Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Acta Odontológica Latinoamericana
versión On-line ISSN 1852-4834
Acta odontol. latinoam. vol.29 no.2 Buenos Aires set. 2016
ARTÍCULOS ORIGINALES
Sequential administration of alendronate and strontium ranelate: histomorphometry and bone biomechanics in ovariectomized animals
Diego H. Díaz1, Julieta A. Rodas1, Clarisa E. Bozzini2, Patricia M. Mandalunis1, Natalia D. Escudero1
1 Histology and Embryology Department, School of Dentistry, University of Buenos Aires.
CORRESPONDENCE Dr. Natalia Daniela Escudero Catedra de Histologia y Embriologia, Facultad de Odontologia, UBA Marcelo T de Alvear 2142, 1o Piso sector A, Catedra de Histologia (C1122AAH). nataliaescudero5@yahoo.com.ar
ABSTRACT
Bisphosphonates are the first choice therapy for the pharmaco logical treatment of osteoporosis. Following reports of cases of bisphosphonaterelated osteonecrosis of the jaw and atypical femur fracture, the safety of longterm use of bisphosphonates has been evaluated, resulting in the proposal of strontium as an alternative drug. No experimental study using a sequential administration design has been reported to date. Hence, the aim of this study was to evaluate the effect on bone tissue of ovariectomized rats of administration of alendronate followed by strontium ranelate. Fortyeight female Wistar rats were ovariectomized on day 1 of the experiment. Beginning on day 30, they were administered 0.3 mg/kg/week of alendronate (ALN) or vehicle (VEH) for 8 weeks. Two groups (ALN and corresponding control) were euthanized at this time, and the remaining animals were divided into 4 groups and given 290 mg/kg/day of strontium ranelate (SR) in their drinking water (TW) or only water for 4 months. Experimental groups were: ALN+SR, ALN+TW, VEH+SR, VEH+TW, ALN and VEH. The tibiae and hemimandibles were resected for histomorphometric evaluation, and the right femur was used to perform biomechanical studies. ANOVA and Bonferroni test were applied. Diaphyseal stiffness, maximum elastic load and fracture load increased in animals that received alendronate, regardless of whether or not they received subsequent SR treatment. Fracture load also increased in VEH+ SR versus control (VEH+TW). Subchondral and interradicular bone volumes were significantly higher in animals that received ALN than in those that received vehicle. No difference was observed in cortical area or thickness of the tibia among treatments. The results obtained with the model presented here, evaluating tibial and mandibular interradicular bone, showed that the combination of ALN and SR and administration of ALN alone are equally effective in preventing bone loss associated with ovariectomyinduced estrogen depletion.
Key Words: Bone tissue; Alendronate; Strontium ranelate.
RESUMEN
Administración secuencial de alendronato y ranelato de estroncio: histomorfometría y biomecánica ósea en animales ovariectomizados
Si bien la primera opción terapéutica para el tratamiento farmacológico de la osteoporosis son los bisfosfonatos (BPs), luego de los primeros reportes en 2003 de los casos de osteone crosis de mandíbula asociada al uso de dichas drogas y las fracturas atípicas de fémur, se ha evaluado su seguridad a largo plazo. Además, en aquellos pacientes que no responden al tratamiento con BPs y mantienen elevado el riesgo de fractura, es necesario suspender su administración y alternar con otras drogas. Una de las que se ha utilizado en la clínica luego del tratamiento con BPs es el ranelato de estroncio (SR). Existen varios trabajos clínicos que reportan los efectos de la administra ción secuencial de ambas drogas, aunque estudios experi men tales con un diseño secuencial aun no se han reportado. Por ello el objetivo de este trabajo ha sido evaluar el efecto de la administración secuencial de alendronato, seguido de ranelato de estroncio sobre el tejido óseo de ratas ovariectomizadas. Se utilizaron 48 ratas Wistar hembras de dos meses de edad divididas en 6 grupos de 8 animales cada uno. El día 1 de experiencia todas fueron ovariectomizadas. El día 30 se comenzó con la administración de alendronato (ALN) en una dosis de 0.3 mg/kg/semana o vehículo (VEH) durante 8 semanas. Luego de este período se sacrificaron dos grupos (uno que recibió ALN y su correspondiente control (sólo vehículo). Los cuatro grupos restantes continuaron con ranelato de estroncio (SR) en el agua de bebida durante 4 meses en una dosis de 290 mg/kg/día o sólo agua corriente( TW) Luego de ese período fueron eutanasiados. Así, los grupos experimentales conformados fueron: ALN+SR, ALN+TW, VEH+SR, VEH+TW, ALN y VEH. Para los estudios histomorfométricos se extrajeron ambas tibias y hemimandíbulas; para el estudio biomecánico se utilizó el fémur derecho. Los resultados fueron analizados mediante el test de ANOVA y el test de Bonferroni. Incrementaron significativamente la rigidez diafisaria, la carga elástica límite y la carga de fractura aquellos grupos que recibieron alendronato versus aquellos que no lo recibieron, independientemente del tratamiento posterior con SR. La carga de fractura además fue mayor en el grupo VEH+SR versus el control (VEH+TW). En cuanto al volumen óseo subcondral e interradicular evaluado histomorfométricamente fue significativamente mayor en aquellos animales que recibieron ALN versus aquellos que recibieron vehículo. No se detectaron diferencias entre aquellos grupos que recibieron SR y sus controles. El área y espesor cortical de la tibia no mostraron diferencias entre grupos. Los resultados obtenidos en el modelo estudiado tanto a nivel del volumen óseo subcondral y cortical de la tibia como a nivel del hueso interradicular del maxilar inferior, mostraron que la combinación de ALN con SR y la administración aislada de ALN son igualmente efectivas para prevenir la pérdida ósea causada por la depleción estrogénica de la ovariectomía.
