Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Kurtziana
On-line version ISSN 1852-5962
Kurtziana vol.37 no.1 Córdoba Jan./June 2012
ARTÍCULOS ORIGINALES
A new species of Pseudoplectania (Sarcosomataceae, Pezizales) from Venezuela
Teresa Iturriaga 1, Melissa Mardones 2 & Héctor Urbina 2
1 Departamento Biología de Organismos, Universidad Simón Bolívar, Caracas, Venezuela, titurri@usb.ve
2 Postgrado en Ciencias Biológicas, Departamento Biología de Organismos, Universidad Simón Bolívar, Caracas, Venezuela.
Summary
A new species of the genus Pseudoplectania (Sarcosomataceae, Pezizales) was collected on wood in a tropical forest in southern Venezuela, at Gran Sabana, Canaima National Park, Bolívar State; species of the genus Pseudoplectania are characterized by having a deep cup shaped sessile or stipitated apothecia, covered by coiled or twisted hairs, J- asci containing 8 globose, smooth, hyaline ascospores; the new species can be distinguished from the rest of the species in the genus in having small short-stipitate apothecia, cylindrical hyaline paraphyses and ascospores with small scattered warts visible only in material stained with aqueous Congo Red; ornamented ascospores have not been previously described in the genus.
Key words: Discomycetes; Ascospore ornamentation; Congo Red; Pseudoplectania; Neotropics; Fungi.
Resumen
Una nueva especie de Pseudoplectania (Sarcosomataceae, Pezizales) de Venezuela
Una nueva especie perteneciente al género Pseudoplectania (Sarcosomataceae, Pezizales) fue coleccionada creciendo sobre madera en un bosque tropical del Sur de Venezuela, en la Gran Sabana, Parque Nacional Canaima, Estado Bolívar; las especies de Pseudoplectania se caracterizan por poseer un apotecio discoide, profundo, que puede ser sésil o estipitado y que se encuentra cubierto por tricomas espiralados o entorchados, ascos J- que contienen 8 ascosporas globosas, hialinas y de pared lisa; la nueva especie se distingue del resto de las especies del género por la presencia de un apotecio pequeño y estipitado, paráfisis hialinas y cilíndricas y ascosporas ornamentadas con pústulas no cianofílicas visibles sólo en montajes en reactivo Rojo Congo; las ascosporas ornamentadas no han sido descriptas para otras especies del género.
Palabras clave: Discomycetes; Esporas ornamentadas; Fungi; Neotrópico; Pseudoplectania; Rojo Congo.
Introduction
Pseudoplectania and Plectania are morphologically similar genera that were proposed by Fuckel (1870); these genera are similar in having blackish discoid apothecia covered on the outside by a dark tomentum. Differences are that Pseudoplectania species have globose spores and Plectania species have spores that are ovoid to ellipsoid. Some authors have suggested that the two genera are synonyms (Korf 1970, 1972, Paden 1983, Zhuang & Wang 1998, Calonge & Mata 2002, Calonge et al. 2003). The primary morphological difference between the genera is a single character, ascospore shape. Korf (1973) observed that Plectania species have globose spore primordia. In addition, both genera have Conoplea anamorphs. Paden (1983), following Korf (1957), separated Plectania into four sections: Sphaerosporae, Plectania, Curvatisporae, and Plicosporae, and the members with globose ascospores were placed into the section Sphaerosporae.
Other authors accept both genera based on morphological and molecular data (Donadini 1987, Landvik et al. 1997, Harrington et al. 1999, Hansen & Knudsen 2000, Perry et al. 2007). According to Li and Kimbrough (1995) the genera differ in spore wall ontogeny; ascospores of Pseudoplectania lack a secondary wall layer but Plectania has a secondary spore wall layer. This layer is cyanophilic when mounted in cotton blue and observed under a light microscope. Donadini (1987) considered the genera different on criteria such as differences in the number of nuclei in the ascospores, the non-cyanophilic spores in Pseudoplectania and low rate of spore germination in Pseudoplectania. Harrington et al. (1999) using 18S rRNA gene sequences found that Plectania is more closely related with the genus Galiella while Pseudoplectania is related to Donadinia. However, Perry et al. (2007) using partial sequences of nuLSU rDNA concluded that Plectania and Donadinia are more closely related. Even when there is not convincing evidence to support one or the other of these positions, both studies separate Plectania from Pseudoplectania, hence, we consider Plectania and Pseudoplectania as different taxa.
