Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO  uBio
uBio
Compartilhar
Kurtziana
versão On-line ISSN 1852-5962
Kurtziana vol.37 no.1 Córdoba jan./jun. 2012
ARTÍCULOS ORIGINALES
Molecular studies reveal a speciation process within Ryvardenia cretacea (Polyporales, Basidiomycota)
Mario Rajchenberg 1, 2, * & María Belén Pildain 1, 2
1 Protección Forestal, Centro de Investigación y Extensión Forestal Andino Patagónico, C.C. 14, 9200 Esquel, Chubut, Argentina
2 Consejo Nacional de Investigaciones Científicas y Técnicas, Argentina.
* Corresponding author: mrajchenberg@ciefap.org.ar
Summary
rDNA sequentiation and phylogenetic analysis of the ITS region of strains of the brown-rot polypore Ryvardenia cretacea from Australia (Tasmania, Victoria) and Argentina (Patagonia) revealed the existence of two well supported clades. Each clade appears restricted to one side of the Pacific indicating a strong biogeographical isolation between strains and populations in the process of speciation. Nevertheless, detailed studies and comparisons did not show the existence of significant morphological differences. This fact, coupled with previously reported dikaryon x monosporic mating tests which showed biological compatibility between specimens from both regions, supports the maintenance of a complex but taxonomically single entity.
Key words: Phylogeny; Polypores; Speciation.
Resumen
Estudios moleculares revelan la existencia de un proceso de especiación en Ryvardenia cretacea (Polyporales, Basidiomycota)
La secuenciación molecular del segmento ITS del ADNr de cepas del políporo de pudrición castaña Ryvardenia cretacea provenientes de Australia (Tasmania, Victoria) y de Argentina (Patagonia) y su posterior análisis filogenético permitió revelar la existencia de dos clados con buen soporte. Cada una de ellos aparece con una distribución geográfica restringida a cada lado del Pacífico austral, indicando la existencia de un proceso de especiación. No obstante, estudios morfológicos comparativos detallados no revelaron la existencia de diferencias morfológicas entre materiales de herbario de esas procedencias. Ello, junto al hecho que investigaciones previas demostraron la compatibilidad entre cultivos secundarios y primarios entre especímenes de ambas regiones, sostiene el considerar a este taxón como un complejo con una única entidad taxonómica a nivel de especie.
Palabras clave: Filogenia; Políporos; Especiación.
Introduction
Ryvardenia Rajchenb. (Polyporales, Basidiomycota) is an austral genus of brown-rot polypores that was established in order to accommodate Polyporus cretaceus Lloyd (type species) and Polyporus campylus Berk. (Rajchenberg, 1994). The genus is characterized by basidiomes with an obscurely dimitic hyphal system in the dissepiments, where long, terminating, thick-walled hyphae resemble skeletal hyphae, while in the context the hyphal system appears to be monomitic; the tissues are characterized by the presence of thickwalled, clamped generative hyphae with glossy/refringent walls that are not metachromatic in cresyl-blue and that contrast with thinwalled generative hyphae; basidiospores are obovate, broad-ellipsoid to drop-shaped and thick-walled. From a biological point of view Ryvardenia is characterized by: brownrot taxa with a heterothallic, bipolar mating system; astatocoenocytic nuclear behavior of the mycelium (Boidin, 1971); and in culture generative hyphae of the advancing mycelium being simple-septate while towards the colony centre generative hyphae are homogeneously sclerified and clamped and fiber hyphae are never irregularly thick-walled (Rajchenberg, 1994).
Species in Ryvardenia were previously treated in different monomitic polypore genera such as Tyromyces P. Karst., Postia Fr., Spongipellis Pat., and Grifola S.F. Gray, or in the dimitic Piptoporus P. Karst. The genus was accepted by Buchanan & Ryvarden (2000) in their review of New Zealand polypores. Its validity from a phylogenetic perspective remains to be tested.
Ryvardenia cretacea (Lloyd) Rajchenb. is distinguished by its large, generally solitary basidiomes, with a consistency that turns chalky upon drying and, microscopically, by its generative hyphae of variable width that become tortuous, with swellings and with hyphal segments appearing as if disarticulated. It decays standing or fallen Nothofagus in southern Argentina and southern Chile and also Eucalyptus in SE Australia. It has been recorded as a wood-rotting species of minor importance for standing Nothofagus pumilio forests in southern Argentina (Cwielong & Rajchenberg, 1995). During a study of Postia s.l. taxa known from southern Argentina (Rajchenberg, 2006) we were able to compare several strains of R. cretacea from different geographic origins using molecular methods. The genus Ryvardenia has not until now been included in molecular phylogenetic analyses of brown-rot polypore genera and its phylogenetic relationships are, thus, unknown. The aim of the present work is to determine the variability of R. cretacea isolates based on the internal transcribed spacer region (ITS) of the nuclear ribosomal DNA, on micro and macro-morphology and on sexuality; and to assess their phylogenetic relationships.