Palabras clave: Tejido óseo; Alendronato; Ranelato de estroncio.
INTRODUCTION
Osteoporosis is a systemic skeletal disease charac terized by low bone mass and structural deterioration of the bone tissue leading to bone fragility and increased risk of fracture. There are currently various options for pharmacological treatment of this common pathology: use of anticatabolic agents (bisphosphonates [BPs], selective estrogen receptor modulators and denosumab), anabolics (peptides of the parathyroid hormone family) or an agent with dual action mechanism (strontium ranelate, SR)1. For decades, BPs have proven to be safe, effective drugs, and are therefore one of the most often used for pharmacological treatment of osteoporosis2,3. However, regarding longterm safety of BPs, adverse effects have been reported which, though rare, are cause for concern: osteonecrosis of the jaw4 and atypical femur fractures5. It should be highlighted that in addition, some patients do not respond to BP treatment and remain at high risk of fracture. Hence, patients who have received BPs for a long time, or who do not respond adequately to the treatment, should stop taking them6,7 and alternate with other drugs. One of the drugs which has been used clinically after treatment with BPs is strontium ranelate (SR)8-11. In general, papers describe better response in bone mineral density (BMD) in BPnaive patients than in patients previously treated with BPs8,9,11. They also report that during the first 6 months’ treatment with SR following treatment with BPs, patients have an attenuated response in BMD, which varies according to the site evaluated (spine, hip or ankle)9. This period during which patients respond less to densito metrically evaluated treatment agrees with other authors’ reports of improvement in the histomorpho metric parameters of the iliac crest at 12 months but not at 6 months after suspending BPs10. BPs are powerful inhibitors of bone resorption and bone turnover general. They become included in the bone matrix on the mineralization fronts due to their great affinity for hydroxyapatite12. Once included, they are not released until subsequent cycles of bone remodeling take place, during the resorption or reversal phase, and can therefore continue to exert their effect for years. SR is a drug composed of ranelic acid and strontium, in which the active component is strontium, which becomes included in the bone matrix at the formation fronts. In cases where BPs and SR are administered sequentially, the inhibitory effect on bone turnover caused by BPs may cause SR to be incorporated less, generating a weaker clinical response in patients.
The literature includes clinical studies such as Rizzoli et al., which used highresolution peripheral quantitative computed tomography to study the effect of ALN and SR administered separately to women with osteoporosis13,14. Other authors studied the effect of sequential administration of these two drugs15 on formation and resorption markers. Still others analyzed the densitometric response to SR of patients who had previously received BP compared to those who had never received BP (BPnaive) 8-11.
Experimental studies have compared the effect of BPs to the effect of SR16,17 or evaluated the combined effect of both drugs administered separately or together,18 but not sequentially. Even though the most frequent clinical situation is sequential administration of BP and SR, to date, the literature contains no experimental evidence of the effect on bone of BP followed by SR. The aim of this study is thus to evaluate the effect of administering alendronate followed by strontium ranelate on bone tissue in ovariectomized rats.
