Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Acta Odontológica Latinoamericana
versão On-line ISSN 1852-4834
Acta odontol. latinoam. vol.28 no.1 Buenos Aires abr. 2015
ARTÍCULOS ORIGINALES
Effect of dexamethasone on mandibular bone biomechanics in rats during the growth phase as assessed by bending test and peripheral quantitative computerized tomography
Clarisa Bozzini, Graciela Champin, Rosa M. Alippi, Carlos E. Bozzini
Department of Physiology, School of Dentistry University of Buenos Aires , Argentina
CORRESPONDENCE Dr. Carlos E. Bozzini, Catedra de Fisiologia, Facultad de Odontologia, Universidad de Buenos Aires, Marcelo T. de Alvear 2142, Buenos Aires 1122, Argentina. e-mail: cebozi@fisio.odon.uba.ar
ABSTRACT
Long-term glucocorticoid administration to growing rats induces osteopenia and alterations in the biomechanical behavior of the bone. This study was performed to estimate the effects of dexamethasone (DTX), a synthetic steroid with predominant glucocorticoid activity, on the biomechanical properties of the mandible of rats during the growth phase, as assessed by bending test and peripheral quantitative computed tomographic (pQCT) analysis. The data obtained by the two methods will provide more precise information when analyzed together than separately. Female rats aged 23 d (n=7) received 500μg.kg-1 per day of DXT for 4 weeks. At the end of the treatment period, their body weight and body length were 51.3% and 20.6% lower, respectively, than controls. Hemimandible weight and area (an index of mandibular size) were 27.3% and 9.7% lower, respectively. The right hemimandible of each animal was subjected to a mechanical 3-point bending test. Significant weakening of the bone, as shown by a correlative impairment of strength and stiffness, was observed in experimental rats. Bone density and cross-sectional area were measured by pQCT. Cross-sectional, cortical and trabecular areas were reduced by 20% to 30% in the DTX group, as were other cortical parameters, including the bone density, mineral content and cross-sectional moment of inertia. The "bone strength index" (BSI, the product of the pQCT-assessed xCSMI and vCtBMD) was 56% lower in treated rats, which compares well with the 54% and 52% reduction observed in mandibular strength and stiffness determined through the bending test. Data suggest that the corticosteroid exerts a combined, negative action on bone geometry (mass and architecture) and volumetric bone mineral density of cortical bone, which would express independent effects on both cellular (material quality) and tissue (cross-sectional design) levels of biological organization of the skeleton in the species.
Key words: Mandible-bone-dexamethasone.
RESUMEN
Efecto de dexametasona sobre el comportamiento biomecanico de la mandibula de la rata en fase de crecimiento, determinado mediante test de flexion y tomografia periferica cuantitativa computarizada
La administracion cronica de glucocorticoides a ratas en fase de crecimiento induce osteopenia y modificaciones negativas del comportamiento biomecanico del hueso. El estudio presente fue realizado para estimar los efectos de dexametasona (DTX), esteroide sintetico con actividad glucorticoide predominante, sobre las propiedades biomecanicas de la mandibula de ratas durante la fase de crecimiento, estimacion realizada mediante el ensayo de flexion a tres puntos, por un lado, y tomografia periferica cuantitativa computarizada (pQCT), por el otro. Los datos obtenidos mediante los dos metodos citados brindaran informacion mas precisa cuando son analizados en forma conjunta que cuando son analizados separadamente. Ratas hembras de 23 d de edad (n = 7) recibieron 500μg.kg-1/d por via subcutanea durante 4 semanas. El peso y la longitud corporales mostraron una disminucion del 51.3% y 20.6%, respectivamente, en las ratas tratadas con respecto a las controles (n = 7). El peso de la hemimandibula derecha y el area mandibular (indice del tamano del hueso) disminuyeron 27.3% y 9.7%, respectivamente. La hemimandibula derecha de cada animal fue analizada biomecanicamente en el test de flexion a tres puntos. Se observo un significativo debilitamiento del hueso, demostrado por la disminucion correlativa de la resistencia (a la fractura) y de su rigidez estructural (medida en la fase elastica de deformacion) en los animales experimentales. La densidad osea y el area de seccion transversal fueron estimadas mediante pQCT. Las areas de seccion transversal, cortical y trabecular, mostraron una reduccion significativa de entre 20% y 30%, asi como la densidad osea, su contenido mineral y el momento de inercia de la seccion transversal. El BSI (indice de resistencia osea), el producto de xCSMI y vCtBMD (medidos topograficamente), disminuyo un 56% en las ratas tratadas, valor semejante al 54% y 52% observado en la resistencia y rigidez mandibulares determinadas mediante el test de flexion. El analisis de los resultados obtenidos sugiere que DXT ejerce una accion negativa y combinada sobre la mandibula, sobre su geometria (masa y arquitectura) y sobre su densidad mineral volumetrica del tejido cortical, acciones que expresarian efectos independientes sobre los niveles celular (calidad material) y tisular (diseno arquitectonico) de organizacion biologica del esqueleto en la especie estudiada.