Material and Methods
Strains and dried specimens studied
Strains are deposited at and retrieved from the author's institutional culture collection (CIEFAPcc), from the International Collection of Micro-organisms from Plants (ICMP) at Landcare Research (Auckland, New Zealand) and from the Center for Forest Mycology Research Culture Collection and Herbarium (CFMR) at the Forest Products Laboratory (USDA Forest Service, Madison, WI, USA).
Herbarium acronyms of specimens studied follow Index Herbariorum at the web page: http://sciweb.nygb.org/science2/IndexHerbariorum.asp; CIEFAP corresponds to the author's institutional herbarium.
Ryvardenia cretacea
CIEFAPcc 38: ARGENTINA, Prov. Chubut, Dto. Futaleufú, Lago Baggilt, Nothofagus pumilio forest, N. Rodríguez & D. Majul (MR 10421), 2-IV-1991, isolated from basidiome (CIEFAP). GenBank JQ677136.
CIEFAPcc 113: ARGENTINA, Prov. Neuquén, Parque Nacional Lanín, Lago Lácar, Hua-Hum, mixed forest of Nothofagus obliqua and N. nervosa, M. Rajchenberg 10667, 10-IV-1992, isolated from basidiome fruiting on N. nervosa (CIEFAP). GenBank JQ677137.
CIEFAPcc 182: ARGENTINA, Prov. Neuquén, Parque Nacional Nahuel Huapi, Lago Espejo, Nothofagus dombeyi forests at the beginning of the road towards San Martín de los Andes, M. Rajchenberg 12329, 17-V-2010, isolated from basidiome (CIEFAP). GenBank JQ677142.
CIEFAPcc 97 (=ICMP 11789): AUSTRALIA, Tasmania, Great Western Tiers, Liffey Falls Res., lower track, on Nothofagus cunninghamii, D. Arora (P.K. Buchanan 87/298), 14-V-1987, isolated from basidiome PDD 59515. GenBank JQ677138.
CIEFAPcc 98 (=ICMP 11791): AUSTRALIA, Tasmania, Franklin Gordon Wild Rivers National Park, Lyell Highway, Franklin River Bridge, on Nothofagus cunninghamii, P.K. Buchanan 87/342, 17-V-1987, isolated from basidiome PDD 55256. GenBank JQ677139.
CIEFAPcc 151 (=CFMR 6449): AUSTRALIA, Victoria, on Eucalyptus regnans, 1950. Basidiome not studied. GenBank JQ677141.
CIEFAP: ARGENTINA, Prov. Neuquén, Parque Nacional Nahuel Huapi, Lago Espejo, ruta 254 ca. 5 km from the turn towards San Martín de los Andes, on standing Nothofagus dombeyi, M. Rajchenberg 12429, 9-V-2011. GenBank JQ677141.
PDD 12295: AUSTRALIA, Victoria, West Tanjil R., on Nothofagus cunninghamii, J.H. Willis, X-1941.
PDD 60300: AUSTRALIA, Tasmania, Arthur River, Sumac Road near Sumac Lookout, in Nothofagus cunninghamii forest, Y. Doi, 15-V-1987.
PDD 97266: AUSTRALIA, Victoria, vic. Marysville, Lady Talbot Drive, The Beeches walk, P. Buchanan PKB 96/388, 6-X-1996.
Ryvardenia campyla
CIEFAPcc 124: ARGENTINA, Prov. Chubut, Parque Nacional Los Alerces, Lago Futalaufquen, pathway to Lago Krugger, on standing Nothofagus dombeyi, M. Rajchenberg 10575, 27-III-1992. GenBank JQ677140.
Phylogenetic analyses
DNA from tissue cultures cited above, grown on malt extract liquid medium, was extracted using the UltraCleanTM Microbial DNA Isolation Kit (MO BIO laboratories inc., Solana Beach, USA) according to the manufacturer's instructions. The ITS region of the extracted DNA was amplified using the universal primers ITS5 and ITS4 (White et al., 1990).