A specimen referred to the genus Pseudoplectania was collected on wood in a gallery forest in the locality of Luepa, Gran Sabana, Canaima National Park, Bolívar State, southern Venezuela. Characters of the specimen include its disc-shaped apothecia, tomentose black receptacle, dark brown to black hymenium and globose hyaline ascospores. It differs from all other species included in the genus Pseudoplectania by a combination of characters: small ascomatal size compared with the rest of species, its short stipe, cylindrical hyaline paraphyses and ornamented ascospores.
Materials and methods
Protocols for morphological examination and sectioning followed those outlined in Iturriaga and Korf (1990). Congo red in water was used as a stain for observing wall details (ornamentation) in asci and ascospores. Cotton blue in lactic acid was used to observe structures and tissues in sections and squashed material.
Results
Pseudoplectania ryvardenii Iturr., Mardones & H. Urbina, sp. nov.
VENEZUELA. Bolívar State, Gran Sabana. Canaima National Park, Gallery forest in Luepa. N 0671180, W 0646970, 1410 msnm, T. Iturriaga, & H. Urbina. 657, 13.VII.2003. On dead wood. Holotypus specie (VEN402017). Figs. 9-12.
MycoBank (MB 563323)
Apothecium dark brown when fresh or rehydrated, black when dry, 5-7 mm diam., 5-8 mm tall, gregarious, deep cup shaped, stipitated, with radiating black anchoring basal mycelium. Disc concave, brown to almost black, smooth. Receptacle dark brown, covered by short-brown hairs on the upper side and long brown hairs at the base. Hairs numerous, dark brown, thick-walled, septate, branched, cylindrical, sinuate, 200-600 µm × 8-10(-12) [x9.5 m15 s1.6 n19] µm at base, 60-130 µm × 6-8(-12) µm at flanks and margin. Ectal excipulum textura angularis to textura globulosa, 60-100 µm wide at base, 50-70 µm wide at the flanks and 30-50 µm wide at the margin; with brown thick-walled cells, 9-20(-24) [x15 m16 s5.2 n14] × (7-)8-13(- 16) [x10 m8 s3 n14] µm, inner cells hyaline, outer cells dark-brown, the later ones giving rise to tomentum hyphae. Medullary excipulum hyaline to yellow, composed of textura intricata in a gelatinous matrix, 750 µm wide at the centre; 200 µm wide at the flanks and 120 µm wide at the margin; hyphae hyaline to yellow, 4-10 [x6,5 m5 s2,2 n22] µm diam., thin-walled. Hymenium yellow, 150-400 µm tall. Asci hyaline, long-cylindrical, narrowing below into a thin flexuous base, apex J-, suboperculate, 8-spored, 250-314(-344) [x284 m270 s14.5 n9] × 11-14.5 [x12.5 m11 s1.7 n16] µm. Ascospores globose, hyaline, with one large central guttule, 10.5-13 [x11 m10.5 s1 n16] µm diam., thick-walled, with randomly scattered pustules seen only under Congo Red. Paraphyses hyaline, cylindrical, septate, straight, thin-walled, 2.4-3.2 µm diam. Hymenial hairs absent.
Ecology & Distribution: Gregarious, on decaying wood in a gallery forest dominated by Dimorphandra macrostachya Benth. Presently only known from the type locality.
Discussion
To date, Pseudoplectania has not been reported from Venezuela (Iturriaga & Minter 2006). The genus Pseudoplectania currently comprises seven species: Ps. ericae Donadini, Ps. kumaonensis Sanwal, Ps. melaena (Fr.) Sacc., Ps. nigrella (Pers.) Fuckel, Ps. sphagnophila, Ps. stygia (Berk. & M.A. Curtis) Sacc. and Ps. vogesiaca Seaver. Among the genus Plectania, only one species has spherical spores: Pl. carranzae Calonge & M. Mata. Only Pl. carranzae and Ps. nigrella have been reported for the Neotropics.
Pl. carranzae, Ps. sphagnophila, Ps. stygia and Ps. vogesiaca have stipitate apothecia, the rest of species have substipitate or sessile apothecia (Figs. 1 & 2). Furthermore, all the species have very large apothecia as compared to Ps. ryvardenii (Table 1).