Palabras clave: Mandibula-hueso-dexametasona.
INTRODUCTION
Long-term glucorticoid (GC) administration to growing rats can induce two main effects in long bones: 1) decrease in longitudinal bone growth, decreased bone formation and increased bone resorption, which may explain the decrease in bone mass (osteopenia)1-6; and 2) decrease in the bone biomechanical behavior, as shown by mechanical testing and tomographic studies3,7-12. GC administration increases bone resorption because it stimulates osteoclastogenesis by increasing the expression of RANK ligand and decreasing the expression of its decoy receptor, osteoprogeterin6. The strong effect of GC on bone formation is associated with a decrease in the number of osteoblasts and their function. The decrease in cell number is secondary to a decrease in osteoblastic cell replication and differentiation, and an increase in the apoptosis of mature osteoblasts6. We have previously reported7 that the effects of GC on rat long bone biomechanics seem to reflect combined, negative action on diaphyseal geometry (mass and architecture) and volumetric mineral density of cortical bone, which may express independent effects on cellular (material quality) and tissue (cross-sectional design) levels of biological organization of the skeleton13 in the species. Bones of the axial or appendicular skeleton show biomechanical properties associated with their condition of "weight-bearing bones". However, they are not only influenced by gravity (body weight) but also by the tensions generated during voluntary or reflex contractions of local muscles. In fact, muscle and bone are anatomically and functionally closely connected14. According to the mechanostat theory,13 striated muscle is essential for bone development and maintenance, modeling and remodeling13. Both tissues are derived from somatic mesoderm and accumulate peak tissue mass synchronously, according to genetic information and environmental stimuli14,15. Excess GC thus results in progressive parallel loss of bone (osteopenia) and skeletal muscle (sarcopenia), with profound consequences for quality of life.
The mandible is both morphologically and functionally different from other bones of the axial skeleton. It also arises from a different embryonic germ layer (neuroectoderm), in contrast to bones of the axial or appendicular skeleton, which arise from the mesoderm. At the organ level, the bone of the mandible is dense with a high proportion of cortical bone. Loading of the mandible during mastication has an impact on the mass, density and microarchitecture of the mandibular alveolar bone16, 17. The mandible is not a "weight-bearing bone". However, since it is influenced by mechanical masticatory loading, it can be considered a "load-bearing bone" that presents similarities to the "weight-bearing bones" from the mechanical standpoint. Mandibular stiffness and strength, including the mechanical properties and distribution of bony tissue, are important because mandibular deformations, stresses and strains occur during static biting and chewing. In order to resist forces, bending and torsional moments, not only the material properties of the mandible but also its geometric design are important16.
The inhibitory effect of GC on mandibular growth and bone biomechanical weakness in the rat when it is administered during the growth phase has been well documented2,8,9,10,12. Most of these studies used three-dimensional peripheral quantitative computed tomography (pQCT) to analyze the biomechanics of the mandible. Densitometric assessment shows that bone mass (the amount of mineralized material present in a bone) offers a certain association with bone strength in vitro18, 19, 20. However, bone strength and bone mass correlated over wide ranges (r = 0.34-0.89; R2 0 0.12-0.79) in in vitro studies, confirming that much of bone strength or fracture risk variance remains unexplained by densitometrically assessed bone mass alone20. Moreover, a lack of correlation between DEXA-assayed BMD (areal bone mineral density) and bone strength or stiffness was reported in studies on bones from rodents of different sizes21. It is thus conceivable that the association between densitometry data and bone fragility may be less reliable than generally supposed for clinical studies20.