MATERIALS AND METHODS
Experimental design
Fortyeight 2monthold female Wistar rats weighing 180±10 g were used, which were ovariectomized at the beginning of the experiment. They were housed in groups of six animals per cage, with a 12hour light, 12hour darkness cycle, temperature 22±2°C and humidity 5256 %, fed ad libitum (standard balanced feed, Cooperacion, Argentina, which contains 23% protein, 11.4% calcium and 0.50.8% phosphorus) and water. Use, care and treatment of the animals followed National Research Council standards (Guide for the Care and Use of Labora tory Animals, 8th Ed. 2011) and guidelines approved by the Ethics Committee of the School of Dentistry of Buenos Aires University (28/11/201238).
The animals were divided into 6 groups (n=8 per group): Alendronate + Strontium Ranelate (ALN+ SR), Vehicle + Strontium Ranelate (VEH+SR), Alendronate + Water (ALN+TW), Vehicle + Water (VEH+TW), Alendronate (ALN) and Vehicle (VEH), as shown in Fig 1. They were euthanized using a solution based on sodium pentobarbital and sodium diphenylhydantoin (Euthanyle, Laboratorios Brouwer, Argentina), after which both tibiae, right femur and hemimandibles were extracted from each animal.

Fig. 1: Experimental design. One month after ovariectomy (OVX), groups ALN, ALN+SR and ALN+TW were given a weekly i.p. injection of alendronate (Gador SA) in a single 0.3 mg/kg dose diluted in sterile saline, for 8 weeks (8 doses) between 11 a.m. and 12 a.m. The dose was adjusted to each animal according to its weight immediately prior to injection. Groups VEH, VEH+SR and VEH+TW were injected with an equivalent volume of saline over the same period. After two months of weekly alendronate or vehicle injections, groups ALN and VEH were sacrificed. Alendronate or vehicle was suspended in the four remaining groups. Groups ALN+SR and VEH+SR were then administered SR in their drinking water at a dose of 290 mg/kg/day p.o., while groups ALN+TW and VEH+TW were only given tap water. Four months later, all animals were euthanized.
Morphometric analysis
Right femurs (free from any soft tissues by dissection) were weighed on precision scales (E. Mettler, Zurich, Switzerland) immediately after dissection and prior to fixing. Their length was measured using a Verniertype caliper and they were kept for biomechanical analysis. They were preserved by freezing at 70° C without fixing.
Biomechanical analysis
For the threepoint flexural test, the test machine (Instron model 4442, Instron Corporation, Canton, MA, USA) was operated in stroke control at a constant rate of 5mm/min, in order to determine diaphyseal stiffness and maximum elastic load19-21.
Histology and histomorphometry
Decalcified sections
Left tibias and hemimandibles were fixed in formalin buffer for 48 hours and then decalcified in EDTA at pH 7 for one month at room temperature. Left tibias were processed for hematoxylin and eosin staining, and longitudinal sections were made of the proximal epiphysis. An area 1 mm high by 1.5 mm wide beneath the mataphyseal cartilage was analyzed in order to determine subchondral bone volume (BV/TV, %). The area studied included primary and secondary spongy bone (Fig 2). Hemimandibles were processed to obtain histological sections stained with hematoxylin and eosin oriented mesiodistally at the level of the first lower molar. Interradicular bone volume was measured in a zone comprised between a tangent line at the level of root apexes and the space limited by the roots of the first molar (BV/TV, %) (Fig 3). For histomorphometric measurement of sections, digital micrographs were taken with a brightfield photomicroscope (Axioskop 2, Carl Zeiss Jena, Germany) and measured using Image Pro Plus 6.1 software (Media Cybernetics).

Fig. 2: Area evaluated in tibia. An area 1mm high by 1.5 mm wide was marked in the subchondral trabeculae zone of the proximal epiphysis, located in the central zone immediately beneath the growth cartilage.

Fig. 3: Interradicular bone tissue area evaluated. We evaluated the area included between the roots of the first lower molar, apically delimited by a line tangential to both apices. The top right corner shows the position of the molar studied in the mandible.
Nondecalcified sections
Right tibias were embedded in methyl methacrylate. In order to determine cortical thickness (Ct.Th, μm) and cortical area (Ct. Ar, μm2) histomor phometrically, the area to be evaluated was first standardized by means of two transversal cuts at the level of the diaphysis: one at 15 and another at 17 mm from the proximal epiphysis, providing a section 2 mm thick. This section was manually worn down with decreasing grits to provide thin sections which were mounted with Canada balsam. Digital micrographs were taken with epifluorescent microscopy. The full cross section was reconstructed by means of photomontage. Measurements were taken using Image Pro Plus on these photomontages. To measure cortical thickness, 4 parallel lines were drawn perpendicular to the bone surface on each face of the tibia. The 12 values for each section were averaged. To determine cortical area on the same photomontages, the area covered by the medullary canal was subtracted from the total area of the section (Fig. 4). Names and abbreviations follow the recommendations of the American Society for Bone and Mineral Research22, 23.

