Servicios Personalizados
Revista
Articulo
Indicadores
-
 Citado por SciELO
Citado por SciELO
Links relacionados
-
 Similares en
SciELO
Similares en
SciELO
Compartir
Revista de la Facultad de Ciencias Agrarias. Universidad Nacional de Cuyo
versión impresa ISSN 1853-8665versión On-line ISSN 1853-8665
Rev. Fac. Cienc. Agrar., Univ. Nac. Cuyo vol.51 no.1 Mendoza jun. 2019
ORIGINAL ARTICLE
Genetic diversity and pathogenicity on root seedlings from three soybean cultivars of Fusarium graminearum isolated from maize crop residues
Diversidad genética y patogenicidad a nivel de raíz en plántulas de soja de tres cultivares de cepas de Fusarium graminearum aisladas de rastrojos de maíz
Martín Bonacci 1,2; German Barros 1,2
1 Universidad Nacional de Río Cuarto. Facultad de Ciencias Exactas Físico Químicas y Naturales. Departamento de Microbiología e Inmunología. Ruta Nacional 36 Km 601, Río Cuarto, Córdoba, Argentina.
2 Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET). gbarros@exa.unrc.edu.ar
Originales: Recepción: 12/10/2016 - Aceptación: 28/06/2017
ABSTRACT
Crops residues are an important source of maintenance of Fusarium graminearum inoculum in the soybean agroecosystem. Given that these populations can interact in the substrate through mechanisms of mycelial recognition and that they can come into direct contact with the implanted seed and cause disease, the following objectives were set: (1) to evaluate the genetic diversity through of the mycelial compatibility of F. graminearum strains isolated from maize crop residues; (2) to analyze the pathogenicity of F. graminearum strains isolated from crop residues towards soybean seedlings from different cultivars treated and untreated with fungicide. Mycelial compatibility studies showed a unique pattern of mycelial compatibility for each strain, indicating a great heterogeneity in the population evaluated. Pathogenicity tests in all strains tested were capable of causing symptoms of root rot with varying degrees of severity and reductions in the height of seedlings. In the factorial statistical analysis, the greatest effect was marked by the soybean cultivar effect. A clear decline in the severity index was also observed with the fungicide application, so this would be a useful prevention tool to reduce the intensity in soybean seedling diseases.
Keywords: Fusarium graminearum; Crop residues; Soybean; Fungicide; Pathogenicity; Mycelial compatibility
RESUMEN
Los rastrojos de cultivos antecesores son una fuente importante de mantenimiento del inóculo de Fusarium graminearum en el agroecosistema de la soja. Teniendo en cuenta que estas poblaciones pueden interactuar en el sustrato a través de mecanismos de reconocimiento micelial y que las mismas pueden entrar en contacto directo con la semilla implantada y causar enfermedad, se plantearon los siguientes objetivos: (1) Evaluar la diversidad genética a través de la compatibilidad micelial en cepas de F. graminearum aisladas de rastrojos de maíz; (2) Analizar la patogenicidad de cepas de F. graminearum aisladas de rastrojos respecto de la podredumbre de raíz en plántulas de distintos cultivares de soja tratadas y no tratadas con fungicida curasemillas. Los estudios de compatibilidad micelial mostraron un único patrón de compatibilidad micelial para cada cepa, indicando una gran heterogeneidad en la población evaluada. En los ensayos de patogenicidad todas las cepas evaluadas fueron capaces de provocar síntomas de podredumbre de la raíz con distintos grados de severidad y reducciones en la altura de plántulas. El análisis estadístico factorial demostró que el mayor efecto observado en las variables independientes estuvo marcado por el efecto del cultivar de soja evaluado. También se observó una clara disminución en el índice de severidad con la aplicación de un fungicida curasemilla, por lo que esta sería una herramienta útil de prevención para disminuir la intensidad en las enfermedades de plántulas de soja.
Palabras Clave: Fusarium graminearum; Rastrojos; Soja; Fungicida; Patogenicidad; Compatibilidad micelial
INTRODUCTION
Fusarium is a cosmopolitan fungal genus that includes agronomically important plant pathogens. Among them, most relevant pathogens are within four species complex: F. fujikuroi, F. oxysporum, F. solani and F. graminearum (5). The F. graminearum species complex is composed of at least sixteen lineages separate structural and biogeographically, however previous work on soybean in Argentina have shown that F. graminearum sensu stricto is the predominant species (6, 10, 11).