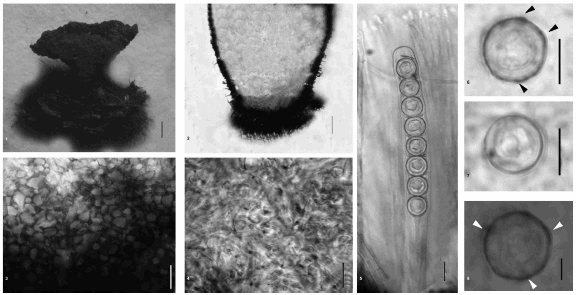
Figs. 1-6. Pseudoplectania ryvardenii. 1. Dry apothecium; 2. Longitudinal section through an apothecium base; 3. Detail of cells of the ectal excipulum ; 4. Detail of hyphae of the medulary excipulum; 5. Ascus and paraphyses; 6. Ascospores. (Scale bars: 1 = 1 mm; 2-8 = 10 µm).

Figures. 9-12. 9. Longitudinal section of the ectal excipulum showing basal tomentum hyphae, ectal excipulum and gelatinized medulary excipulum; 10. Ascospores with ornamentations and gelatinous sheath; 11. Apical and basal parts of an ascus; 12. Paraphysis apex. (Scale bar = 10 µm).
Table 1
Comparison of morphological characters among Pseudoplectania species
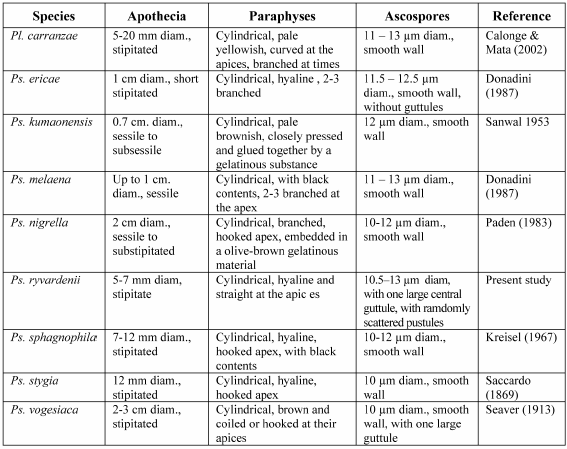
Paraphyses in Ps. ryvardenii are quite different from those of any of the other similar species: showing cylindrical, hyaline and straight paraphyses, while other species as Pl. carranzae, Ps. nigrella, Ps. sphagnophila, Ps. stygia, and Ps. vogesiaca have curved paraphyses with hooked apices. Paraphyses contents have been described as brown or black in Ps. melaena, Ps. nigrella and Ps. sphagnophila; 2 or 3 times branched at the apices in Ps. ericae, Ps. melaena and Ps. nigrella, or with agglutinated paraphyses in Ps. kumaonensis (Table 1).
P. ryvardenii is easily distinguished from all known species of the genus by the small size of its apothecia, up to 7 mm diam; cylindrical, hyaline paraphyses; and ornamented ascospores. This combination of characters does not exist in any of the already known descriptions of species belonging to the genus (Table 1).
Ornamented spores have not been reported in any other species of Pseudoplectania. However, Li and Kimbrough's (1995) ultrastructural studies show that walls of ascospores in Pseudoplectania nigrella are undulate and electron-dense dots are attached to the perisporic sac, outside their primary wall. These are not visible under light microscopy. It is possible that Ps. ryvardenii has large deposits of material than other species in the genus and consequently these can be observed under light microscopy.
Congo red is a very effective stain for showing ascospore wall ornamentations by it binding to the chitin of the cell-wall (Matsuoka et al. 1995). Thus, ornamentations seen under Congo Red and reported by us in this paper suggest that these are chitinous and might be part of the spore wall.
Acknowledgments
The authors thank Richard P. Korf, Plant Pathology Herbarium, Cornell University, for valuable taxonomic comments. We are grateful to FONACIT, (Fondo Nacional de Ciencia, Tecnología e Innovacion) Caracas-Venezuela for funding project Nº S1-2001000663:"Sistemática Molecular y Morfológica de Hongos Ascomycetes y Basidiomycetes presentes en las Sucesiones Fúngicas Descomponedoras de Madera en Bosques Tropicales" under the direction of TI which permitted research in a forest in Luepa, Parque Nacional Canaima as well as giving a benefit to several of the projects being undertaken in the Mycology Lab at USB during that same period.
