After one day of water storage, SB2 showed the highest bond strength values (38.22±3.90) (p < 0.05). However, after one year, SB2 mean (30.90±3.12) showed a statistically significant decrease (p < 0.05) and was statistically similar (p > 0.05) to ASE after 1d (31.22±4.85) and 1yr (30.95±2.22) of water storage. ASE mean was not affected by water storage (p > 0.05) (Table 2). The overall failure modes of this study are depicted in Table 3, showing that the majority of failures were adhesive. The number of pre-testing failures was similar among groups.
Table 2: Mean bond strength (MPa) and standarddeviation for each experimental condition.
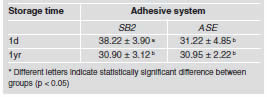
Table 3: Number of specimens (percentage %) according to the fracture pattern* or pre-test failures (PTF) for each experimental condition.
Scanning electron microscopy
The resin-dentin interfaces of all groups are shown in Fig. 1. Hybrid layer and resin tag formation were observed for both adhesives. Silver uptake within the hybrid layer and water tree formation within adhesive layer were observed for both adhesives independently of water storage period.

Fig. 1: Representative scanning electron micrographs (1200x) of SB2 (a and b) and ASE (c and d) after 1d (a and c) and 1y (b and d) of water storage. Both adhesives showed silver nitrate uptake within the hybrid layer (white arrow) and "water-tree like" formations in the adhesive layer (asterisk). (Bar = 10 μm) (Co= composite resin; AL= adhesive layer; HL= hybrid layer; De= dentin).
DISCUSSION
Although clinical studies are preferred for predicting the behavior of adhesive systems, in vitro studies can provide results more quickly and inexpensively4. One year of water storage did not affect the bond strength of ASE. Although it has two bottles, it only reacts with dental surfaces once both liquids, A and B, are mixed into cavities. Hydrophilic and hydrophobic components are separated to avoid phase separation and premature degradation in the bottle5. The high water content of liquid A (80-90%) is necessary for ionization of the acidic functional monomers of liquid B, but had no deleterious effect on bond strength even after 1yr of water storage. Hydrolysis of unprotected collagen is unexpected since demineralization and monomer infiltration occurs at the same time and depth, resulting in the formation of a thin but uniform hybrid layer.
Our results disagree with those obtained by Sauro et al6. The lack of differences between 1d and 1y results could be explained because they used stored whole teeth, whereas our study used stored bonded sticks. As mentioned above, indirect water storage, i.e. storage of entire teeth, showed significantly better bond strength values than direct water storage because the surrounding enamel-resin bond warrants protection against degradation6-8. The bond strength values obtained in our study for ASE are almost three times greater than those obtained by Belli et al.9, who attributed this to the low capacity of ASE to dissolve the smear layer as consequence of the formation of an uneven layer of liquid A resulting in an inhomogeneous etching of the Class I cavities. As mentioned above, our study was performed on a flat surface that lacks angles facilitating the application of the adhesive 7. A previous study showed that the decrease in bond strength of self-etch adhesives was material-dependant. A low pH value produces complete smear layer and smear plug dissolution, facilitating the contact between water and hybrid and adhesive layers10. The major content of more hydrophilic monomers increases water sorption and water entrapment, affecting adhesive polymerization and contributing to hydrolytic degradation7,11-13. The bond strength values of SB2 after 1d of storage were significantly higher than those of ASE. Nevertheless, one year of water storage produced a significant decrease in SB2. Our results are in agreement with the values obtained by previous studies14,15. Simplified adhesives have more hydrophilic monomers that can lead to water sorption, decreasing the mechanical properties of the resin and making the polymer swell8,16. Another factor mentioned is the discrepancy between the depth of demineralized dentin and resin infiltration, which left unprotected collagen fibrils beneath the hybrid layer more prone to hydrolysis13. According to Osorio et al.7, when resin-dentin bonds are protected by surrounding resin-infiltrating enamel, no decrease in bond strength was observed for SB after one-year aging. Metalloproteinases (MMPs) are endogenous enzymes released from the dentin matrix once the conditioning step is performed and have been shown to participate in the degradation of the hybrid layer16. Phosphoric acid is a strong acid (pH= 0.03-0.05) used as a conditioning step for SB2, and ASE reaches a pH of approximately 1.5 when liquids A and B are mixed. Both are sufficiently acidic to activate gelatinolytic and collagenolytic activity, but only phosphoric acid was also able to denature the MMP activity16. However, as shown by De Munck et al.17, this anticolagenolytic activity is not enough to prevent degradation for longer periods, even after the adhesives were modified with MMP inhibitors such as chlorhexidine (CHX). This agrees with the abovementioned results, poor monomer infiltration left unprotected collagen fibrils beneath hybrid layer that can be affected by hydrolytic or enzymatic activity18.
In contrast to phosphoric acid, the pH of ASE was insufficient to destroy the MMPs16. Previous studies have shown that modification of the primer of a two-step self-etching adhesive with an MMP inhibitor (chorhexidine) depends on the concentration of the inhibitor19,20. Nevertheless, our results suggested that demineralized collagen fibrils were not affected by ASE because most of them were covered by resin and thus protected from enzymatic and hydrolytic action. Longer periods of water storage are also suggested for a better understanding of the behavior of the current dental adhesives.
