INTRODUCTION
Veneers are indirect restorations made with conservative techniques with the aim of harmonizing smiles by restoring the color, shape and adequate function of esthetically compromised teeth. Dental ceramic has excellent properties, including color stability, translucence and high capacity to imitate tooth structure. It is the restorative material with the most similar optical properties to those of teeth 1 . There is currently a wide range of available dental ceramics for indirect restorations, which can be used to make veneers, including sintered or machined feldspathic ceramics, injected or machined glass ceramics, resin matrix ceramics such as resin nano ceramics (Lava Ultimate) and glass ceramic in a resin interpenetrating matrix (Vita Enamic) 2, 3 . Highly translucent ceramic restorations allow passage and dispersion of light, especially when they are thin. Successful clinical treatment therefore depends on the perfect combination of colors between the restoration and the tooth. Many variables are involved, including color of the tooth structure, thickness, color and type of ceramic and resin cement, as well as the translucence of the restorative material as a result of refracted and transmitted light. 4, 5
Light-activated resin-based cement is used for bonding ceramic veneers. Controlling its activation by light makes it possible to optimize the technique and lengthen work time. It also provides color stability because there is no redox reaction of the tertiary aromatic amines that are present in chemically cured materials 4 . It must be considered that light-cured materials may be affected by irradiance of the curing unit. In clinical practice, they may be affected by the type and thickness of the ceramic, as well as by other factors that determine the translucence of the ceramic, such as color, thickness, surface smoothness, polish, and volume ratio between amorphous and crystalline phases 6 . Almeida et al. concluded that light-cured resins (including resin cement, fluid composite and preheated restoration composite) had greater color stability than dual-cured resin 7 . Many other authors (e.g., Marchionatti, Ghavam, Magalhaes) have reported that, due to their polymerization efficacy and high degree of conversion, dual-cured resins do not undergo significant color change, similarly to light-cured resins 8-11 .
The color stability of composite resins depends on intrinsic factors owed to the material’s physicochemical reactions, and extrinsic factors related to diet 12, 13 . Color can change in resin-based materials as a result of aging, and has been studied in vitro using methods such as exposure to UV radiation/condensation, thermocycling and storage in water 14-16 . Accelerated Artificial Aging (AAA) has been shown to be efficient for evaluating the longevity of different materials used in dentistry. AAA simulates the clinical parameters, as far as possible, due to the action of different conditions to which the material is subjected, such as UV light, and continuous alterations in temperature and humidity, and it has been used to evaluate the color stability of dental restorations. Color undergoes change due to degradation of the polymer matrix and unreacted components in the system, as well as the type of initiators, inhibiting agents, polymerization systems, filling types and free double bonds 17, 18 . Moharamzadeh et al. 19 say that residual monomers are the main components released into the environment. Therefore, color changes in the resin cement used for bonding may become visible, affecting the final esthetic appearance of the restoration and leading to failed treatment 14 . The choice of color for each patient has always been a great challenge in dentistry. Dentists have traditionally determined tooth color by comparing it to a sampler of artificial tooth colors. However, it is difficult to unify the perception of a given color by different observers. Variables such as lighting, experience, fatigue of the human eye, and color-blindness interfere with the objective definition of color 20, 21 . Today, instruments that analyze color objectively and quantitively -such as colorimeters, spectrophotometers, spectroradiometers, and digital cameras- can be used to measure tooth color. A spectrophotometer is a standardized scientific colorimetric device for measuring colors which provides information on the reflectance curve as a function of wavelengths throughout the visible range, specifying the perceived color numerically or on a scale 22 . The CIELAB system is a 3-dimensional system that identifies any color in the entire visible spectrum by means of three numbers. The parameter L* represents lightness, with L* equal to 0 being black and L* equal to 100 being white. The parameter a* indicates red to green, with lower values indicating green and higher values indicating red. The parameter b* indicates the position between yellow and blue, with lower valúes corresponding blue and higher valúes to yellow 23, 24 .