In Argentina, more than 90% of soybean is grown using conservative tillage practices, involving reduced tillage or no-tillage in order to reduce erosion and maintain soil moisture, conserve energy, reduce costs and increase crop yields (21).
As consequence, a greater amount of crop residue on the soil surface after harvest creates a favorable environment for maintaining high inoculum levels of F. graminearum which survives in saprophytic way between hosts. During favorable conditions, F. graminearum produces perithecia with ascospores (sexual spores) which are expelled and dispersed over long distances by air movements (29) and together with conidia (asexual spores) are the primary sources of inoculum (Shaner, 2003). The conidia are produced in large quantities on the crop residues spreading through the rain, and thus arrive at hosts in which produce disease, mycotoxins, or both (28). According to Chiotta et al. (2015a), the crops residues would be the main source of F. graminearum inoculum in soybean agroecosystem compare to the surrounding air crop.
Several studies have analyzed the genetic diversity of F. graminearum populations isolated from grains both wheat and soybean (3, 6, 26, 27); however this diversity has not yet been evaluated in populations isolated from crop residues. One approach that has been used successfully in many filamentous fungi to examine the population structure, clonality and potential gene flow is the study of vegetative incompatibility. Mycelial incompatibility assay at macroscopic level is a simple and reliable way to indicate a reaction of self or non-self-recognition and has been used in several fungal genera. Thus, the ability of strains from crop residues to recombine poses the danger of introducing virulence and/or toxigenic genes intolocal pathogen populations (16). Assessing genotypic diversity is therefore important to align plan protection to the existing and potentially changing pathogen population, especially where sexual reproduction play a role in the pathogen life cycle. An understanding of the genetic structure of F. graminaerum populations may provide insights into their epidemiology and evolutionary potential and may lead to improved strategies for their management (1).
The focus of studies on F. graminearum species complex in Argentina largely has been directed to wheat and maize since these species produce Fusarium head blight and ear or stalk rot, respectively (8). In soybean, reports about the pathogenicity of F. graminearum strains have been contradictory. In some cases, these strains were considered to be secondary colonizers of soybean seeds previously damaged by other fungi or by freezing (15, 22). At present different investigations in the America carried out in Brazil (18), United States (9, 12, 14) and Argentina (7, 25) have recognized to F. graminearum as a primary pathogen of soybean. Considering that the crop residues are the main source of maintenance of F. graminearum inoculum in the soybean agroecosystem, these populations can interact in the substrate and that they may can come into direct contact with the implanted seed and cause disease. For this reason, the active ingredients used in seed-treatment fungicides would be an important strategy to prevent diseases in soybean seedlings. Of these fungicides, fludioxonil was the only one that inhibited mycelial growth (9).
The objective of the present study was: to assess the genetic diversity through the mycelial compatibility of F. graminearum strains isolated from maize crop residues; and to analyze the pathogenicity of F. graminearum strains toward soybean seedlings from different soybean cultivars treated and untreated with fungicide.
MATERIALS AND METHODS
Evaluation of mycelial compatibility in F. graminearum strains from maize crop residues
Mycelial compatibility testing of 50 F. graminearum strains isolated from maize crop residues in a previous study in Cordoba Province (Argentina) (10), was carried out following the methodology described by Akinsanmi et al. (2008). The mycelial compatibility was evaluated on V8-soybean medium (150 ml V8 juice (Campbell's, UK), 20g agar, 20g of ground soybean seed and distilled water to a liter). A maximum of nine mycelial plugs of the isolates were placed equidistant from one another on agar medium in a single 90-mm Petri dishand incubated at 25°C for 7 days.
Two replicates of each isolate were paired with every other isolate in all possible combinations and themselves in a repeated experiment. A compatible reaction was indicated by mycelium continuity between the interacting colonies without a zone line, while an incompatible reaction was evidenced by the formation of a barrier between the interacting isolates (Figure 1a and 1b).

Figure 1. Compatibility (A) and incompatibility (B) reactions between Fusarium graminearum strains on V8-soybean medium. Multiple crosses among different Fusarium graminearum strains in V8-soybean plate (C).
Figura 1. Reacciones de compatibilidad (A) e incompatibilidad (B) entre cepas de Fusarium graminearum sobre medio V8-soja. Placa de medio V8-soja donde se observan múltiples cruzamientos entre distintas cepas de Fusarium graminearum (C).