To improve our knowledge of the real biomechanical effects of glucocorticoids on the rat mandible, dexamethasone (DTX), a synthetic steroid with predominant glucocorticoid activity, was chronically administered to rats during the growth phase, and the biomechanical behavior of the excised bone was assessed by bending test and peripheral quantitative computed tomographic analysis.
MATERIALS AND METHODS
Two groups (Control = C, and Experimental = E) of 7 female Sprague-Dawley rats aged 23 days were housed in cages under natural light cycle and controlled temperature (23oC), and fed a normal 1.0%Ca/0.8%P standard diet covering nutritional requirements to support normal mandibular growth22. The E group was administered 500 μg/kg per day of DXT (Dexamethasone; Sidus SA, Buenos Aires, Argentina) for 4 weeks, while the C group was injected with the vehicle. Body weight was recorded at the end of treatment in a Mettler P 600 scale to the nearest 0.1 g. Body length was measured from the tip of the snout to the base of the tail. Animals were euthanized by intramuscular ketamine (0.1 ml/100g b.wt) and xylasine (0.02 ml) injection at the end of the treatment period. The hemimandible of each rat was then removed, cleaned of adherent soft tissue, weighed, split at the midline suture, and stored at -20oC wrapped in gauze soaked in Ringer's solution, as recommended by Turner and Burr23. Each bone was thawed at room temperature before analysis. Growth was estimated directly by taking measurements between anatomical points with digital calipers, following Eratalay et al.2 with some modifications24. As an indicator of mandibular size, the mandibular area was calculated from a triangle formed between the most anterior inferior point of the inderdental spine and the angular process, the most posterior point of the angular process, and the most superior point of the coronoid process. The excised left hemimandible was measured by single-energy peripheral quantitative computed tomography on a developed, purpose-built scanner (XCT 960A), Stratec Medizintechnic, Pforzheim, Germany) equipped with an X-ray tube (38.5 kV) as the source of radiation. A region of interest was defined after recording a "scout" scan along the mandibular long axis. The third molar was identified in that scan and the reference line for the crosssectional image was set approximately adjacent to the third molar. The resulting cross-sectional image of the mandible at this site was then encompassed manually by a square region of interest, allowing the software to detect automatically the outside edge of the bone, excluding the molar. This site was chosen because it does not include the incisor root. The threshold density was fixed at a linear attenuation coefficient of 0.900 cm-1 in order to distinguish between cortical and trabecular bone. The fixed threshold refers to mineralized cortical bone. Voxel size was set at 0.148 mm3. Variables related to bone mass, density and architecture were determined as follows: 1) Crosssectional area, as an indicator of the total area of the slice cross-section, 2) Cross-sectional area of cortical bone, as an indicator of cortical bone mass, 3) Volumetric cortical bone mineral content, as an indicator of cortical bone mineral mass, 4) Crosssectional moment of inertia, as an indicative of architectural fitness concerning bending strength, and 5) Volumetric cortical bone mineral density, as an indirect indicator of bone material quality. Cortical area was calculated as the area encompassed by the endosteal and periosteal circumferences. Volumetric bone mineral density was calculated from the single slice cross-sections. The area moment of inertia was calculated along the mesiodistal and craniocaudal axes. The pQCT calculated the cross-sectional moment of inertia as the integral sum of products of each of the infinitesimal fractions into which the whole bone area can be divided and their respective square distances to the reference axis. The reference axis was horizontal and perpendicular to the longest axis. Therefore, we assumed a symmetrical cross-section. The right hemimandible was subjected to a three-point bending mechanical test23 in an Instron test machine (model 4442, Instron Corp., Canton, MA, USA). Each bone was placed on two lower supports (11 mm span) with the lateral aspect facing down and centered along its length. Loads were applied transversally to the bone axis at a point immediately posterior to the posterior surface of the third molar at a rate of 5.00 mm/min. The resulting load / deformation (W/d) curves23 enabled graphic determination of the main structural properties of the mandibles, which essentially measure the resistance to both deformation (stiffness) and fracture (strength). The structural properties are those corresponding to the whole bone as an organ. They are: 1) load at the yielding point (Wy, represents the end point of the elastic deformation of the bone and defines a threshold about which unrecoverable permanent deformation occurs (plastic deformation), marking the initiation of damage accumulation with the first appearance of the first microcracks that occur on the periosteal surface of the bone; it is a measure of the bone strength); 2) structural stiffness (represents the slope of the linear or elastic phase of the W/d curve and is a measure of the resistance of the bone to deformation, or bone rigidity); 3) structural strength (Wf, represents the value of the load at fracture and expresses directly the resistance of the whole bone to fracture). The data are expressed as mean } standard error (SEM). The tomographic "Bone Strength Index" (BSI) was estimated as the product of CSMI and vCtBMD, Statistical analyses were performed with the t-test using GraphPad Prism Software. The experiment was conducted in accordance with the principles outlined in the European Convention for the Protection of Vertebrate Animals used for Experimental and Other Scientific Purposes, and approved by the University of Buenos Aires Ethics Committee.
