Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Acta Odontológica Latinoamericana
versão On-line ISSN 1852-4834
Acta odontol. latinoam. vol.29 no.2 Buenos Aires set. 2016
ARTÍCULOS ORIGINALES
Immunolocalization of the TGFB1 system in submandibular gland fibrosis after experimental periodontitis in rats
Candela R. González1, Mariano A. R. Amer2, Alfredo D. Vitullo1, Silvia I. González-Calvar3, María I. Vacas4
1 Research Center of Biomedical, Biotechnology, Environmental and Diagnostic Studies, Maimónides University, Buenos Aires, Argentina.
2 Department of Anatomy, School of Dentistry, University of Buenos Aires, Buenos Aires, Argentina.
3 Reproductive Molecular Endocrinology Laboratory, Institute of Experimental Biology and Medicine (IBYME-CONICET), Argentina.
4 University Institute, Italian Hospital, Buenos Aires, Argentina.
CORRESPONDENCE Dr. Mariano Axel Ramon Amer.Catedra de Anatomia, Facultad de Odontologia, UBA. Marcelo T. de Alvear 2142 1B, (C1122AAH), Buenos Aires, Argentina. marianoamer@gmail.com
ABSTRACT
Saliva is the first barrier to entry of bacteria and viruses into the body. The submandibular glands (SMG) contribute to the maintenance of oral health and regulation of immune/ inflam matory responses. Previous studies suggest that transforming growth factor beta 1 (TGFB1) may contribute to salivary gland fibrosis but the expression of the TGFB1 system in the SMG has not been elucidated. Thus, the aim of this study was to analyze in rat SMG the immunolocalization of TGFB1 and its specific receptors ALK5 (profibrotic) and ALK1 (proproliferative) and the coreceptor endoglin (EDG) in a bilateral experimental periodontitis (EP) model (cotton thread ligature around the neck of the first lower molars) for 1 and 6 weeks. Fixed SMG were embedded in paraffin and serially cut for routine hematoxylin–eosin staining for histological analysis or immunohistochemical techniques by diaminobenzidine detection. SMG histology from animals with EP showed timedependent structural changes involving marked reduction in the height of the contoured ducts, cell destruction, loss of secretory granules, periductal congestion and excess connective tissue surrounding these ducts indicative of a fibrotic process, compared to control SMG. TGFB1, ALK5 and ALK1 receptors and the coreceptor EDG were mainly immunolocalized in ductal cells and in the fibrotic areas in EP groups. The expression of the profibrotic ALK5 receptor was increased in areas of fibrosis in SMG of animals with EP. In SMG of rats with EP, the localization of the TGFB1 specific receptors in the ducts and cells from fibrotic areas, due to the expression of TGFB1 in the surrounding areas, might indicate paracrine and autocrine actions exerted by TGFB1 via its specific receptors. The results of this study suggest that TGFB1 promotes fibrosis, inducing cell proliferation via ALK1 and EDG receptors and stimulates fibrosis relatedprocesses via ALK5 receptor, which could lead to abnormal secretor activity of the SMG during periodontal disease.
Key words: Submandibular gland; Transforming Growth Factor beta1; Activin Receptors; Fibrosis; Periodontitis.
RESUMEN
Inmunolocalización del sistema TGFB1 en fibrosis de la glándula submandibular bajo periodontitis experimental en ratas
La saliva es la primera barrera para la entrada de bacterias y virus en el cuerpo. Las glándulas submandibulares (GSM) contribuyen al mantenimiento de la salud oral y a la regulación de las respuestas inmunoinflamatorias. Estudios previos sugieren que el factor de crecimiento transformante beta 1 (TGFB1) puede contribuir a la fibrosis de las glándulas salivales, pero la expresión y localización del sistema TGFB1 en las GSM no ha sido dilucidada. El objetivo del presente trabajo fue analizar por inmunohistoquímica en las GSM de ratas la expresión de TGFB1 y sus receptores específicos ALK5 (profibrótico) y ALK1 (proproliferativo) y el coreceptor endoglina (EDG) en un modelo de periodontitis bilateral experimental (PE) (hilo de algodón alrededor del cuello de los primeros molares inferiores) durante 1 y 6 semanas. Las GSM fueron fijadas y embebidas en parafina para realizar cortes seriados los cuales se tiñeron con hematoxilinaeosina para analizar la histología o se procesaron para realizar la técnica de inmunohistoquímica mediante detección con diaminobenzidine. La histología de las GSM de animales con PE reveló cambios estructurales tiempo dependientes, con una marcada reducción de la altura de los conductos, destrucción celular, pérdida de gránulos secretores, congestión periductal y exceso de tejido conectivo que rodea los conductos, indicando un proceso de fibrosis respecto de las GSM de animales control. TGFB1, ALK5 y ALK1 y el coreceptor EDG fueron principalmente inmunolocalizados en las células que forman los ductos y en las áreas de fibrosis en los grupos con PE. La expresión del receptor profibrótico ALK5 se incrementó en las áreas de fibrosis en GSM de animales con PE. En GSM de ratas con PE, la localización de los receptores específicos de TGFB1 en las células de los conductos y áreas de fibrosis, junto con la expresión de TGFB1 en las áreas circundantes, podría indicar acciones paracrinas y autocrinas ejercidas por TGFB1 a través de sus receptores específicos. Los resultados de este estudio sugieren que TGFB1 podría inducir un proceso de fibrosis promoviendo la proliferación celular a través de los receptores ALK1 y EDG, y favoreciendo procesos relacionados con la fibrosis a través de su receptor ALK5, lo que conduciría a una actividad secretora anormal de la GSM durante la enfermedad periodontal.