Mycelial reactions for each strain were recorded in a binary matrix where 1 represented a compatible reaction and 0 an incompatible one. Compatibility was scored 1 because it requires a higher level of matching between mycelial compatibility than incompatibility, which can occur from differences at a few loci. The mycelial compatibility group data were transported to obtain a complete matrix.
The genetic similarity was estimated using the SinQual program (NTSYS.pc version 2.01), taking into account all pairs of strains tested according to Dice coefficient. The dendrogram of genetic similarity was constructed using the method of arithmetic means UPGMA and comparison program NTSYS matrices (NTSYS.pc version 2.01).
Pathogenicity assays
Out of 50 F. graminearum strains included in the compatibility analysis we arbitrary selected 10 strains to perform the pathogenicity tests. Two F. meridionale strains (F5043 y F5048) were included as positive control since were highly aggressive in a previous study on soybean seedlings (6). Three soybean cultivars were used in the present study: Nidera 5009, Nidera 5258 and Don Mario 5.1 i. The evaluation of seedlings height and disease severity was performed in a rolled-towel assay described by Xue et al. (2007). Soybean seeds were surface disinfected by soaking in 0.5% (v/v) NaOCl for 30 sec and rinsed twice in sterile distilled water.
Then disinfected seeds were spread evenly on two layers of sterilized paper towels moistened with 50 ml of sterilized water and covered with two layers of paper towels in plastic containers to allow the germination. Containers were covered with plastic lids and kept in darkness at 24°C for 3-4 days until plants were at the early VE growth stage and root hairs were visible. Visually healthy seedlings were selected and disinfected as described above. Seedlings were infested by transferring agar plugs (5 mm in diameter) with a sterilized metal needle from 7-day-old fungal cultures to the root-hair zone, about 1.5 cm behind the primary-root tip.
Control plants were inoculated with agar plugs from sterile PDA. Inoculated plants were each placed vertically on precut covering sheets consisting of two layers of the sterilized paper tissue laid on top of an aluminum foil sheet. The entire root along with the attached agar disc of an individual plant was then covered by folding the covering sheets in the middle. A growth unit for each plant was formed by further folding the covering sheets on the opening side.
The growth units were placed in plastic trays containing sterilized distilled water at a depth of 0.5 cm. The open top of the growth unit allowed for plant growth and the open bottom for root development and water absorption. The aluminum foil sheet was used to separate each unit and for moisture retention. The trays were placed in a growth chamber operated at 24°C with a 16 hour photoperiod of fluorescent light.
The water level in the tray was checked daily and the water was added as needed. Ten days after inoculation, plants were removed from the growth unit and visually assessed for root-rot severity. Symptoms were rated using a 0-4 scale (figure 2, page 152): 0, no visible disease symptoms; 1, lesion visible, but infection confined to the inoculation site, with normal seedling growth; 2, lesion size extended and the plant growth retarded; 3, infection of the entire root, and the plant growth halted; and 4, massive infection of the entire root resulting in plant death.

Figure 2. Ordinal scale used to evaluate disease severity.
Figura 2. Escala ordinal utilizada para evaluar la severidad de la enfermedad.
The experiments were repeated twice in independent way. In seedlings subjected to fungicide treatment, the seeds from the three cultivars were not superficially disinfected and were directly treated with the fungicide prior to inoculation of the fungal isolate using the above methodology for the untreated seed with fungicide.
The fungicide used was MAXIM®XL (Syngenta, Brazil) containing Fludioxonil (4- (2,2-difluoro-1,3-benzodioxol- 4-yl) -1H-pyrrole-3-carbonitrile) and metalaxyl - M (N- (2,6-dimethylphenyl) -N- (2'-methoxyacetyl) alanine methyl ester-D) as active agents. The application was performed according to the manufacturer's recommendation: per 100 g of seeds, 0.15 ml of fungicide and 0.35 ml of water were added.
Statistical analysis
Data from the two independent pathogenicity experiments were combined and analyzed as one.
Dependent variables seedling height and disease severity were subjected to an analysis of variance (ANOVA) and treatment means were separated by Fisher's least significant difference (LSD) test at a probability level of P<0.05 using the INFOSTAT software version 2012 (13). For disease severity, multifactorial analyses across soybean cultivar, isolates, fungicide treatment and their interactions were subjected to an analysis of variance (ANOVA)(13).