Fig. 4: Determination of cortical thickness and cortical area. Left: cortical thickness was determined by measuring four lines perpendicular to the endosteal and periosteal surfaces of the three faces of the tibia. Right: cortical volume was determined by measuring total area occupied by the bone tissue and the medullary cavity (area shaded in gray), from which the area occupied by the medullary space (lined area) was subtracted.
Statistical analysis
Results are expressed as arithmetic mean and standard deviation. All results were analyzed using oneway Anova and posthoc Bonferroni. Data were analyzed using Primer software (Mc GrawHill 1992). Values of p lower than 0.05 were consi dered statistically significant. Some para meters were also tested using the hypothesis test for planned comparisons by means of the orthogonal contrast technique (SSPS Statistics, IBM USA). The three contrasts used were: groups receiving alendronate vs. groups receiving vehicle (whether or not they received strontium ranelate), groups receiving alendronate combined with strontium ranelate vs. groups receiving only alendronate and groups receiving only strontium ranelate vs. those not receiving any treatment.
RESULTS
Morphometric analysis
For femur length and weight, it was found that femurs of animals euthanized 7 months after the beginning of the experiment were significantly heavier than those of animals euthanized 3 months after the beginning of the experiment (Table 1).
Table 1. Femur weights and lengths.

Biomechanical analysis
The following values were recorded for diaphyseal stiffness (N/mm): ALN+SR: 398.8±50.1 (n=6), ALN+TW: 421±36.7 (n=8), VEH+SR: 384.8±47.5 (n=6), VEH+TW: 337.0±85.5 (n=8), ALN: 356.4±60.0 (n=4), VEH: 329.0±32.9 (n=4), Anova p=0.059. In addition, orthogonal contrasts detected differences between groups that received alendronate vs. those that did not (ALN+SR and ALN+TW vs. VEH+SR and VEH+SR) (Fig 5). Maximum elastic load (N) showed the same trend: ALN+SR: 111±17.8 (n=6), ALN+TW: 116.1±30.1 (n=8), VEH+SR: 104.8±12.4 (n=6), VEH+TW: 83.4±19.3 (n=8), ALN: 103.0±33.8 (n=4), VEH: 87.7±22.5 (n=4), Anova p=0.043, Bonferroni ALN+TW vs. VEH+TW. Orthogonal contrasts: there are differences between groups that received alendronate vs. groups that did not (ALN+SR and ALN+TW vs. VEH+SR and VEH+SR) (Fig. 5). Fracture load (N): ALN+SR: 165.8±32.7 (n=6), ALN+TW: 170.2±30.1 (n=8), VEH+SR: 164.1± 18.7 (n=6), VEH+TW: 141.7±25.5 (n=8), ALN: 125.5±35.5 (n=4), VEH: 147.7±13.8 (n=4), Anova p=0.02, Bonferroni ALN+SR vs. VEH+TW, ALN+TW vs. ALN+TW and VEH+SR vs. VEH+ TW. Orthogonal contrasts did not detect any differences (Fig. 5).
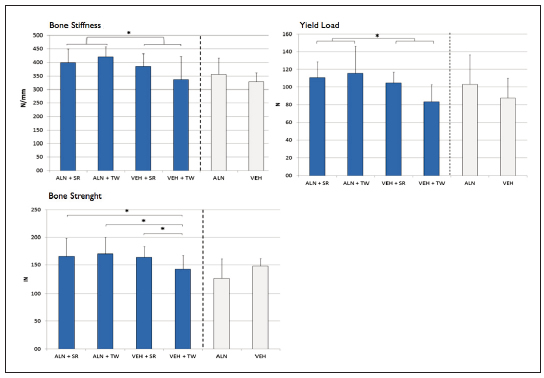
Fig. 5: Biomechanical results. Diaphyseal stiffness showed differences between groups that received ALN (ALN+SR and ALN+TW) versus those that did not (VEH+SR and VEH+TW). Maximum elastic load showed the same tendency as diaphyseal stiffness, with differences between the groups that received ALN (ALN+SR and ALN+TW) versus those that did not (VEH+SR and VEH+TW). Fracture load showed differences between all groups versus the absolute control: ALN+SR, ALN+TW and VEH+SR vs. VEH+TW.