RESULTS
As expected, E rats failed to attain normal weight gain compared with C, age-matched rats (Fig. 1). The 51.3 % reduction in final body weight found in DXTtreated rats was accompanied by a significant, 20.6% reduction in body length. Like body size, hemimandible weight and area (an index of mandibular size) were significantly lower (27.3 % and 9.7 %, respectively) in E than in C rats at the end of the experimental period (Fig. 1). Fig. 2 shows the structural properties of the mandible, as derived from the slope of the load/deformation curve in the linear region of the elastic behavior. The values for the fracture load, yielding load, and structural stiffness were significantly (P < 0.001) lower (36%, 40% and 38%, respectively) in DXT-treated than in untreated rats, while the deformation at yielding was 20% higher in the former than in the latter. The "yielding load / fracture load ratio" did not differ significantly between E and C groups (0.55 0.02 vs. 0.50 0.02, P > 0.05), indicating that the elastic and plastic components of the load /deformation curve were not altered by treatment. The results of bone density and bone-cross sectional area as measured by pQCT are summarized in Fig. 3. I. Consistently with the effects on mechanical properties, the cross-sectional parameters of the mandible were reduced. Cross-sectional, cortical and trabecular areas were significantly lower (between 20% and 30%) in DXT-treated than in control rats. All the other cortical bone parameters including the density (vctBMD, 7%), mineral content (vctBMC, 43%), and cross-sectional moment of inertia (xCSMI, 49%) were significantly lower in the DXT-treated than in the untreated group.
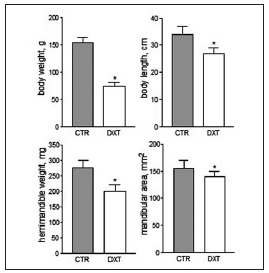
Fig. 1: Morphometric data (Mean } SEM) from untreated (CTR) and dexamethasone-treated (DXT) rats. Asterisks above bars indicate p < 0.05..

Fig. 2: Morphometric data (Mean } SEM) from untreated (CTR) and dexamethasone-treated (DXT) rats. Asterisks above bars indicate p < 0.05..
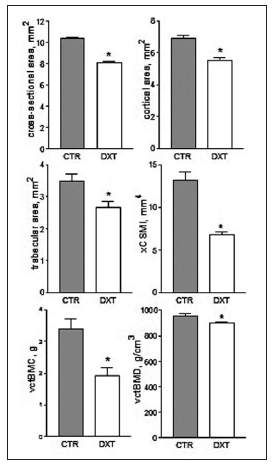
Fig. 3: Morphometric data (Mean } SEM) from untreated (CTR) and dexamethasone-treated (DXT) rats. Asterisks above bars indicate p < 0.05..
DISCUSSION
This study demonstrated that one-month treatment of rats during their growth phase with 500 μg/kg of DXT administered daily impaired body mass growth in general and, directly related to the main purpose of the study, mandible growth and the biomechanical behavior of the bone. The corticosteroid dose chosen has been shown in a previous study7 to induce negative effects in geometric properties and bone material quality of the femur, both of which play a significant role in determining the deleterious biomechanical consequences seen.