RESULTS AND DISCUSSION
During the 1990s, agriculture changed significantly in Argentina through the adoption of transgenic crops such as soybeans, maize and cotton under no-tillage system (23).
The adoption of this type of conservation tillage was a major change affecting the F. graminearum populations that can colonize easily crop residues of wheat, maize and soybeans (10, 24). Soybean is often part of a succession using wheat and other cereal crops in Argentina, therefore, F. graminearum strains colonizing soybean residues could provide the primary inoculum for infections wheat and maize and vice versa. For this reason, it was considered analyzing genetic diversity and pathogenicity of F. graminearum strains that colonize crop residues.
In filamentous fungi, the ability of self-recognition is essential for vegetative growth, sexual reproduction and defense against invading pathogens. Thus, the mycelial compatibility test can serve as a tool to measure the ability of self-recognition and in this study we used that tool to assess the genetic diversity and the potential of different genotypes to interact in the crop residues. A total of 1275 pairings were performed, representing all possible combinations between F. graminearum isolates, in which all isolates were self-compatible.
Regardless self-compatibility reactions, out of 1225 pairings between F. graminearums trains, 104 were compatible representing 8.4% of crosses (figure 3, page 154).

Figure 3. Cluster analyses of mycelial reactions of compatibility and incompatibility of the 50 Fusarium graminearum strains.
Figura 3. Análisis de cluster con base a las reacciones miceliales de compatibilidad/ incompatibilidad en las 50 cepas de Fusarium graminearum.
In Argentina, similar results were found by assessing the diversity of F. graminearum populations from wheat and soybean using VCG analysis (Vegetative Compatibility Groups) (26) and neutral molecular markers AFLPs(polymorphism amplified fragment length) (6), respectively. Other findings in the world have shown that none of F. graminearum populations are completely panmictic or clonal (2, 19, 32). Taking into account that the strains included in the mycelial compatibility analysis were isolated from maize crop residues recovered from a single field, the population showed a high degree of genetic diversity. This suggests that sexual recombination is probably the main factor affecting the genetic diversity of this population. Moreover, balancing selection between parasitic and saprophytic subpopulations can generate genetic variation.
Naef and Défago (20) compare the genetic diversity within a saprophytic field population from maize stubbles with a pathogenic population from wheat using SSR markers. The study found that genetic diversity of the saprophytic population was significantly higher than parasitic population. This result may partly explain the high genetic diversity found in this study considering the origin of our strains.
In the present study it was observed that mutually compatible isolates had very different patterns of compatibility with other isolates (figure 1c, page 150), and this fact suggest that many factors are involved in the control of compatibility and incompatibility reactions (2). However, we found the presence of “multi-compatible” strains (figure 3, page 154) that showed hyphal anastomosis with several others strains which could have implications for gene flow through somatic recombination, which could contribute to increase genetic diversity.
In the present study, the pathogenicity of 10 F. graminearum strains isolated from maize crop residues and 2 F. meridionale used in a previous study, were evaluate-dunder controlled conditions in seedlings from three soybean cultivars widely sown by growers in the Cordoba Province. The pathogenicity of each strain was assessed by two parameters: seedling height and severity in terms of root rot.
Regarding the seedling height, it was significantly reduced (P<0.05) by all strains compared to the control non-inoculated seedlings (table 1).
Table 1. Variation among Fusarium graminearum and Fusarium meridionale strains in relation to the growth parameters evaluated in the pathogenicity assays.
Tabla 1. Variabilidad en las cepas de Fusarium graminearum y Fusarium meridionale analizadas respecto de las variables altura de plántula y severidad de la enfermedad.
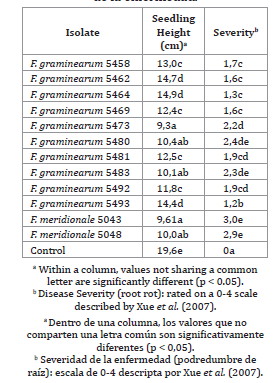
Reductions in the seedlings heights ranged from 24% to 52.5%. However, it was not observed in seedling height parameter significant differences among soybean cultivars and treatment with and without fungicides.
By the other hand, significant differences (P<0.001) were detected for cultivar, isolate, fungicide treatment and their interactions for disease severity. All genotypes tested showed symptoms of root rot with all the strains evaluated and were significantly different (P<0.05) respect to control treatment, in which non-inoculated seedlings remained healthy throughout the experience.