Histology and histomorphometry
The tibia and maxillary sections were evaluated qualitatively. Tibia sections from animals that received alendronate and strontium ranelate (ALN+ SR) or alendronate followed by tap water (ALN+ TW) showed a zone of high trabecular density corresponding to the period during which they received alendronate. Between this highdensity trabecular zone and the growth cartilage, there was a zone with fewer trabeculae, corresponding to the period of time during which the animals received strontium ranelate or only tap water (Fig 6). In addition, giant osteoclasts were observed in all groups that received ALN (ALN, ALN+SR and ALN+TW) (Fig 7). In groups ALN+TW and ALN+SR these osteoclasts were observed both in the highdensity trabecular zone corresponding to the 8 weeks during which they received BP and in recent trabecular zones formed during the BPfree period.

Fig. 6: Subchondral bone volume in tibia. Animals that received ALN ( A and B) had a zone of high trabecular density (1). During the bisphosphonatefree period, they developed a less dense zone (2) between the growth cartilage and the zone with high trabecular density; whether during that period they received strontium ranelate (A) or tap water (B). The animals that received strontium ranelate only (C) had bone volume similar to the control group (D). Bar, 1000 μm.

Fig. 7: Giant osteoclasts. Present in all groups treated with alendronate (ALN+SR, ALN+VEH and ALN). A: partially active giant osteoclast in group ALN+SR in the trabecular zone formed during the period in which alendronate was administered. B: partially active osteoclast in group ALN+VEH, with cytoplasmic heterogeneity. C: osteoclast in group ALN, surrounding a mixed trabecula. Bar, 30 μm.
The same occurred with the apoptotic forms of osteoclasts, which were present in the animals treated with ALN, in both tibia (Fig. 8 A) and maxillae (Fig. 8 B). It was also noted that there were intensely basophilic zones on the bone surface of animals treated with bisphosphonates, both on tibiae and on maxillae, corresponding to apposition lines (Fig 9 A). This was observed both in the group that was euthanized after receiving alendronate for eight weeks, and in the groups which had a BPfree period (ALN+TW and ALN+SR). These basophilic surface areas were also apparent in the interra dicular bone, where reversal lines could also be clearly seen (Fig. 9 B), and were located on the bone matrix delimiting the lacunocanalicular system of some superficial osteocytes (Fig. 9 C and D).

Fig. 8: Apoptosis in osteoclasts. Apoptotic forms of osteoclasts were present in all animals in the groups that received ALN, both in tibia (arrows, A), and in interradicular bone (B). Bar, 20 μm.

Fig. 9: Intensely basophilic zones in animals treated with ALN in tibia and maxillae bone tissue. Appositional lines strongly stained with hematoxylin, in tibia (A), and in the reversal lines of maxillaries (B). Basophilia of bone matrix in bone tissue delimiting part of some superficial osteocyte lacunae (C and D). Bar, 20 μm.
Static histomorphometry
Fig. 10 shows the bone volume results for tibiae and hemimandibles. Tibia cortical thickness (Ct.Th, μm) showed no significant variation among groups: ALN+SR: 545.66±32.16, ALN+TW: 554.06±47.23, VEH+ SR: 534.12±55.78, VEH+TW: 522.65±33.9, ALN: 487.33 ±24.63, VEH: 478.35± 33.56. Anova p=0.58. Cortical area (Ct.Ar μm2): ALN+SR: 5.05±0.47, ALN+TW: 5.02±0.40, VEH+SR: 4.78±0.29, VEH+ TW: 4.74±0.37, ALN: 4.58 ±0.63, VEH: 4.71± 0.20. Anova p=0.34.

Fig. 10: Bone volume in tibias and hemimandibles. For tibia there were statistically significant differences between animals that received ALN versus those that did not (ALN+SR and ALN+TW vs. VEH+SR and VEH+TW; Anova p<0.001, posthoc Bonferroni). For hemimandibles, there were also differences between groups with and without ALN (ALN+SR and ALN+TW vs. VEH+SR and VEH+TW).
DISCUSSION
The results of this study show that administering bisphosphonate (alendronate) followed by strontium ranelate produces the same effect on bone as administering bisphosphonate alone. To date, there are few studies on animals using alendronate and strontium ranelate16-18, and none using them sequentially.