When the effect of DTX on the structural biomechanical properties of the mandible, considering the bone as a whole entity, were analyzed directly through the mechanical bending test, it was observed that the corticosteroid decreased the external load (yielding load) necessary to induce unrecoverable deformation of the bone, probably due to the appearance of microcracks on its periosteal surface. At this point, the deformation of the bone was 20% higher than that in the control bone. When the external load applied to the mandible was increased, it was also found that the microcracks progressed until the mandible fractured. Fracture occurred in the treated bone (fracture load) at a load level that was about 60% lower than that needed to fracture an untreated control bone. The load at yielding showed a very similar decrease percentage. As both parameters are very good indicators of bone strength, it seems acceptable to conclude at this point of the discussion that DXT markedly reduced the structural "strength" of the rat mandible under these conditions. The resistance of the bone to deformation in elastic conditions was also negatively influenced by DXT: the structural stiffness, which is an indicative predictor of bone rigidity, was about 60% lower in treated rats than in controls. By considering the results for mandibular structural strength and stiffness together, it is evident that the treated bone was weaker than that of untreated animals and, therefore, structurally incompetent. In the pQCT analysis, DXT administration reduced the total area of the slice cross-section (-21.2%), the cortical bone mass (-20%), the trabecular bone mass (-30%), the cortical bone mineral mass (-43%), the architectural fitness concerning bending strength (-49%), and the volumetric cortical bone mineral density (-6%). Analysis of the morphometrical and geometrical properties of the mandible by the two methods outlined showed that the DXT-treated bone was smaller than the untreated bone, with a significant reduction in cross-sectional area, cortical and trabecular bone mass and architectural design (xCSMI). At this point in the discussion, it is reasonable to conclude that the mandible of the DXT-treated growing rat was weaker than the untreated one because of its smaller bone mass and cross-sectional area, and inadequate spatial distribution of the resistive material over its cross-section.
However, the pQCT assessment of vCtBMD in this experiment allowed determination of the actual bone mineral concentration in cortical tissue. Its significant decrease induced by DXT administration could be taken as evidence of a negative effect of the glucocorticoid on a poor indicator of bone material quality in terms of mineralization, because other indicators of bone quality, such as tissue composition, amount of secondary mineralization, collagen cross-linking, mineral composition, particle size and distribution, presence of microdamage, cannot be estimated tomographically. In fact, vCtBMD is directly proportional to bone material stiffness or E, with a coefficient of determination ranging from 18% to 27%25.
Ferretti et al.20 showed in the rat femur that the product of the pQCT-assessed CSMI and vCtBMD, called BSI (Bone Strength Index) is an accurate and precise indicator of the actual mechanical quality of rat long bone tested by bending. It is interesting to note that in the rat mandible in our study, the BSI calculated and bone strength measured (fracture load) and the structural stiffness were 56%, 54%, and 52% lower than control values, respectively, in response to DXT treatment. This reinforces the concept that BSI is a precise indicator of bone strength. Ferretti et al.25 have estimated that the index is predictive of the actual fracture load with about 89% determining power. It should be noted, however, that the mechanical quality of a whole bone can only be directly assessed by destructive means, such as the one used in this study. The biomechanical response of the mandible to DTX was similar to that found in the femoral shaft in our previous study7 and suggests that corticosteroids exert a combined, negative action on bone geometry (mass and architecture) and volumetric mineral density of cortical bone, which would express independent effects on both cellular (material quality) and tissue (cross-sectional design) levels of biological organization of the skeleton in the species13.
ACKNOWLEDGMENTS
This study was supported by research grants from the University of Buenos Aires (UBACyT 20020100100389 and 20020100100039). RMA and CEB are Career Investigators from National Research Council (CONICET)
1. Loeb JN. Corticosteroids and growth. N Engl J Med 1976; 295:547-552. [ Links ]
2. Eratalay YK, Simmonds DJ, El-Mofty SK, Rosenberg GD, Nelson W, Haus E, Halberg F. Bone growth in the rat mandible following everyday or alternate-day methylpredisolone treatment schedules. Arch oral Biol 1981; 26:769-777. [ Links ]
3. Ortroft G, Oxlund H. Reduced strength of rat cortical bone after glucocorticoid treatment. Calcif Tissue Int 1988; 43:376-382. [ Links ]
4. Karabelios Cs, Horvath Cs, Hollo I, Csaba G. Effect of neonatal glucocorticoid treatment on bone mineralization of adult nontreated, dexamethasone-treated or vitamin D3- treated rats. Gen Pharmacol 1998; 31:789-791.