Palabras clave: Glándula submandibular; Factor de crecimiento transformante beta1; Receptores de activina; Fibrosis Periodontitis.
INTRODUCTION
Periodontal diseases have been well described and characterized according to different degrees of inflammation, eventually leading to the destruction of teethsupporting tissue. Periodontitis is characte rized by chronic activation of the immune response and synthesis of several cytokines and proteases. Cytokines induce alterations in the cellular metabolism of the connective tissue concomitant with the destruction of the periodontal tissue and tooth loss1. The pair of submandibular glands (SMG) is involved in the maintenance of the oral health and regulation of immune/inflammatory responses2. The SMG has a ductal structure that opens into the oral cavity with secretory end pieces, the mucous and serous acini, producing saliva3. A timedependent link has been observed between experimental periodontitis (EP) and SMG activity, mediated by systemic and neural mechanisms. Initially, the EP stimulates glandular secretion, while the progression of the disease affects the normal function of the gland, leading to histological and biochemical changes, including an inflammatory response and reduced secretion4,5.
There is growing evidence showing the relation between cytokines and the pathological fibrotic process of different tissues, characterized by the development of excess fibrous connective tissue, as a consequence of a tissue repair process that triggers an increase in the production and deposition of extracellular matrix. Among these cytokines, the transforming growth factor beta 1 (TGFB1) plays an important role. The TGFB1 inhibitory and stimulatory properties have been described in the regulation of cellular homeostasis in both physiological and pathological stages6-9. The effects of TGFB1 are exerted via specific type I and type II serine/threonine kinase receptors. Type II receptor (TGFBRII) transphosphorylates and activates type I receptor (TGFBRI). Two TGFBRI have been described: activin receptorlike kinase 1 (ALK1), which signals via phosphorylation of Smads 1/5, and activin receptorlike kinase 5 (ALK5), which signals via phosphorylation of Smads 2/310. In line with this, endoglin (EDG) appears to be a key coreceptor that contributes to an efficient transduction pathway of ALK111. In this context, it has been observed that deregulation mechanisms of TGFB1 appear to be involved in the progression of diseases such as cancer and autoimmune fibrosis6. However, the expression of the TGFB1 system in the SMG affected by periodontal disease is unknown. The aim of this study was to analyze the localization of TGFB1 and its specific receptors ALK5 and ALK1 and the coreceptor EDG in rat SMG after 1 and 6 weeks of EP induction.
MATERIALS AND METHODS
Animals
Adult male Wistar rats from our own colony with initial body weight 220-250 g were randomly divided into 4 groups (n=8 animals per treatment): (1) 1week control, (2) 1week bilateral EP, (3) 6week control, (4) 6week bilateral EP. They were kept in group cages in an animal room with a photoperiod of 12 h light (07.00–09.00 h), room temperature 22–25° C, humidity: 52–56% and fed standard Purina chow pellets and tap water ad libitum. All experiments were performed following the National Institute of Health guidelines for the care and use of laboratory animals (NIH 8523, revised in 1985) and protocols were approved by the Ethical Commission of the School of Dentistry, University of Buenos Aires.
Induction of experimental periodontitis
EP was performed as previously reported5. EP was induced under general anesthesia with a mixture of 2% xylazine hydrochloride (5 mg/kg; i.p.) and 5% ketamine hydrochloride (50 mg/kg; i.p.). A cotton thread ligature was placed around the neck of both first lower molars (bilateral EP) and served as a retention device for oral microorganisms. The ligature was pushed into the gingival sulcus and left in place until sacrifice (1and 6 weeks post surgery). In the 6 week EP treatment, the thread was renewed every 2 weeks in order to prevent the ligature from coming out of the sulcus, thereby not meeting the desired objective. The SMG and submandibular lymph nodes were removed and weighed (mg), after which they were fixed in 4% PFA for histological and immunohistochemical studies.