References
1. Calonge F.D., A. García, M. Sanz & J. Bastardo. 2003. Plectania zugazeae (Ascomycotina), especie nueva para la ciencia. Bol. Soc. Micol. Madrid 27: 17-20. [ Links ]
2. Calonge F.D. & M. Mata. 2002. Plectania carranzae sp. nov. (Ascomycotina), from Costa Rica. Mycotaxon 81: 237-241. [ Links ]
3. Donadini J.C. 1987. Étude des Sarcoscyphaceae ss. Le Gal (1). Sarcosomataceae et Sarcoscyphaceae ss. Korf. Le genre Pseudoplectania emend. nov. P. ericae sp.nov. (Pezizales). Mycol.helv. 2: 217-246. [ Links ]
4. Fuckel L. 1870. Symbolae mycologicae. Beiträge zur Kenntnis der rheinischen Pilze. Jahrbücher des Nassauischen Vereins für Naturkunde 23-24: 1-459. [ Links ]
5. Harrington F.A., D.H. Pfister, D. Potter & M.J. Donoghue. 1999. Phylogenetic Studies within the Pezizales. I. 18S rRNA Sequence Data and Classification. Mycologia 91: 41-50. [ Links ]
6. Hansen L. & Knudsen H. 2000. Nordic Macromycetes. Vol. 1, Ascomycetes. Nordsvamp, Copenhagen. [ Links ]
7. Iturriaga T. & D. Minter. 2006. Hongos de Venezuela. www.cybertruffle.org.uk/venefung [sitio internet, versión 1.00]. [ Links ]
8. Iturriaga T. & R.P. Korf. 1990. A monograph of the discomycete genus Strossmayeria (Leotiaceae), with comments on it's anamorph Pseudospiropes (Dematiaceae). Mycotaxon 36: 383-454. [ Links ]
9. Korf R.P. 1957. Two bulgarioid genera: Galiella and Plectania. Mycologia 49: 107-111. [ Links ]
10. Korf R.P. 1970. Nomenclatural notes. VII. Family and tribe names in the Sarcoscyphineae (Discomycetes) and a new taxonomic disposition of the genera. Taxon 64: 937-994. [ Links ]
11. Korf R.P. 1972. Synoptic key to the genera of the Pezizales. Mycologia 64: 937-964. [ Links ]
12. Korf R.P. 1973. Discomycetes and Tuberales. In: Ainsworth G.C, Sparrow F.K, Sussman A.S. eds. The Fungi, An Advanced Treatise 4A. Academic Press. New York: 249-319. [ Links ]
13. Landvik, S., K. N. Egger & T. Schumacher. 1997. Towards a subordinal classification of the Pezizales (Ascomycota): phylogenetic analyses of SSU r DNA sequences. Nordic J. Bot 17: 403-418. [ Links ]
14. Li, L.T. & J. W. Kimbrough. 1995. Spore wall ontogeny in Pseudoplectania nigrella and Plectania nannfeldtii (Ascomycotina, Pezizales). Can J. Botany 73: 1761- 1767. [ Links ]
15. Matsuoka, H., H.-C. Yang, T. Homma, Y. Nemoto, S. Yamada, O. Sumita, K. Takatori & H. Kurata. 1995. Use of Congo red as a microscopic fluorescence indicator of hyphal growth. Appl. Microbiol Biotechnol 43: 102-108. [ Links ]
16. Paden, J. W. 1983. Sarcosomataceae (Pezizales, Sarcoscyphineae). Flora Neotropica Monograph 37: 1-17. [ Links ]
17. Perry B.A., Hansen, K., Pfister, D.H. 2007. A phylogenetic overview of the family Pyronemataceae (Ascomycota, Pezizales). Mycol. Res. 111: 549-571. [ Links ]
18. Zhuang W.Y. & Z. Wang. 1998. Sarcosomataceous Discomycetes in China. Mycotaxon 67: 355-364. [ Links ]
Original recibido el 15 de Noviembre de 2011;
primera decisión: 18 de Noviembre de 2011;
aceptado el 8 de Diciembre de 2011.
Editor responsable: Andrea Romero.