PCR reaction mixtures for amplification of the ITS region were as described by Rajchenberg et al. (2011): dNTPs (0.25 mM of each), 2.5 mM MgCl2; PCR buffer supplied with the polymerase enzyme; 0.1 µM of each primer; 100-500 ng DNA; and 1.25 U of GoTaq polymerase (Promega, Madison, WI, USA). The final reaction volume was 50 µL. The PCR reactions were performed in a thermal cycler (My CyclerTM, BioRad) and the conditions were as described by Rajchenberg et al. (2011). PCR products were separated on a 1% (w/v) ethidium bromide-stained agarose gel and the bands were visualised under UV illumination. The amplified fragments were purified and sequenced at the DNA Synthesis and Sequencing Facility, Macrogen (Seoul, Korea). GenBank accession numbers of the sequences obtained are shown in Table 1.
Table 1
Fungal strains from Argentina and Australia used in this study, and GenBank sequences included in the phylogenetic analysis.

DNA sequences were aligned on MEGA version 4.0 (Tamura et al., 2007; available from the authors upon request). Phylogenetic trees based on neighbourjoining (NJ) and maximum parsimony (MP) were generated. A NJ tree was generated using MEGA version 4.0 (Tamura et al., 2007), with gaps and missing data deleted prior to the analysis. A maximum composite likelihood model was used to calculate the distances between taxa. Bootstrap analysis included 1,000 replicates using the same settings. MP trees were generated using PAUP* 4.0b10 (Swofford, 2000). For MP phylogenetic analyses, gaps were treated as missing characters and trees were generated by heuristic searches with random addition of sequences (1000 replicates), TBR (tree bisection reconnection) branch swapping and MULTREES effective. Bootstrapping (1000 bootstrap replicates) was used to determine confidence in the branches.
Antrodiella semisupina (Berk. & M.A. Curtis) Ryvarden was used as outgroup (GenBank AF126904). Sequences of Postia tephroleuca (Fr.) Jülich and Antrodia serialis (Fr.) Donk were retrieved from GenBank as indicated in Table 1.
Morphological studies
Herbarium specimens were studied with classical methods in order to corroborate morphological features described previously (Rajchenberg, 1994, 2006). This included the study of 30 spores of each collection, which were measured in sections mounted in Melzer's reagent; their mean length and width were determined in order to compare and/or distinguish differences in their sizes. One-way ANOVA analyses were performed using SPSS v.17
Results
The ITS-sequence dataset included 718 total characters, of which 98 were parsimony informative. Heuristic tree searches under MP analysis resulted in two equally most parsimonious trees. The tree length (TL) was 333, consistency index (CI) and retention index (RI) were 0.9 and 0.78, respectively. One of these trees is presented in Fig. 1. Bootstrap values obtained using MP and NJ were very similar and both analyses resulted in a similar tree topology, with high statistical support for the Ryvardenia cretacea species complex. Geographical sub-groups within R. cretacea were well supported; bootstrap values for Argentinean specimens (ARG): MP= 99, NJ = 96; and for Australian specimens (AUST): MP= 100, NJ = 99.
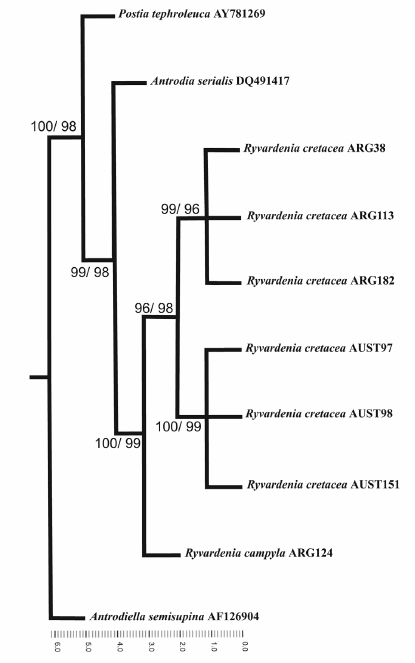
Fig. 1. One of two most parsimonious trees inferred from MP analysis of the ITS region dataset for cultures from 10 specimens. Numbers at the internal nodes indicate MP and NJ bootstrap values, respectively.
Morphological studies corroborated previous descriptions based on basidiomes from both sides of the Pacific (Rajchenberg, 1994). No significant differences between Argentinean and Australian specimens were observed in terms of pores size, basidiome size and consistency, hyphal construction of the basidiome, hyphal features including branching, variable width and wall thickness, basidia, and spores shape and size; all features were as recorded previously [Rajchenberg (1994, 2006); cfr. also Reid (1957) and Cunningham (1965)]. In particular, basidiospores showed homogeneity in being obovate, broad-ellipsoid to drop-shaped, with thickened walls and similar in size. A statistical analysis of spore sizes did not show grouping of specimens according to geographic distribution (Table 2).