SMG histology and immunohistochemistry
The tissues mounted in paraffin were cut at 5μm sections, dewaxed in xylene and rehydrated in graded alcohols. For each specimen, at least 3 to 5 slides were stained with hematoxylin/eosin for general histological inspection. Two specialists reviewed all the SMG samples. For immunohisto chemical analysis, endogenous peroxidase activity was inhibited in tissue sections using 0.5% v/v H2O2/methanol. Sections were blocked for 1 h with 15% normal goat serum in phosphatebuffered saline (PBS) and then incubated overnight at 4°C with primary antibody (1:100 diluted rabbit antiTGFB1, sc146; 1:100 diluted rabbit anti TGFB RI [V22], sc402; 1:100 diluted rabbit anti TGFB RI [T19], sc398; 1:100 diluted rabbit antiendoglin, sc20632, Santa Cruz Biotechnology, Inc., USA). After 3 rinses in PBS, sections were incubated for 1h at room temperature with the appropriate 1:200 diluted biotinylated secondary antibody (Vector Labs, UK). After further washing in PBS, sections were incubated with 1:100 diluted streptavidin– peroxidase complexes (ABC kit, Vector Labs, UK). Development of peroxidase activity was achieved with 0.05% w/v 3,3diaminobenzidine and 0.1% v/v H2O2 in Tris–HCl. Negative controls were processed simultaneously by omitting the primary antibody or preabsorbing the primary antibody with specific synthetic peptides.
Statistical Analysis
Mean and standard error (SEM) were calculated and the InfoStat Software (version 2011, developed by Statistics Department, National University of Cordoba) was used for calculating differences between 2 groups using Student´s t test. A pvalue of less than 0.05 was considered significant.
RESULTS
Histology of the SMG in rats with bilateral EP
The weight of the SMG and submandibular lymph nodes in the EP groups was significantly higher than control SMG in both 1and 6week treatments (p<0.05) (Fig. 1A). The histology of the SMG of animals with 1and 6 week EP showed alterations of the epithelial architecture and ducts, partial loss of material granular secretion and periductal edema, as well as an excess in the connective tissue surrounding the ducts, indicative of a fibrotic process compared to the control SMG (Fig. 1B).

Fig. 1: Weight of SMG and submandibular lymph nodes (A) and general histology of SMG (B) in animals with 1 and 6week EP. (B) Note the SMG of periodontitis groups with fibrotic areas surrounding ducts (red arrows). Data are plotted as mean ± SEM (n=6). *p<0.05 different from control group. EP: experimental periodontitis; D: duct; MA: mucous acini; SA: serous acini.
Detection of TGFB1, ALK5, ALK1 and coreceptor EDG in SMG from rats with EP
TGFB1 and its specific receptors ALK1 and AKL5 and coreceptor EDG were immunolocalyzed in the cell cytoplasm of the ducts of control SMG and SMG from animals with 1 and 6 week EP (Fig. 2). TGFB1, ALK1 and EDG were localized in the apical zone and nearest to the lumen while ALK5 showed diffuse localization within cell cytoplasm (Fig. 2).
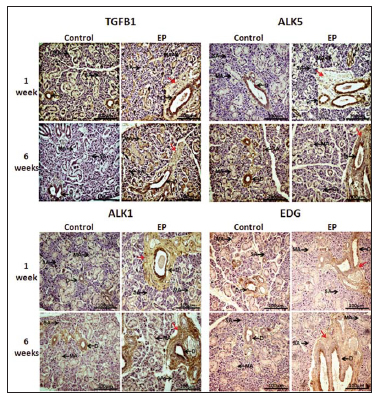
Fig. 2: Immunolocalization of the TGFB1 system in SMG from animals with 1and 6week EP. Note that fibrotic areas (red arrows) of the SMG of periodontitis groups are intensively immunostaned with TGFB1. These areas were also immunopositive for ALK5 and ALK1 receptor and coreceptor EDG, particularly, in 6week EP. EP: experimental periodontitis; D: duct; MA: mucous acini; SA: serous acini; EDG: endoglin.