Don Mario 5.1i was the most susceptible cultivar, while the cultivar Nidera 5009 showed lower severity index with all strains tested (figure 4, page 156).

Figure 4. Differences in performance of the three soybean cultivars evaluated for the root rot severity.
Figura 4. Diferencias en el comportamiento de los tres cultivares evaluados respecto de la severidad en la podredumbre de raíz.
These results are not coincidental with those obtained by Xue et al. (2007), who showed no differences in behavior for root rot in three soybean cultivars widely used in Canada. According to these authors, the lack of differences between cultivars is not compatible with the notion that F. graminearum is pathogen to soybean in a strict sense, coinciding with other authors who considered to this fungus secondary colonizer of soybean seeds previously damaged by other fungi or by freezing (15, 22). However, in the present work we observed not only significant differences between cultivars, but also in the multifactorial statistical analysis, the greatest effect was marked by the cultivar effect, with a value F twice that the fungicide treatment (table 2, page 156).
Table 2. Analyses of variance of the disease severity of soybean cultivars inoculated with Fusarium graminearum strains.
Tabla 2. Análisis de la varianza para la variable severidad en los tres cultivares inoculados con cepas de Fusarium graminaerum.

The fact that F. graminearum generate differences in disease severity among cultivars makes us think that it could be a primary pathogen in roots of soybean seedlings as has been observed by other researchers in Brazil (18), United States (9, 12, 14) and Argentina (7, 25).
The strains included in the pathogenicity assay were scattered throughout the dendrogram (figure 3, page 154) and showed varying degrees of aggressiveness.
In both experiments, it was found less aggressive strains such as F. graminearum F5464 and highly aggressive strains as F. graminearum F5480 (figure 5) and the two F. meridionale F5043 and F5048 evaluated in previous studies using Don Mario 4613 cultivar (7).

Figure 5 Seedlings with different symptoms at the root level.
Figura 5 Las plántulas con diferentes síntomas a nivel de raíz.
However, in the multifactorial analysis it was observed that the isolate effect was significantly lower than the cultivar and fungicide treatment effect.
The mean disease severity averaged over the isolates evaluated in this study was approximately 2. This result indicates that the majority of the isolates, although showed low aggressiveness, produced a decrease in the seedling growth of that could influence the future crop production.
The value of mean disease severity found in the present study is similar to the average reported by Xue et al. (31) for Canadian wheat isolates evaluated on different soybean cultivars.
The seed treatment with fungicides is the most common practice for handling problems related to soil-borne, seeds and seedlings pathogens, including pathogenic Fusarium species (17). In this study, fungicide treatment of seed was carried out using fludioxonil, the active ingredient recommended to protect soybean against fungi belonging to the Fusarium genus (28).
In the present study, disease severity was affected by fungicide treatment, showing higher severity levels the seedlings not treated with the fungicide (figure 6).

Figure 6. Assessment of disease severity according to seed treatment with and without fungicide treatment.
Figura 6. Evaluación de la severidad de acuerdo con el tratamiento de la semilla con y sin fungicida curasemilla.
Similar results were obtained by Broders et al. (9) and Ellis et al. (14) who tested 4 and 6 fungicides respectively, in the treatment of soybean in the US and found that fludioxonil was more effective fungicide in preventing damage by F. graminearum.
CONCLUSIONS
Given that no-tillage system in wheat/ soybean or maize/soybean rotations has allowed an increase in the primary inoculum of F. graminearum, the use of fludioxonil as the active ingredient could be seen as a prevention tool to reduce seedling diseases by an emerging potential pathogen of soybean in Argentina. So far four species within the F. solani complex (F. virguliforme, F. tucumaniae, F. crassistipitatum and F. brasiliense) are considered the most important pathogens in causing sudden death syndrome and root rot of soybean Argentina (4). However, considering the present work, further studies are needed to determine the contribution of F. graminearum isolates in these soybean diseases in different agro-ecological zones showing high inoculum rates.
1. Akinsanmi, O. A; Backhouse, D.; Simpfendorfer, S.; Chakraborty, S. 2006. Genetic diversity of Australian Fusarium graminearum and F. pseudograminearum.Plant Pathol.55: 494–504.