Ceramic crown color stability has been thoroughly studied. However, for restorations with thin, translucent veneers, resin color stability too, is of the utmost importance and may affect long-term clinical success. The aim of this study was to determine color change after accelerated artificial aging of different composite resin cements indicated for esthetic veneers.
MATERIALS AND METHODS
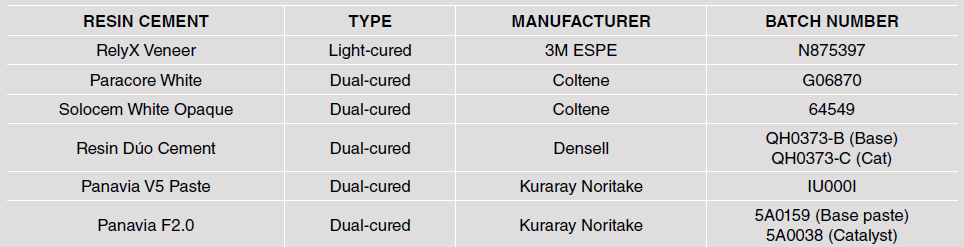
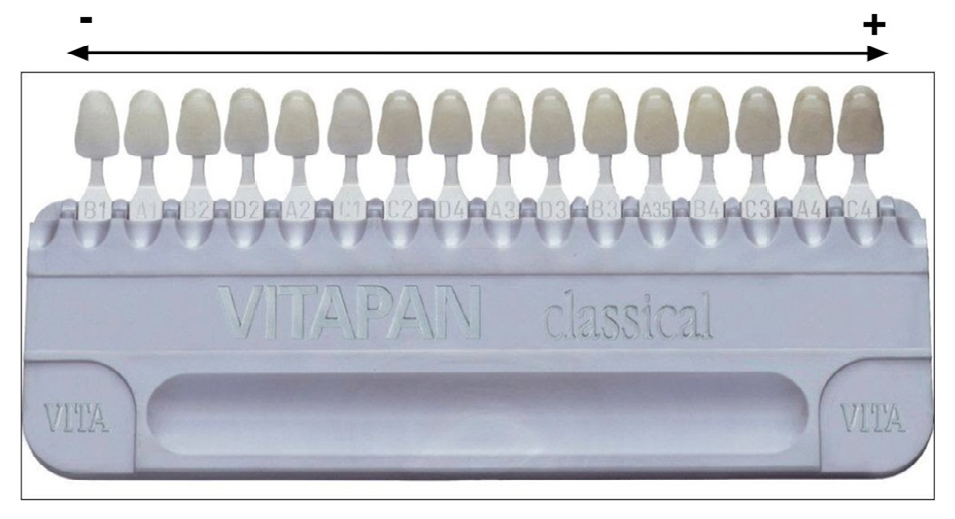
Five cylindrical test specimens 15 mm in diameter and 2 mm thick were made from each of the following six materials (total 30 specimens): RelyX Veneer by 3M ESPE (RX), Paracore White by Coltene (PC), Solocem White Opaque by Coltene (SO), Resin Duo Cement by Densell (DC), and Panavia V5 Paste (PA) and Panavia F 2.0 (PF) by Kuraray Noritake (Table 1). They were all prepared following the manufacturers’ instructions. To make the test specimens, material was placed in a silicone-coated mold, and pressed with a glass slide to minimize inclusion of bubbles that would affect specimen shade. Excess material was removed. The specimens were light-cured using a COLTOLUX Coltene LED light-curing unit (1200 mW/cm 2 ). Then they were fixed to a strip of double-sided adhe si ve tape attached to a sheet of white paper, and numbered from 1 to 5 for each material. Color was measured with a Vita Easyshade spectrophotometer which had been calibrated before use following the manufacturer’s instructions. The spectrophotometer has a color temperature of 6500 K (D65). Color was measured in the center of each test specimen (Fig. 1) against a white background, according to the Vita Classic and Vita 3D Master color scale and considering the Vita Classic sampler (Fig. 2). Specimens were stored in a closed box to prevent contact with light. They were subsequently subjected to an accelerated artificial aging procedure at the National Institute of Industrial Technology (Instituto Nacional de Tecnología Industrial, INTI), in a chamber where they were exposed for 14 days to cycles of 4 hours of UV radiation with a fluorescent lamp at 60 °C and 4 hours of water vapor condensation at 50 °C. After the aging procedure, the color of each specimen was recorded again in the same way. Color change was evaluated on the color scale, ordered according to shade. The differences between the initial and final colors for each group
were analyzed using the Kruskal Wallis test and múltiple comparisons, with significance level p<0.05. A table providing equivalencies between colors on the Vita Classic and CIELAB scales 25 (Table 2) was used to transform the recorded colors to equivalent values on the CIELAB scale. The difference in color (ΔE) was calculated for each group according to the following formula:
where ΔL, Δa and Δb are the differences in the values of L, a and b before and after AAA. Mean and standard deviation were analyzed statistically
RESULTS