The expression of TGFB1 and its specific receptors was also predominantly detected in the cytoplasm of cells from the fibrosis areas surrounding the ducts in the affected SMG from animals with 1and 6week EP (Fig. 2). TGFB1 was also detected in mucous acinus (MA) and predo minantly observed only in SMG of EP groups. Con cerning the expression of ALK5, most of the cells from fibrosis areas were immuno positive for this receptor in animals with 6 week EP while the localization of ALK5 was detected in cell clusters in animals with 1week EP (Fig. 2). Serous acinus (SA) showed no immunos taining for ALK5 and only a few immunopositive MA were observed in SMG from all treatment groups. The expression of ALK1 and its coreceptor EDG was mainly observed in cells of fibrosis areas with intense immunos taining in the affected SMG from animals with 1and 6 week EP (Fig. 2). Some immuno positive MA for ALK1 and EDG was observed in the SMG from EP treatment groups.
DISCUSSION
TGFB1, a profibrotic cytokine, participates in diverse biological processes including inflammation, fibrosis, tissue regeneration and epithelialmesenchymal transition6,12-15. In this study, we followed the expression of TGFB1 and its specific receptors ALK5 and ALK1 and the coreceptor EDG in normal SMG and in SMG after 1 and 6 weeks of induction of EP in adult rats. To our knowledge, this is the first report of a global analysis of the expression of the TGFB1 system in SMG in EP conditions.
Normal SMG secretor activity is essential because saliva is the first barrier to entry of bacteria into the body, so changes in secretion are important in the onset and progression of oral infectious processes16. Using a 1and 6 week EP model, we observed histological changes in the SMG, mainly including the appearance of large fibrotic areas that could affect the normal secretor function of the gland. We detected alterations of the epithelial architecture as well as of the ducts, partial loss of material granular secretion and excess in the connective tissue surrounding the ducts indicative of a fibrotic process in SMG after induction of EP. Moreover, SMG and lymph nodes weighed more in animals with EP than in control animals regardless of the duration of the treatment. This may be explained by the fact that EP produced severe structural changes involving a marked reduction in the height of the granular convoluted ducts, cell destruction, loss of secretory granules, increased fibrosis and periductal congestion.
Under these experimental treatments, there is an excess of immunopositive cells for TGFB1 in the connective tissue surrounding the ducts, indicative of a fibrotic process. In line with this, Hall et al.17 have reported an increase in TGFB1 expression in salivary glands leading to hypofunction of the tissue due to the replacement of normal glandular parenchyma with interstitial fibrous tissue. We recently reported altered function of the SMG, including partial loss of secretor granular material with reduced salivary secretion and periductal oedema in EP treatments5. Concerning the type of the TGFB1 receptor, it has been shown that the presence of the coreceptor EDG promotes TGFB1 signaling via ALK1 receptor leading to cell proliferation and attenuates the ALK5 signaling that leads to cell cycle arrest 9,18. There is also growing evidence indicating that in pathological conditions, TGFB1 stimulates fibrosis through deposition of extracellular matrix and epithelialmesenchymal transition via ALK5 receptor and activation of Smad2/3 19-21. In the present study, the expression pattern of ALK1 and its coreceptor EDG was similar and remained constant in the fibrotic areas of SMG in all EP treatments. This could indicate that TGFB1 induces a proliferative response in the connective tissue surrounding the ducts, stimulating the fibrotic process. In line with this, the quantity of cells in fibrotic areas expressing ALK5 increased in SMG concomitant with the days of the EP treatment, which may reflect a fibrotic process that becomes more aggressive with the duration of the periodontitis.
In conclusion, to our knowledge, this is the first report of the expression and localization of TGFB1 and its receptors ALK5 and ALK1 and coreceptor EDG in the SMG. We postulate that TGFB1 would be a key factor in SMG during periodontitis condi tions, probably stimulating tissue damage, fibrosis and morphological alterations associated to abnormal secretor activity. The SMG of animals with EP appears to be an interesting model for studying diverse mechanisms that promote gland fibrosis exerted by TGFB1. Further insights into TGFB1 signaling in SMG could identify novel therapeutic targets for salivary gland fibrosis, whether or not associated to periodontal disease.
ACKNOWLEDGEMENT
The authors acknowledge the collaboration of Ricardo Orzusa, technical assistant, Department of Biochemistry, School of Dentistry, University of Buenos Aires, Argentina.
1. Niculescu EA, Crăiţoiu MM, Baniţă MI, Drăghici E, Crăiţoiu Ş. The involvement of TGFβ1 and CTGF in regional gingival overgrowth. Rom J Morphol Embryol 2012; 53:143-150.