Table 2
Spores measurements of Ryvardenia cretacea specimens studied. *Same letter indicates no significant difference between specimens.

Ecological differences, i.e. differences in substrates between specimens from Argentina and Tasmania were not detected through the phylogenetic analyses.
Discussion
Results of this study show that populations of R. cretacea from Australia and southern Argentina could be considered as distinct at the species level on phylogenetic grounds. The bootstrap values that support both sub-groups are high enough to sustain this conclusion and accord to a phylogenetic species concept. Nevertheless, the typological and biological species concepts do not align with our phylogenetic results. Detailed morphological study failed to identify morphological differences between specimens from the two widely separated geographic regions; on the contrary our results corroborated previous descriptions that included specimens from Australia, southern Argentina and southern Chile. In addition, mating tests by Rajchenberg (1994), when he introduced the genus Ryvardenia, suggested one biological species. In that study three tissue/polysporic cultures from Tasmania and Victoria (Australia), that were also used in the present phylogenetic study, were confronted against 6 monosporic cultures from basidiomes MR 10621 (Argentina, Prov. Chubut, Sierra Colorada, M. Rajchenberg 10621¸1-IV- 1992, on standing Nothofagus pumilio, at CIEFAP herbarium) and MR 10674 (Argentina, Prov. Neuquén, Parque Nacional Lanín, Lago Hermoso, margin N, M. Rajchenberg 10674, 11-IV-1992, on fallen branch of Nothofagus dombeyi, at CIEFAP herbarium). These confrontations gave positive results after 7 weeks and, therefore, gave support to the idea that the specimens are conspecific from a biological point of view.
Several examples of non-congruence between the biological and phylogenetic species concepts are present in the mycological literature and it is the taxonomist's decision to recommend one or the other. Among only a few examples from the Southern Hemisphere, Pildain et al. (2009, 2010) showed for Armillaria that A. novae-zelandiae (G. Stev.) Herink forms a clade with specimens from southern Argentina and Chile, New Zealand, Australia and Malaysia, but there was a strong substructure formation with four subclades, each corresponding to geographically different areas. Strains from Patagonia grouped together and apart from those of other geographic regions in a well supported subclade, suggesting the possibility that they represent a distinct taxon. However, based on morphology they were similar to specimens from New Zealand and additional (i.e., biological) evidence is considered necessary before describing new species in that group. Paulus et al. (2000) also found a geographically based, phylogenetic substructure for populations of the polypore Schizopora radula (Pers.: Fr.) Hallenb. from southern Argentina, New Zealand and Australia. In this case biological intercompatibility was also demonstrated, precluding further taxonomic differentiation at the species level.
Geographically delimited populations of a fungal species may often be distinguished phylogenetically (Hallenberg et al. 2007). This may be due to their long history of isolation (Jin et al. 2001) as might be the case for many austral fungal species. A vicariant event such as the separation of Gondwana and the following tectonic drift of land masses may have dominated their history. One should be warned, though, that trans-oceanic dispersal could also have taken place during the millions of years of progressive separation, as was documented by Sanmartín & Ronquist (2004) and Muñóz et al. (2004) for the austral biomes. Maintenance of intercompatibility (i.e., breeding processes) could be explained by such more recent events, but also by the lack of need to develop reproductive isolation due to the geographic isolation. Because species are biological entities of relevance in ecological, pathological and biodiversity contexts, it is better to understand them as species complexes when no morphological, ecological and/or compatibility differences exist. For this reason it is our conclusion that R. cretacea is a species that is undergoing a speciation process as evidenced by the existence of two phylogenetic lineages. This work is a first step to assess the relationships within this taxon. More research including monosporic x monosporic compatibility tests and multigenic phylogenetic studies are necessary to further understand the taxonomic circumscription of this complex.
Acknowledgments
This paper is dedicated to Prof. Leif Ryvarden in honor of his numerous contributions to Polypore taxonomy, his untiring collecting activity throughout the world, and in recognition to his open and kind spirit. Thanks for that Leif! This study was funded by PIP-CONICET 80101000. The authors are members of the "Carrera del Investigador Centífico", National Research Council of Argentina (CONICET). Dr. P.K. Buchanan (Landcare Research, Auckland, New Zealand) kindly arranged the shipment of specimens kept at PDD. He and an anonymous reviewer made most valuable suggestions to improve the manuscript.