Mean and standard deviation values for the difference in Vita Classical Shades for each group were: RX 8.40 (1.52); PC 8.60 (3.13); SO 6.40 (3.51); DC 10.00 (0.00); PA 7.60 (3.29); PF 2.00 (0.00) (Fig. 3). The Kruskal Wallis test showed significant difference for the factor ‘material’ (p<0.05) and comparison of means showed a difference between PF and the rest of the materials.

Fig. 3 Arithmetic mean and standard deviation of the difference on the Vita Classic scale. Differences between groups identified with the same lowercase letters are not statistically significant.
Mean and standard deviation values for the difference in in color ΔE for each group were: RX 14.94 (2.01); PC 14.51 (4.01); SO 12.07 (4.53); DC 16.31 (0.00); PA 10.90 (3.38); PF 7.23 (0.00) (Fig. 4). Analysis with ANOVA showed significant differences among the materials evaluated (p<0.05). Comparison of means with Tukey’s test showed the presence of two groups (PF-DC, RX, PA).
DISCUSSION
Analysis based on CIELAB showed that all test specimens underwent darkening, with reduction of L* values. This may be associated to the presence of oxides or unreacted components in the polymerization system, which can act as intrinsic pigments 17, 18 . There was an increase in a* values, indicating a trend towards reddish discoloration. For the b* coordinate, all specimens presented a yellowing effect during aging.
Composite resin color stability involves both intrinsic factors owed to the material’s physicochemical reactions and extrinsic factors 12 . Regarding composition, most bonding agents used in this study contain TEGDMA and Bis-GMA monomers with structural chemical groups prone to hydrolysis and/ or hydrogen bond with water: ether in TEGDMA, hydroxyl in Bis-GMA, and ester in both. Hydrolytic degradation and hygroscopic effects are determinant factors in color variation in resin-based materials. Color changes may be related to the nature of the organic matrix 13 . TEGDMA-based resins release larger quantities of monomers in aqueous environments than do Bis-GMA- and UDMA-based materials. Regarding water absorption, Bis-GMA-based resins are more absorbent than TEGDMA. UDMA has lower absorbance and greater color stability 19 . Resins with larger filler particles are more susceptible to color change, possibly because color perception is directly related to dispersion. Since the resin-filler interface is one of the weakest points of the material, with high sensitivity to water absorption, it may be assumed that hydrolytic degradation of the interface could modify the way in which light is dispersed by the particles. Differently sized particles may determine differences in the degree of dispersion due to aging in water 13 . In the current study, both light-cured and dual resin-based cements underwent significant color change. Many studies have reported that the mode of polymerization activation (dual polymerization and photopolymerization) had no influence on color change 8-10 . However, other authors, such as Almeida, noted that dual polymerization resin-based cement underwent significant color change. Color change in composite resins has been associated to hydrolytic degradation of organic components in resin-based materials, especially with chemical degradation of polymerization promoters 7 .