2. Akinsanmi, O. A; Backhouse, D.; Simpfendorfer, S.; Chakraborty, S. 2008. Mycelial compatibility reactions of Australian Fusarium graminearum and F. pseudograminearum isolates compared with AFLP groupings. Plant Pathol. 57: 251-261. [ Links ]
3. Alvarez, C. L.; Somma, S.; Proctor, R. H.; Stea, G.; Mulè, G.; Logrieco, A.; Fernandez Pinto, V.; Moretti, A. 2011. Genetic diversity in Fusarium graminearum from a major Wheat-producing region of Argentina. Toxins 3: 1294-1309. [ Links ]
4. Aoki, T.; O’Donnell, K.; Scandiani, M. M. 2005. Sudden death syndrome of soybean in South America is caused by four species of Fusarium: Fusarium brasiliense sp. nov., F. cuneirostrum sp. nov., F. tucumaniae and F. virguliforme. Mycoscience 46:162-183.
5. Aoki, T.; O’Donnell, K.; Geiser, D. 2014. Systematics of key phytopathogenic Fusarium species: current status and future challenges. J. Gen. Plant Pathol. 80:189-201.
6. Barros, G.; Alaniz Zanon, M. S.; Abod, A.; Oviedo, S.; Ramirez, M. L.; Reynoso, M. M., Torres, A.; Chulze, S. 2012. Natural deoxynivalenol occurrence and genotype and chemotype determination of a field population of the Fusarium graminearum complex associated with soybean in Argentina. Food Add. Contam. 29: 293-303. [ Links ]
7. Barros, G.; Alaniz Zanon, M. S.; Chiotta, M. L.; Reynoso, M. M.; Scandiani, M. M.; Chulze, S. 2014. Pathogenicity of species in the Fusarium graminearum complex on soybean seedlings in Argentina. Eur. J. Plant Pathol. 138: 215-222. [ Links ]
8. Bottalico, A.; Perrone, G. 2002. Toxicogenic Fusarium species and mycotoxins associated with head blight in smaill-grain cereals in Europe. Eur. J. Plant Pathol. 108: 611-624. [ Links ]
9. Broders, K. D.; Lipps, P. E.; Paul, P. A.; Dorrance, A. E. 2007. Evaluation of Fusarium graminearum associated with corn and soybean seed and seedling in Ohio. Plant Dis. 91: 1155-1160. [ Links ]
10. Chiotta, M. L.; Chulze, S.; Barros, G. 2015. Inoculum sources of potential toxigenic Fusarium species in the soybean agroecosystem. Revista de la Facultad de Ciencias Agrarias. Universidad Nacional de Cuyo. Mendoza. Argentina. 47 (2): 171-184. [ Links ]
11. Chiotta, M. L.; Alaniz Zanon, M. S.; Giaj-Merlera, G.; Tessmann, D.; Barros, G.; Chulze, S. 2015b. Phylogenetic analyses of the Fusarium graminearum species complex isolated from soybean in Argentina and Brazil. Australasian Plant Dis. Notes 10:32. [ Links ]
12. Diaz Arias, M. M.; Leandro, L. F; Munkvold, G. P. 2013. Aggressiveness of Fusarium species and impact of root infection on growth and yield of soybeans. Phytopathology 103: 822-832. [ Links ]
13. Di Rienzo, J. A.; Casanoves, F.; Balzarini, M. G.; González, L.; Tablada, M.; Robledo, C.W. 2012. INFOSTAT. Grupo InfoStat, FCA, Universidad Nacional de Córdoba, Argentina. [ Links ]
14. Ellis, M. L.; Broders, K. D.; Paul, P. A.; Dorrance, A. E. 2011. Infection of soybean seed by Fusarium graminearum and effect of seed treatments on disease under controlled conditions. Plant Dis. 95: 401-407. [ Links ]
15. Jacobsen, B. J.; Harbin, K. S.; Swanson, S. P.; Lambert, R. J.; Beasley, V. R.; Sinclair, J. B.; Wei, L. S. 1995. Occurrence of fungi and mycotoxins associated with field mold damage soybeans in the Midwest. Plant Dis. 79: 86-88. [ Links ]
16. Leslie, J. F. 1993. Fungal vegetative compatibility. Annual Rev. Phytopathol. 31: 127-50. [ Links ]
17. Lipps, P. E.; Dorrance, A. E.; Milles, D. 2004. Corn disease management in Ohio. Ohio Agric. Res. Dev. Cent. Bull. 802. [ Links ]
18. Martinelli, J. A.; Bocchese, C. A. C.; Xie, W.; O’Donnell, K.;Kistler, H. C. 2004. Soybean pod blight and root rot caused by lineages of Fusarium graminearum and the production of mycotoxins. Fitopatol. Bras. 29: 492-498.