2. Sabadini E, Berczi I. The submandibular gland: a key organ in the neuroimmunoregulattory network? Neuroimmunomo dulation 1995; 2:184-202. [ Links ]
3. Holmberg KV, Hoffman MP. Anatomy, biogenesis and regeneration of salivary glands. Monogr Oral Sci 2014; 24:113. [ Links ]
4. Vacas MI, Amer M, Chiarenza AP, Luchelli MA, Mandalunis PM, Elverdin, JC. Influence of submandibulectomy on alveolar bone loss in rats. J Periodontol 2008; 79:1075-1080. [ Links ]
5. Amer M, Elverdin JC, Fernandez Solari J, Medina V, Chiarenza AP, Vacas MI. Reduced methacholineinduced submandibular salivary secretion in rats with experimental periodontitis. Arch Oral Biol 2011; 56:421-427. [ Links ]
6. Ingman WV, Robertson SA. Defining the actions of transforming growth factor beta in reproduction. Bioessays 2002; 24:904-914. [ Links ]
7. Ingman WV, Robertson SA. The essential roles of TGFB1 in reproduction. Cytokine Growth Factor Rev 2009; 20:233-239. [ Links ]
8. Gonzalez CR, Matzkin ME, Frungieri MB, Terradas C, Ponzio R, Puigdomenech E, Levalle O, Calandra RS et al. Expression of the TGFbeta1 system in human testicular pathologies. Reprod Biol Endocrinol 2010; 8:148-159. [ Links ]
9. Gonzalez CR, Vallcaneras SS, Calandra RS, Gonzalez Calvar SI. Involvement of KLF14 and egr1 in the TGFbeta1 action on Leydig cell proliferation. Cytokine 2013; 61:670-675. [ Links ]
10. Feng XH, Derynck R. Specificity and versatility in TGFbeta signaling through Smads. Annu Rev Cell Dev Biol 2005; 21:659-693. [ Links ]
11. Lebrin F, Goumans MJ, Jonker L, Carvalho RL, Valdimarsdottir G, Thorikay M, Mummery C, Arthur HM et al. Endoglin promotes endothelial cell proliferation and TGFbeta/ ALK1 signal transduction. EMBO J 2004; 23:4018-4028. [ Links ]
12. Thiery JP. Epithelialmesenchymal transitions in tumour progression. Nat Rev Cancer 2002; 2:442-454. [ Links ]
13. Kalluri R, Neilson EG. Epithelialmesenchymal transition and its implications for fibrosis. J Clin Invest 2003; 112: 1776-1784. [ Links ]
14. Xu J, Lamouille S, Derynck R. TGFβinduced epithelial to mesenchymal transition. Cell Res 2009; 19:156-172.
15. Woods LT, Camden JM, ElSayed FG, Khalafalla MG, Petris MJ, Erb L, Weisman GA. Increased Expression of TGFβ Signaling Components in a Mouse Model of Fibrosis Induced by Submandibular Gland Duct Ligation. PLoS One 2015; 10:124.
16. Dodds MW, Johnson DA, Yeh CK. Health benefits of saliva: a review. J Dent 2005; 33: 223-233. [ Links ]
17. Hall BE, Zheng C, Swaim WD, Cho A, Nagineni CN, Eckhaus MA, Kathleen CF, Ambudkar IS, et al.Conditional overexpression of TGFβ1 disrupts mouse salivary gland development and function. Lab Invest 2010; 9:543-555.
18. Lebrin F, Mummery CL. Endoglinmediated vascular remodeling: mechanisms underlying hereditary hemorrhagic telangiectasia. Trends Cardiovasc Med 2008; 18:25-32. [ Links ]
19. Kemaladewi DU, Pasteuning S, van der Meulen JW, van Heiningen SH, van Ommen GJ, Ten Dijke P, Aartsma Rus A, ‘t Hoen PA et al. Targeting TGFβ Signaling by Antisense Oligonucleotidemediated Knockdown of TGFβ Type I Receptor. Mol Ther Nucleic Acids 2014; 3:e156.
20. Park JH, Ryu SH, Choi EK, Ahn SD, Park E, Choi KC, Lee SW. SKI2162, an inhibitor of the TGFβ type I receptor (ALK5), inhibits radiationinducedfibrosis in mice. Oncotarget 2015; 6:4171-4179.
21. Smoktunowicz N, Alexander RE, Franklin L, Williams AE, Holman B, Mercer PF, Jarai G, Scotton CJ, et al. The antifibrotic effect of TGFβ/ALK5 inhibition in experimental pulmonary fibrosis is attenuated in the presence of concurrent γherpesvirus infection. Dis Model Mech 2015; 9:1129-1139.