Biomechanics showed that administering alendronate makes femurs stiffer, at the same time increasing maximum elastic load and fracture load, while administering strontium ranelate alone also improves femur fracture load. However, sequential administration of the two drugs showed no difference compared to administration of either bisphosphonate alone or strontium ranelate alone. A paper by Chen et al.17 partially agrees with our findings for long bones, as it reports that administering alendronate to ovariectomized rats improves the biomechanical properties of the femur and increases subchondral bone volume, but it also describes histomorphometric differences between groups that received strontium ranelate and the control, which was not observed in our study. Sun et al.16 used a model of bone loss associated to administration of glucocorticoids and also reported that alendronate and strontium ranelate were equally efficient at preventing bone loss (measured by densitometry and histomorphometry) and at improving biomechanical properties. Interradicular bone showed the same behavior as long bones regarding bone volume, with less marked differences. Considering that in rat subchondral bone there is constant presence of trabeculae produced by endochondral ossification which does not cease throughout the animal’s lifetime, any changes in the bone are more evident in interradicular bone, which has already completed its formation.
Once bisphosphonates are deposited in the bone matrix, they can only be released into circulation by bone resorption performed by osteoclasts. Considering that bisphosphonates affect the osteoclast, and therefore also resorption, the bisphosphonates themselves prolong the time they remain in the bone matrix. It is estimated that after suspending treatment, bone tissue releases quantities of bisphosphonate equivalent to 25% of the dose absorbed while actively receiving the drug24. Considering this aspect of bisphosphonates, in our experimental design we decided that the bisphosphonatefree period should be double the time during which it was administered, with the aim of observing its residual effects. It is worth highlighting that no change in bone volume was found between the group that received alendronate and was immediately euthanized (group ALN) and the groups euthanized after 4 months (ALN+SR and ALN+TW) (statistics not included in this paper), indicating a residual effect of ALN. This effect also was also apparent in the persistence of giant osteoclasts and apoptotic forms of osteoclasts in animals from the groups ALN+SR and ALN+ TW. Both effects are known to be produced by aminobiphosphonates25-30. The persistence of the effects of alendronate deposited in the matrix and the reduction in bone turnover that it produces is the most probable cause of the lack of effect of strontium administered after alendronate. It should be taken into account that strontium is deposited at bone formation sites where there are active BMUs in that phase of the remodeling cycle. As bisphosphonates cause a lower BMU activation frequency, they reduce the sites at which strontium may be deposited, as shown by Wu et al. in the model administering risedronate and strontium ranelate simultaneously, using Electron Probe MicroAnalysis18. Moreover, strontium ranelate is described as a dual agent: anticatabolic and anabolic. Due to the fact that bisphosphonates inhibit remodeling, they may inhibit the anabolic effect of strontium. Although in our study the effects of strontium ranelate alone were subtle compared to the effects of bisphosphonates, they were not observed in the experimental group that received the drugs sequentially (ALN+SR) as a result of alendronate “slowing down” bone tissue. This experimental finding agrees with clinical findings reported in the literature8-11 where bone response to the effect of strontium ranelate administered after alendronate is “attenuated” during the first 6 months. After this time, there is an improvement in densitometric parameters and fracture rate. Nevertheless, this improvement does not catch up with the response in bisphosphonatenaive patients who were treated with strontium ranelate.
Weinstein et al.27 and our group26 were the first to describe giant osteoclasts in patients receiving bisphosphonates or who had received bisphospho nates, in papers published in 2009. This finding is important to histopathological diagnosis, since the histological picture could be mistaken for Paget´s disease, hyperparathyroidism and even bone tumors, in which giant cells are often present. The intensely basophilic zones observed on the bone surface and within the matrix in animals treated with bisphosphonates, both in tibia and maxillae, corresponding to apposition lines and reversal lines, are not a minor finding, considering the role of bone matrix components in various processes such as matrix mineralization and bone resorption31. Osteopontin is a noncollagenous protein synthesized by osteoblasts. During the bone formation stage, osteopontin is the first and last secretion of osteoblasts, and is therefore preferentially deposited on the reversal and appositional lines32. It is a phospho rylated acid protein, and therefore basophilic, one of its functions being to act as a bonding site between osteoclast and bone matrix. Any alteration in matrix composition at the level of the superficial appositional lines can condition osteoclast adhesion. One of the known effects of bisphospho nates is altering the organization of the osteoclast cytoskeleton by interfering in the mevalonate pathway and inhibiting prenylation of small GTPases33. The qualitative changes observed in the bone matrix require further study because they may be another potential point of interference of bisphosphonates with osteoclast adhesion to their bone substrate. Moreover, this topic requires further study to elucidate any potential bisphosphonate actions on the osteoblast lineage.