References
1. Boidin, J. 1971. Nuclear behavior in the mycelium and the evolution of the Basidiomycetes. In Petersen R.H. (Ed.) Evolution in the higher Basidiomycetes, p.129-148, Knoxville, USA: University of Tennessee Press, 562 pp. [ Links ]
2. Buchanan, P. K. & L. Ryvarden. 2000. An annotated checklist of polypore and polypore-like fungi recorded from New Zealand. New Zeal. J. Bot. 38: 265-323. [ Links ]
3. Cunningham, G. H. 1965. Polyporaceae of New Zealand. New Zealand DSIR Bulletin 164: 1-304. [ Links ]
4. Cwielong, P. P. & M. Rajchenberg. 1995. Wood rotting Fungi on Nothofagus pumilio in Patagonia, Argentina. Eur. J. For. Pathol. 25: 47-60. [ Links ]
5. Hallenberg, N., R. H. Nilsson, A. Antonelli, S.-H. Wu, N. Maekawa & B. Nordén. 2007. The Peniophorella praetermissa species complex (Basidiomycota). Mycol. Res. 111: 1366-1376. [ Links ]
6. Jin, J., K.W. Hughes & R. H. Petersen. 2001. Biogeographical patterns in Panellus stipticus. Mycologia 93: 308-315. [ Links ]
7. Muñoz, J., Á. M. Felicísimo, F. Cabezas, A. R. Burgaz & I. Martínez. 2004. Wind as long-distance dispersal vehicle in the Southern Hemisphere. Science 304: 1144-1147. [ Links ]
8. Paulus, B., N. Hallenberg, P. K. Buchanan & G. K. Chambers. 2000. A phylogenetic study of the genus Schizopora (Basidiomycota) based on ITS DNA sequences. Mycol. Res. 104: 1155-1163. [ Links ]
9. Pildain, M. B., M. P. A. Coetzee, B. D. Wingfield, M. J. Wingfield & M. Rajchenberg. 2010. Taxonomy of Armillaria in the Patagonian Forests of Argentina. Mycologia 102: 392-403. [ Links ]
10. Pildain, M. B., M. P. A. Coetzee, M. Rajchenberg, R. H. Petersen, M. J. Wingfield & B. D. Wingfield. 2009. Molecular phylogeny of Armillaria from the Patagonian Andes. Mycol. Progress 8: 181- 196. [ Links ]
11. Rajchenberg, M., S. P. Gorjón & M. B. Pildain. 2011. The phylogenetic disposition of Antrodia s.l. from Patagonia, Argentina. Aust. Syst. Bot. 24: 111-120. [ Links ]
12. Rajchenberg, M. 1994. A taxonomic study of the subantarctic Piptoporus (Polyporaceae, Basidiomycetes) I. Nord. Jour. Bot. 14: 435-449. [ Links ]
13. Rajchenberg , M. 2006 . Los Políporos (Basidiomycetes) de los Bosques Andino Patagónicos de Argentina. Polypores (Basidiomycetes) from the Patagonian Andes Forests of Argentina. Versión bilingüe. Bibliot. Mycol. Band 201, J. Cramer Verlag, Stuttgart. 300 pp. [ Links ]
14. Reid, D. A. 1957. New or interesting records of Australasian basidiomycetes III. Kew Bull. 1957: 127-143. [ Links ]
15. Sanmartín I. & F. Ronquist. 2004. Southern Hemisphere biogeography analyzed with event-based models: Plant versus animal patterns. Systematic Biology 53: 216-243. [ Links ]
16. Swofford, D. L. 2002. PAUP*: phylogenetic analysis using parsimony (*and other methods). Version 4.0b10. Sunderland, Massachusetts: Sinauer Associates. [ Links ]
17. Tamura K., J. Dudley, M. Nei & S. Kumar. 2007. MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0. Molec. Biol. Evol. 24: 1596-1599. doi: 10.1093/molbev/msm092 [ Links ]
18. White, T. J., T. Bruns, S. Lee & J. W. Taylor. 1990. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: M.A. Innis, D.H. Gelfand, J.J. Sninsky, T.J. White (Eds.), PCR protocols: a guide to methods and applications. New York: Academic Press, pp. 315-322. [ Links ]
Original recibido el 18 de Agosto de 2011;
primera decisión: 6 de Febrero de 2012;
aceptado el 17 de Febrero de 2012.
Editor responsable: Gerardo Robledo.