Dual polymerization resin-based bonding agents are believed to be more prone to color change due to oxidation of tertiary aromatic amines present in their composition and of inhibitors, while light-cured resin-based bonding agents have greater color stability due to their aliphatic amines being less susceptible to oxidation 14 . The chemical reaction between amine and benzoyl peroxide is not very efficient because it depends on the physical bonding between molecules during the polymerization reaction 17 . Initiator mobility becomes restricted as the material polymerizes and its viscosity increases, leaving unreacted initiators in the polymer network 9 . Kilinc et al. say that inhibitor breakdown leads to yellowing 15 . Some authors report that the proportion between light-cured and chemical-cured components in the system is determinant. When there is a high concentration of light-sensitive components, greater color stability may be expected, while greater presence of chemical activation components leads to greater color change 4,11, 14 . However, manufacturer information regarding the quantity of each component is unclear. Based on the results of the current study showing significant color change in dual resin-based bonding agents, it may be inferred that they contain a high proportion of self-curing components.
The initiator in the light-cured resin-based material used in the current study, RelyX Venner, is camphorquinone, which is yellowish, and produces slight color changes during polymerization. The effectiveness of photopolymerization depends on the quantity of amine molecules available to bond with the camphorquinone, and is directly related to the material’s mechanical and optical properties. Photopolymerization materials increase the yellowish appearance during the first 24 hours following polymerization because the free camphorquinone molecules return to their original yellow color 7 .
The AAA procedure consists of aggressive conditions of humidity, ultraviolet radiation and high temperature. It is not yet possible to reliably simulate the clinical situation, since factors such as absorption of pigments from food, bacterial plaque accumulation and mechanical loading are excluded 8 . Pissaia et al. used the method involving immersion in distilled water, and concluded that color changed less in light-cured resin-based cements than in dual-cured ciments 1 . The aging procedure can be used to compare different materials to each other, but cannot be correlated to a clinical time frame 15 . Some authors estimate that the color changes within the first 300 hours of AAA at 37 °C are equivalent to one clinical year 10, 26 .
Spectrophotometers are considered to be objective instruments for measuring color, and due to their precision, sensitivity, reproducibility and ease for communication, have been used in many studies to quantify tooth color. Despite these advantages, the translucence of the tooth structure and curved surfaces of teeth can lead to systematic errors in measurement 20 . The current study measured test specimens with flat surfaces, avoiding this type of measurement error.
Based on the individual capacity of the human eye to appreciate differences in colors, three different intervals are used to distinguish changes in color valúes: ΔE<1 is imperceptible to the human eye; 1<ΔE<3.3 is clinically acceptable, and ΔE>3.3 is not clinically acceptable. Restorations may thus be considered clinically acceptable when ΔE is less than 3.3. Turgut et al. studied color change using 0.5 mm ceramic blocks and cement with a thickness of 0.1 mm, finding that although there was a change in color, the change in the block was clinically acceptable according to ΔE valúes. They suggested that the resin-based cement was the main cause of color change in restoration treatments with ceramic veneers because ceramic materials have high color stability 4 . Ceramic veneers are thinner than ceramic crowns, and therefore present greater color change 5 .
In the current study, ΔE was greater than 3.3 for all materials, which may be attributed to the greater thickness of solid cement specimens. The literature shows that discoloration is less in resin-based cements that are polymerized beneath ceramic structures than in solid cement specimens, because color change of the entire block is minimized, and possibly also due to the cement being protected by the surface layer of porcelain, the low volume of cement, and its low degree of direct exposure to the environment 6 , all of which are favorable to reducing color change. Tests on solid resins show higher pigmentation due to the intrinsic properties of the material. The current study used cement specimens 2 mm thick, which is much thicker than the amount used clinically, and it was not covered by a ceramic layer, which may affect color change.
Although an in vitro study cannot be directly transferred to the clinical situation, it provides an interesting level of evidence. It would be ideal to perform this analysis using a different design, to evaluate the effect of the same variables on color change. Resin-based cements are known to undergo color change, and further studies could analyze whether color change with the ceramic is significant.