ACKNOWLEDGEMENTS
This work was supported by Grant UBACyT 20020120 100190BA from the University of Buenos Aires. The authors wish to thank GADOR SA for supplying alendronate; Prof. Dr. Ricardo L Macchi for help with statistics; Ms. Lewicki DVM for collaboration during the experiments; Ms. Mariela Lacave and Ms. Ivana Sanchez Rojas CLT for obtaining and processing histological sections; DDS Sharon Oyhanart for preparing various figures and DDS Lorena Cabirta for help during the drug administration stage of this experiment.
1. Reginster JY, Neuprez A, Dardenne N, Beaudart C, Emonts P, Bruvere O. Efficacy and safety of currently marketed antiosteoporosis medications. Best Pract Res Clin Endocrinol Metab 2014; 28:809-834. [ Links ]
2. National Institute for Health and Clinical Excellence. Alendronate, atidronate, risedronate, raloxifene, strontium ranelate and teriparatide for the secondary prevention of osteoporotic fragility fractures in postmenopausal women. URL: http://www.nice.org.uk/guidance/ta161/resources/guidancealendronateetidronaterisedronateraloxifenestrontiumranelateandteriparatideforthesecondarypreventionofosteo poroticfragilityfracturesinpostmenopausalwomenamendedpdf [ Links ]
3. Salica D, Buceta A, Palacios S, Sanchez A. Consenso Iberoamericano de Osteoporosis SIBOMM 2013. Osteopo rosis: Prevencion, Diagnostico y Tratamiento. Guia para el Diagnostico, Prevencion y Tratamiento de la Osteoporosis para la Argentina. URL: https://www.iofbonehealth.org/sites/default/files/PDFs/National%20Guidelines/Argentine_guidelines_2013QSchurman_y_col1QMedicina2013.pdf [ Links ]
4. Marx RE. Pamidronate (Aredia) and zoledronate (Zometa) induced avascular necrosis of the jaws: a growing epidemic. J Oral Maxillofac Surg 2003; 61:1115-1117. [ Links ]
5. Cheung RK, Leung KK, Lee KC Chow TC. Sequential nontraumatic femoral shaft fractures in a patient on longterm alendronate. Hong Kong Med J 2007; 13:485-489. [ Links ]
6. Whitaker M, Guo J, Kehoe T, Benson G. Bisphosphonates for osteoporosis—where do we go from here? N Engl J Med 2012; 366:2048-2051.
7. Food and Drug Administration. Background document for meeting of Advisory Committee for Reproductive Health Drugs and Drug Safety and Risk Management Advisory Committee. Sep 9. URL: http://www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/Drugs/DrugSafetyandRiskManagementAdvisoryCommittee/UCM270958.pdf. [ Links ]
8. Middleton ET, Steel SA, Aye M, Doherty SM. The effect of prior bisphosphonate therapy on the subsequent BMD and bone turnover response to strontium ranelate. J Bone Miner Res 2010; 25:455-462. [ Links ]
9. Middleton ET, Steel SA, Aye M, Doherty SM. The effect of prior bisphosphonate therapy on the subsequent therapeutic effects of strontium ranelate over 2 years. Osteoporos Int 2012; 23:295-303. [ Links ]
10. Jobke B, Burghardt AJ, Muche B, Hahn M, Semler J, Amling M, Majumdar S, Busse B. Trabecular reorgani zation in consecutive iliac crest biopsies when switching from bisphosphonate to strontium ranelate treatment. PLoS One 2011; 6:e236-38. [ Links ]
11. Brun LR, Galich AM, Vega E, Salerni H, Maffei L, Premrou V, Costanzo PR, Sarli MA et al Strontium ranelate effect on bone mineral density is modified by previous bisphosphonate treatment. Springerplus 2014; 3:676. [ Links ]
12. Fleisch H. Bisphosphonates: a new class of drugs in diseases of bone and calcium metabolism. Recent Results Cancer Res 1989; 116:12-8. [ Links ]
13. Rizzoli R, Laroche M, Krieg MA, Frieling I, Thomas T, Delmas P, Felsenberg D. Strontium ranelate and alendronate have differing effects on distal tibia bone microstructure in women with osteoporosis. Rheumatol Int 2010; 30:1341-1348. [ Links ]
14. Rizzoli R, Chapurlat RD, Laroche JM, Krieg MA, Thomas T, Frieling I, Boutroy S, Laib A, et al. Effects of strontium ranelate and alendronate on bone microstructure in women with osteoporosis. Results of a 2year study. Osteoporos Int 2012; 23:305-315. [ Links ]