19. Monds, R. D.; Cromey, M. G.; Lauren. D. R.; di Menna, M.; Marshall, J. 2005. Fusarium graminearum, F. cortaderiae and F. pseudograminearum in New Zealand: molecular phylogenetic analysis, mycotoxin chemotypes and co-existence of species. Mycol. Res. 109: 410-20. [ Links ]
20. Naef, A.; Défago, G. 2006. Population structure of plant-pathogenic Fusarium species in overwintered stalk residues from Bt-transformed and non-transformed maize crops Eur. J. Plant Pathol. 116: 129-143. [ Links ]
21. Nocilli Pac, S. Estimación de superficie en siembra directa. Campaña 2014-2015. AAPRESID (Asociación Argentina de Productores en Siembra Directa). 9 p. Available in: www. aapresid.org.ar/superficie/ [ Links ]
22. Osorio, J. A.; McGee, D. C. 1992. Effect of freezing damage on soybean seed mycoflora and germination. Plant Dis. 76: 879-882. [ Links ]
23. Pengue, W. 2005. Transgenic crops in Argentina: the ecological and social debt. Bull. Sci. Tech. Soc. 25: 314-322. [ Links ]
24. Pereyra, S. A.; Dill-Macky, R. 2008. Colonization of the residues of diverse plant species by Gibberellazeae and their contribution to Fusarium head blight inoculum. Plant Dis. 92 (5): 800-807. [ Links ]
25. Pioli, R. N.; Mozzoni, L.; Morandi, E. N. 2004. First report of pathogenic association between Fusarium graminearum and soybean. Plant Dis. 88: 220. [ Links ]
26. Ramirez, M. L.; Reynoso, M. M.; Farnochi, M. C.; Chulze, S. 2006. Vegetative compatibility among Fusarium graminearum (Gibberellazeae) isolates from wheat spikes in Argentina. Eur. J. Plant Pathol. 115: 129-138. [ Links ]
27. Ramirez, M. L.; Reynoso, M. M.; Farnochi, M. C.; Torres, A. M.; Leslie, J. F.; Chulze, S. N. 2007. Population genetic structure of Gibberellazeae isolated from wheat in Argentina. Food Addit. Contam. 24: 1115-1120. [ Links ]
28. Scandiani, M. M. 2014. Tratamiento de la semilla de soja con curasemillas. Available in http:// www.rizobacter.com/assets/ensayos_rizobacter/10_tratamiento_con_fungicidas.pdf [ Links ]
29. Schmale, D. G.; Leslie, J. F.; Zeller, K. A.; Saleh, A. A.; Shields, E. J.; Bergstrom, G. C. 2006. Genetic structure of atmospheric populations of Gibberellazeae. Phytopathology 96: 1021-1026. [ Links ]
30. Shaner, G. E. 2003. Epidemiology of Fusarium Head Blight of small grain cereals in North America. In: Leonard, K. J., Bushnell, W. (Eds), Fusarium Head Blight of Wheat and Barley. APS Press. St. Paul. MN USA. 88-119. [ Links ]
31. Xue, A. G., Cober, E., Voldeng, H. D., Babcock, C.; Clear, R. M. 2007. Evaluation of the pathogenicity of Fusarium graminearum and Fusarium pseudograminearum on soybean seedlings under controlled conditions. Canadian J. Plant Pathol. 29: 35-40. [ Links ]
32. Zeller, K. A.; Vargas, J. I.; Valdovinos-Ponce, G.; Leslie, J. F.; Bowden, R. L. 2003. Population genetic differentiation and line age composition among Gibberellazeae (Fusarium graminearum) in north and South America. Fun. Gen. News. 50 (Suppl.):143. [ Links ]
ACKNOWLEDGEMENTS
This work was supported by grants from Secretaría de Ciencia y Técnica, Universidad Nacional de Río Cuarto and Agencia Nacional de Promoción Científica y Tecnológica (PICT 2457/11).