CONCLUSION
One year of water storage promoted a decrease in bond strength of the two-step etch-and-rinse adhesive system.
ACKNOWLEDGEMENTS
This study was partially supported by 3M ESPE and grants from CNPq (142751/08-1).
1. Van Meerbeek B, De Munck J, Yoshida Y, Inoue S, Vargas M, Vijay P, Van Landuyt K, Lambrechts P, Vanherle G. Buonocore memorial lecture. Adhesion to enamel and dentin: current status and future challenges. Oper Dent 2003;28:215-235. [ Links ]
2. Saboia VP, Nato F, Mazzoni A, Orsini G, Putignano A, Giannini M, Breschi L. Adhesion of a two-step etch-andrinse adhesive on collagen-depleted dentin. J Adhes Dent 2008;10:419-422. [ Links ]
3. Pashley DH, Tay FR, Yiu C, Hashimoto M, Breschi L, Carvalho RM, Ito S. Collagen degradation by host-derived enzymes during aging. J Dent Res 2004; 83:216–221.
4. Loguercio AD, Bittencourt DD, Baratieri LN, Reis A. A 36- month evaluation of self-etch and etch-and-rinse adhesives in noncarious cervical lesions. J Am Dent Assoc 2007;138: 507-514 [ Links ]
5. Inoue H, Inoue S, Uno S, Takahashi A, Koase K, Sano H. Microtensile bond strength of two single-step adhesive systems to bur-prepared dentin. J Adhes Dent 2001;3: 129-136. [ Links ]
6. Sauro S, Watson TF, Tay FR, Chersoni S, Breschi L, Bernardi F, Prati C. Water uptake of bonding systems applied on root dentin surfaces: a SEM and confocal microscopic study. Dent Mater 2006;22:671-680. [ Links ]
7. Osorio R, Pisani-Proenca J, Erhardt MC, Osorio E, Aguilera FS, Tay FR, Toledano M. Resistance of ten contemporary adhesives to resin-dentine bond degradation. J Dent 2008; 36:163-169. [ Links ]
8. Abdalla AI, Feilzer AJ. Morphological characterization of the interface between self-etching adhesives and vital dentin. Am J Dent 2007;20:305-308. [ Links ]
9. Belli R, Sartori N, Peruchi LD, Guimaraes JC, Araujo E, Monteiro S Jr., Baratieri LN, Lohbauer U. Slow progression of dentin bond degradation during one-year water storage under simulated pulpal pressure. J Dent 2010;38: 802-810. [ Links ]
10. Tay FR, Pashley DH. Aggressiveness of contemporary self etching systems. I. Depth of penetration beyond dentin smear layers. Dent Mater 2001;17:296-308. [ Links ]
11. Reis A, Loguercio AD, Carvalho RM, Grande RHM. Durability of resin dentin interfaces: effects of surface moisture and adhesive solvent component. Dent Mater 2004; 20:669-676. [ Links ]
12. Klein-Junior CA, Zander-Grande C, Amaral R, Stanislawczuk R, Garcia EJ, Baumhardt-Neto R, Meier MM, Loguercio AD, Reis A.. Evaporating solvents with a warm air-stream: effects on adhesive layer properties and resin-dentin bond strengths. J Dent 2008; 36:618-625. [ Links ]
13. Hosaka K, Tagami J, Nishitani Y, Yoshiyama M, Carrilho M, Tay FR, Agee KA, Pashley DH. Effect of wet vs. dry testing on the mechanical properties of hydrophilic selfetching primer polymers. Eur J Oral Sci 2007;115:239-245. [ Links ]
14. Garcia RN, de Goes MF, Giannini M. Effect of water storage on bond strength of self-etching adhesives to dentin. J Contemp Dent Pract 2007;8:46-53. [ Links ]
15. Tay F, Pashley DH. Have dentin adhesives become too hydrophilic? J Can Dent Assoc 2003;69:726-731. [ Links ]
16. Nishitani Y, Yoshiyama M, Donnelly AM, Agee KA, Sword J, Tay FR, Pashley D. Effects of resin hydrophilicity on dentin bond strength. J Dent Res 2006;85:1016-1021. [ Links ]
17. De Munck J, Mine A, Van den Steen PE, Van Landuyt KL, Poitevin A, Opdenakker G, Van Meerbeek B. Enzymatic degradation of adhesive-dentin interfaces produced by mild self-etch adhesives. Eur J Oral Sci 2010;118:494-501. [ Links ]
18. Pashley DH, Tay FR, Yiu C, Hashimoto M, Breschi L, Carvalho RM, Ito S. Collagen degradation by host-derived enzymes during aging. J Dent Res 2004; 83:216-221. [ Links ]
19. Zhou J, Tan J, Chen L, Li D, Tan Y. The incorporation of chlorhexidine in a two-step self-etching adhesive preserves dentin bond in vitro. J Dent 2009; 37:807-812. [ Links ]
20. De Munck J, Van den Steen PE, Mine A, Van Landuyt KL, Poitevin A, Opdenakker G, Van Meerbeek B. Inhibition of enzymatic degradation of adhesive-dentin interfaces. J Dent Res 2009;88:1101-1106. [ Links ]