15. Lima H, Maia J, Bandeira F. Trajectories of bone remodeling markers and bone mineral density during treatment with strontium ranelate in postmenopausal women previously treated with bisphosphonates. Clin Med Insights Endocrinol Diabetes 2014; 7:7-11. [ Links ]
16. Sun P, Cai DH, Li QN, Chen H, Deng WM, He L, Yang L. Effects of alendronate and strontium ranelate on cancellous and cortical bone mass in glucocorticoidtreated adult rats. Calcif Tissue Int 2010; 86:495-501. [ Links ]
17. Chen B, Li Y, Yang X, Xie D. Comparable effects of alendronate and strontium ranelate on femur in ovariecto mized rats. Calcif Tissue Int 2013; 93:481-486. [ Links ]
18. Wu Y, Adeeb SM, Duke MJ, Munoz Paniagua D, Doschak MR. Compositional and material properties of rat bone after bisphosphonate and/or strontium ranelate drug treatment. J Pharm Pharmaceut Sci 2013; 16: 52-64. [ Links ]
19. Tuner CH, Burr DB. Basic biomechanical measurements of bone: a tutorial. Bone 1993; 14:595-608. [ Links ]
20. Bozzini C, Olivera MI, Huygens P, Alippi RM, Bozzini CE. Longterm exposure to hypobaric hypoxia in rat affects femur crosssectional geometry and bone tissue material properties. Ann Anat 2009; 191:212-217 [ Links ]
21. Hogan HA, Groves JA, Simpson HW. Longterm alcohol consumption in the rat affects cross sectional geometry and bone tissue material properties. Alcohol Clin Exp Res 1999; 23:1825-1833. [ Links ]
22. Parfitt AM, Drezner MK, Glorieux FH, Kanis JA, Malluche H, Meunier PJ, Ott SM, Recker RR. Bone histomorphometry: standardization of nomenclature, symbols, and units. Report of the ASBMR Histomorphometry Nomenclature Committee. J Bone Miner Res 1987; 2:595-610. [ Links ]
23. Dempster DW, Compston JE, Drezner MK, Glorieux FH, Kanis JA, Malluche H, Meunier PJ, Ott SM, et al.Standardized nomenclature, symbols, and units for bone histomorphometry: a 2012 update of the report of the ASBMR Histomorphometry Nomenclature Committee. J Bone Miner Res 2013; 28:21-7 [ Links ]
24. Rodan G, Reszka A, Golub E, Rizzoli R. Bone safety of longterm bisphosphonate treatment. Curr Med Res Opin 2004; 20:1291-1300. [ Links ]
25. Jain N, Weinstein RS. Giant osteoclasts after longterm bisphosphonate therapy: diagnostic challenges. Nat Rev Rheumatol 2009; 5:341-346. [ Links ]
26. Escudero ND, Lacave M, Ubios AM, Mandalunis PM. Effect of monosodium olpadronate on osteoclasts and megakaryocytes: an in vivo study. J Musculoskelet Neuronal Interact. 2009; 9:109-120. [ Links ]
27. Weinstein RS, Roberson PK, Manolagas SC. Giant osteoclast formation and longterm oral bisphosphonate therapy. N Engl J Med 2009, 360:53-62. [ Links ]
28. Jobke B, Milovanovic P, Amling M, Busse B. Bisphospho nateosteoclast: changes in osteoclast morphology and function induced by antiresorptive nitrogencontaining bisphosphonate treatment in osteoporosis patients. Bone 2014; 59:37-43. [ Links ]
29. Morrissey C, Roudier MP, Dowell A, True LD, Ketchanji M, Welty C, Corey E, Lange PH, et al. Effects of androgen deprivation therapy and bisphosphonate treatment on bone in patients with metastatic castrationresistant prostate cancer: results from the University of Washington Rapid Autopsy Series. J Bone Miner Res 2013; 28:333-340. [ Links ]
30. MacWay F, Trombetti A, Noel C, Lafage Proust MH. Giant osteoclasts in patients under bisphosphonates. BMC Clin Pathol 2014; 8:14-31. [ Links ]
31. Davies JE. Bone bonding at natural and biomaterial surfaces. Biomaterials 2007; 28: 5058-5067. [ Links ]
32. McKee MD, Nanci A. Osteopontin at mineralized tissue interfaces in bone, teeth, and osseointegrated implants: ultrastructural distribution and implications for mineralized tissue formation, turnover, and repair. Microsc Res Tech. 1996; 33:141-164. [ Links ]
33. Benford HL, McGowan NW, Helfrich MH, Nuttall ME, Rogers MJ. Visualization of bisphosphonate induced caspase 3 activity in apoptotic osteoclasts in vitro. Bone 2001;28: 465-473. [ Links ]