5. Li M, Shen Y, Halloran BP, Baumann BD, Miller K, Wronski TJ. Skeletal response to corticosteroid deficiency and excess in growing male rats. Bone 1996; 19:81-88. [ Links ]
6. Canalis E, Delany AM. Mechanisms of glucocorticoid action in bone. Ann NY Acad Sci 2002; 966:73-81. [ Links ]
7. Ferretti JL, Gaffuri O, Capozza R, Cointry G, Bozzini C, Olivera MI, Zanchetta JR, Bozzini CE. Dexamethasone effects on mechanical, geometric and densitometric properties of rat femur diaphyses as described by peripheral quantitative computerized tomography and bending tests. Bone 1995; 16:119-124. [ Links ]
8. Fujita Y, Nishioka T, Kinjo K, Maki K. Influence of prednisolone on craniofacial and long bones in growing rats: a cephalometric and peripheral quantitative computed tomographic analysis. Ped Dent J 2007; 17:107-117. [ Links ]
9. Kimura E, Nishioka T, Hasegawa K, Maki K. Effect of biphosphonate on the mandible of rats in the growing phase with steroid-induced osteoporosis. Oral Dis 2007; 13:544-549. [ Links ]
10. Fujita Y, Konoo T, Maki K. Short-term etidronate treatment prevents glucocorticoid-induced bone debility on the mandible in growing rats. Orthod Craniofac Res 2008; 11:187-195. [ Links ]
11. Fujita Y, Watanabe K, Uchicanbon S, Maki K. Effects of risedronate on cortical and trabecular bone on the mandible in glucocorticoid-treated rats. Am J Orthod Dentofacial Orthop 2011; 139:267-277. [ Links ]
12. Bouvard B, Gallois I, Legrans E, Audran M, Chappard D. Glucocorticoids reduce alveolar and trabecular bone in mice. Joint Bone Spine 2013; 80:77-81. [ Links ]
13. Frost HM. The intermediary organization of the skeleton.. Boca Raton, FL, USA CRC Press.1987 [ Links ]
14. DiGirolamo DJ, Kiel DP, Esser KA. Bone and skeletal muscle: neighbors with close ties. J Bone Miner Res 2013; 28:1509-1518. [ Links ]
15. Cianferotti L, Brandi ML Muscle-bone interactions: basic and clinical aspects. Endocrine 2014; 45:165-177. [ Links ]
16. van Eijden E. Biomechanics of the mandible. Crit Rev Oral Biol Med 2000; 11:123-136. [ Links ]
17. Mavropoulos A, Rizzoli R, Ammamm P. Different responsiveness of alveolar and tibial bone loss stimuli. Bone Miner Res. 2000; 22:403-407. [ Links ]
18. Dalen N, Hellstrom LG, Jacobson B. Bone mineral content and mechanical strength of the femoral neck. Acta Orthop Scand 1976; 47:503-508. [ Links ]
19. Beck TJ, Ruff CB, Warden KE, Scott FW, Rao GU. Predicting femoral neck strength from bone mineral data. A structural approach. Invest Radiol 1990; 25:6-18. [ Links ]
20. Ferretti JL, Capozza RF, Zanchetta JR. Mechanical validation of a tomographic (pQCT) index for noninvasive estimation of rat femur bending strength. Bone 1996; 18: 97-102. [ Links ]
21. Ferretti JL, Spiaggi EP, Capozza R, Cointry G, Zanchetta JR. Interrrelationship between geometric and mechanical properties of long bones from three rodent speciews with very different biomass. Phylogenetic implications. J Bone Miner Res 1992; 7(S2):5423-5425. [ Links ]
22. Bozzini C, Barcelo AC, Alippi RM, Bozzini CE. The concentration of dietary casein required for normal mandibular growth in the rat. J Dent Res 1989; 68:840-842. [ Links ]
23. Turner CH, Burr DB. Basic biomechanical measurements of bone: a tutorial. Bone 1993; 14:595-608. [ Links ]
24. Alippi RM, Meta MD, Olivera MI, Bozzini C, Schneider P, Meta IF, Bozzini CE. Effect of protein-energy malnutrition in early life on the dimensions and bone quality of the adult rat mandible. Arch Oral Biol 2002; 47:47-53. [ Links ]
25. Ferretti JL. Noninvasive assessment of the architecture and biomechanical properties in animals and humans employing pQCT technology. J Jpn Soc Bone Morphom 1997; 7:115-125. [ Links ]














